Trematurid mites (Mesostigmata: Uropodina) associated with bark beetles (Coleoptera: Scolytinae) in Mexico
Chaires-Grijalva, Martha Patricia  1
; Estrada-Venegas, Edith Guadalupe
1
; Estrada-Venegas, Edith Guadalupe  2
; Equihua-Martínez, Armando
2
; Equihua-Martínez, Armando  3
; Moser, John C.4
; Blomquist, Stacy R.
3
; Moser, John C.4
; Blomquist, Stacy R.  5
; Pérez-Silva, Mauricio
5
; Pérez-Silva, Mauricio  6
; Arriola-Padilla, Víctor Javier
6
; Arriola-Padilla, Víctor Javier  7
; Rodríguez-Ortega, Alejandro
7
; Rodríguez-Ortega, Alejandro  8
and Kontschán, Jenő
8
and Kontschán, Jenő  9
9
1✉ Unidad Académica Multidisciplinaria Mante, Universidad Autonoma de Tamaulipas, Mexico.
2Programa de Entomología y Acarología. Colegio de Postgraduados. Campus Montecillo. Mexico.
3Programa de Entomología y Acarología. Colegio de Postgraduados. Campus Montecillo. Mexico.
4Alexandria Forestry Center, Forest Service. United States Department of Agriculture, USA. Deceased.
5Alexandria Forestry Center, Forest Service. United States Department of Agriculture, USA.
6Instituto de Biología, Universidad Nacional Autonoma de México. Mexico.
7Centro Nacional de Investigación Disciplinaria en Conservación y Mejoramiento de Ecosistemas Forestales, Instituto Nacional de Investigaciones Forestales Agrícolas y Pecuarias. Mexico.
8Ingeniería en Agrotecnología. Universidad Politécnica de Francisco I. Madero. Mexico.
9Széchenyi István University. Győr. Hungary.
2026 - Volume: 66 Issue: 2 pages: 364-376
https://doi.org/10.24349/qznl-uogsOriginal research
Keywords
Abstract
Introduction
Mites of the superfamily Uropodoidea are common inhabitants of soil and forest litter. They are predominantly found in microhabitats rich in decaying organic matter, such as decomposing plant material, manure, and the nests of insects, birds, and mammals (Błoszyk, 1999; Błoszyk et al., 2006; Gwiazdowicz et al., 2015; Karg, 1989; Mašán, 2001; Mašán and Krištofík, 1995; Wiśniewski and Hirschmann, 1993). This diverse group includes 47 families and over 300 currently recognized genera (Halliday, 2016, 2019). With a complete life cycle up to one year, the deutonymph stage is particularly critical for species persistence due to its remarkable resilience to adverse environmental conditions and its capacity for phoresy (Athias-Binche, 1984).
Phoretic interactions between mites and bark-dwelling insects have been documented by several authors (e.g., Gwiazdowicz et al., 2011, 2012; Moser, 1976; Pernek et al., 2012). Notably, some deutonymphs exhibit striking morphological adaptations for phoresy, most spectacularly exemplified by the presence of an anal pedicel. These modified deutonymphs attach to their insect host by pressing their slightly enlarged anal openings against a smooth surface on the host's body or legs. They then expel a sticky, liquid secretion that hardens upon air exposure, forming this pedicel for secure attachment (Evans, 1972).
These relationships can be opportunistic or exhibit strong host specificity, mediated by unique physical, chemical, and behavioral cues (Moser et al., 2005; Walter and Proctor, 2013). Trematurid mites are frequently found associated with or directly on bark beetles (Coleoptera: Curculionidae: Scolytinae) (Chaires-Grijalva, 2013; Chaires-Grijalva et al., 2016; Knee et al., 2012; Quiroz-Ibáñez et al., 2016), which provide the primary means of dispersal for deutonymphs. While the influence of mites on bark beetle populations is not yet fully understood, their high relative abundance and diversity suggest a potential impact on beetle dynamics and the physical condition of their hosts (Chaires-Grijalva, 2013; Halliday, 2019).
Download as Note: Species previously described under the genus Trichouropoda (e.g., T. ovalis, T. hirsuta) are here listed as Oodinychus following the classification of Kontschán et al. 2019.
Species
Host
Country
Reference
Oodinychus hirsuta (Hirschmann, 1972)
D. approximatus Dietz, D. brevicomis LeConte, D. frontalis Zimmerman, D.valens LeConte, Dryocoetes affaber Mannerheim, Ips avulsus Wood & Bright, I. calligraphus Germar, I. grandicollis Wood & Bright, I. pini Say & T., Hylastes porculus Erichson, H. pinifex Fitch, Polygraphus rufipennis Kirby
Canada, Mexico, USA
Moser & Roton 1971; Kinn, 1976; Hirschmann & Wiśniewski 1986; Kinn & Linit 1989; Hofstetter 2008; Knee et al. 2012a, b
Oodinychus karawaiewi (Berlese, 1904)
I. typographus (Linnaeus)
Central Europe, Slovakia, Russia
Berlese, 1904; Mašán, 2001; Khaustov et al. 2018
Oodinychus ovalis (Koch, 1839)
I. typographus (Linnaeus)
Norway, Poland
Kielczewski et al 1983; Gwiazdowicz et al. 2012
Oodinychus scolytanaKontschán et al. 2019
Polygraphus proximus Blanford
Russia
Kontschán et al. 2019
Trichouropoda alascae Hirschmann & Wisniewski, 1987+
Dendroctonus rufipennis Kirby
Alaska
Cardoza et al. 2008
Trichouropoda australis (Hirschmann, 1972) ++
Dendroctonus brevicomis LeConte, D. frontalis Zimmerman, D. ponderosae Hopkins, D. simplex LeConte, D. terebrans (Olivier), D. valens LeConte, I. avulsus Wood & Bright, I. bonanseai Hopkins, I. calligraphus Germar, I. grandicollis Wood & Bright, I. lecontei Swaine, I. perturbatus (Eichhoff), I. pilifrons Swaine, I. pini Say & T.
Canada, Honduras, Mexico, USA
Moser & Roton, 1971; Moser et al. 1974; Kinn, 1976; Kinn & Linit 1989; Hofstetter 2008; Knee et al. 2012b; Pfammatter et al. 2015
Trichouropoda bipilis (Vitzthum, 1921)
Hylesinus aculeatus Say, Scolytus multistriatus (Marsham), S. pygmaeus (Fabricius), S. amygdali Guerin
Austria, Canada, Iran
Hirschmann & WiÏniewski 1989; Ahadiyat et al. 2004; Moser et al. 2005; Knee et al. 2012b
Trichouropoda californica Wiśniewski & Hirschmann 1988 ++
Ips confusus LeConte, I. hoppingi Lanier
USA
Knee et al. 2012b
Trichouropoda dalarenaensis Hirschmann & Zirngiebl-Nicol, 1961 ++
Dendroctonus rufipennis (Kirby)
Alaska
Cardoza et al. 2008
Trichouropoda fallax (Vitzthum, 1926) +
Dendroctonus adjunctus Blandford, Hilastes ater Paycur, H. cunicularius Erichson, H. interstitialis Chapuis, H. ruber Swaine, Hylurgops pinifex Fitch, H. rugipennis Mannerheim
Canada, Belgica, Siberia, USA
Hirschmann & WiÏniewski, 1989; Knee et al. 2012a, b; Hofstetter 2008
Trichouropoda guatemalensis Hirschmann, 1972 ++
Dendroctonus frontalis Zimmermann
Guatemala
Moser et al. 1974
Trichouropoda idahoensis Hirschmann & Wiśniewski 1986
Ips pini Say & T.
USA
Knee et al. 2012b
Trichouropoda lamellosa Hirschmann, 1972 ++
Dendroctonus frontalis Zimmermann, D. ponderosae Hopkins, D. pseudotsugae Hopkins, Pityokteines curvidens Germar, P. spinidens Wood & Brigth, Hylastes porculus Erichson, D. valen LeConte, Ips avulsus Wood & Bright, I. calligraphus Germar, I. grandicollis Wood & Bright
Canada, Croatia, Honduras, Turkey, USA
Moser & Roton 1971; Moser et al. 1974; Kinn 1987; Kinn & Linit 1989; Pernek et al. 2008; Hofstetter 2008; Knee et al. 2012a, b; Cilbircioğlu et al. 2021
Trichouropoda moseri Hirschmann, 1972 ++
Dendroctonus simplex LeConte, Pityokteines sparsus LeConte, Polygraphus rufipennis Kirby
Canada
Knee et al. 2012b
Trichouropoda obscura (Koch, 1836) ++
Ips typographus (Linnaeus), Hylurgops palliatus Gyllenhal, Hylastes curnicularius Erichson
Central Europe
Kielczewski et al 1983; Zach et al. 2016
Trichouropoda parisiana Hirschmann & Wisniewski, 1987+
Ips sexdentatus Boerner, I. typographus (Linnaeus), Gnathotrichus materiarius Fitch
Canada, Francia
Hirschmann & WiÏniewski, 1987, Knee et al. 2012b
Trichouropoda pecinaituberosa Hirschmann & Wisniewski, 1987 ++
Hylastess cunicularius Erichson, H. palliatus Gyllenhal
Central Europe
Zach et al. 2016
Trichouropoda polonica Wisniewski & Hirschmann, 1988 +
Ips typographus (Linnaeus)
Norway, Poland
Gwiazdowicz et al. 2011, 2015
Trichouropoda polygraphi (Vitzthum, 1923) ++
Polygraphus sp.
Thailand
Knee et al. 2012b
Trichouropoda polytricha (Vitzthum, 1923) ++
Ips amitinus Eichhof, I. bonanseai Hopkings, I. borealis Swaine, I. cembrae (Heer), I. duplicatus Sahlberg, I. hunteri Swaine, I. montanus Eichhof, I. perturbatus (Eichoff), I. pilifrons Wood, I. typographus (Linnaeus), I. sexdentatus Boerner, I. woodi Thatcher, Pityogenes chalcographus (Linnaeus),Hylastes cunicularis Erichson Dryocoetes autographus Ratzerburg, Dentroctonus rhizophagus Thomas & Brigth.
Alaska, Austria, Bulgaria, Canada, Central Europe, Slovenia, Czech Republic, España, Finland, Germany, Norway, Poland, Romania, Russia, Serbia, Switzerland, Turkey, Turkmenistan, USA
Hirschmann & Zirngiebl-Nicol, 1961; Moser & Bogenschutz, 1984; Moser et al. 1989; Karg 1989; Kofler & Schmölzer 2000; Mašán 2001; Takov et al. 2009; feketeovà 2011;Gwiazdowicz& Gutwski 2012; Gwiazdowicz et al. 2012, 2015; Vrabec et al. 2012; Chaires-Grijalva et al. 2013; Fernández et al. 2013; Moraza et al. 2013; Penttinen et al. 2013; ČeJka & Holuša 2014; Zach et al. 2016; Khaustov et al. 2016, 2018; Kontschán et al. 2019; Paraschiv & Isaia, 2020; Milosavljević et al. 2022
Trichouropoda polytrichasimilis Hirschmann & Wisniewski, 1987 ++
Ips emarginatus Wood & Bright, Ips montanus Eichhoff, Ips pilifrons* Swaine
USA
Knee et al. 2012b
Trichouropoda sociata (Vitzthum, 1923) ++
Ips typographus (Linnaeus)
Norway
Gwiazdowicz et al. 2012
Trichouropoda structura Hirschmann & Zirngiebl-Nicol,1961 ++
Ips typographus (Linnaeus), Pytogenes chalcographus Linnaeus, Polygraphus proximus Brandford
Russia
Kiełczewski & Wiśniewski 1983; Hirschmann & Wiśniewski 1987; Karg 1989; Khaustov et al. 2018; Kontschán et al. 2019
Trichouropoda sp.
Ips typographus (Linnaeus)
Norway
Gwiazdowicz et al. 2012
Trichouropoda tegucigalpae Hirschmann & Wiśniewski 1986
Ips bonanseai Hopkings
Mexico
Knee et al. 2012b
Trichouropoda tuberosa Hirschmann & Zirngiebl-Nicol, 1961 ++
Polygraphus poligraphus Blanford
Central Europe
Zach et al. 2016
Trichouropoda vitzthumilongisetae Hirschmann & Wiśniewski 1987
Scolytidae
Central Europe
Hirschmann 1993, Kontschán et al. 2021
The family Trematuridae represents the primary group of phoretic mites associated with bark beetles. This family exemplifies the global nature of these associations, with 20 species recorded from 21 countries (Table 1). Their extensive distribution and phoretic associations underscore the ecological plasticity of these mites across diverse ecosystems. However, much of the foundational research on Uropodina has historically focused on temperate zones of Europe and North America. This geographic bias has created a significant knowledge gap regarding the diversity and ecology of phoretic Trematuridae within tropical and subtropical regions.
Our study, conducted across the diverse climatic range of Mexico, contributes to filling this void by documenting new records and attachment sites in the Neotropical and Nearctic transition zones. The main objective of this study is to assess the diversity of Trematuridae mites associated with bark beetles across various states in Mexico. To ensure taxonomic accuracy, we relied on recent comprehensive revisions (e.g., Halliday, 2016; Knee et al., 2012; Kontschán et al., 2019), particularly regarding the complex historical nomenclature established by Hirschmann and collaborators.
Materials and methods
Study area and sampling
Sampling was conducted across 24 Mexican states between 2009 and 2023. At each site, samples were obtained from both live and dead trees exhibiting signs of bark beetle infestation. The sampling effort covered Aguascalientes, Baja California, Ciudad de México, Chiapas, Chihuahua, Coahuila, Colima, Durango, Guanajuato, Hidalgo, Jalisco, México, Michoacán, Morelos, Nuevo León, Oaxaca, Puebla, Querétaro, San Luis Potosí, Sinaloa, Sonora, Tlaxcala, Veracruz, and Zacatecas. Bark and log samples were transported in plastic bags to the Forest Entomology Laboratory at Colegio de Postgraduados. Bark samples were examined under a Carl Zeiss Stemi DV4 stereomicroscope, while logs were placed in climate-controlled chambers to collect emerging adult insects, which were subsequently processed for phoretic mites. Detailed georeferenced coordinates for all collection sites are provided in the Results section.
Bark beetle identification
Bark beetles and associated arthropods were collected from bark and log samples using forceps and preserved in 70% ethanol. Specimens were identified using the taxonomic keys of Cibrián et al. (1995) and Wood (2007). Identifications were confirmed by Ph.D. Armando Equihua-Martínez and Mauricio Pérez-Silva.
Mite processing and identification
Phoretic mites were carefully removed from each beetle specimen and transferred to a lactic acid solution for clearing. Specimens were then mounted on glass slides using Hoyer's medium for microscopic examination. In addition to phoretic individuals, mites found within bark beetle galleries were also collected. The identification of Trichouropoda species was primarily conducted using the keys provided by Karg (1989), supplemented by specific original descriptions from the complex literature of Hirschmann and collaborators, and confirmed by the late co-author, Dr. John C. Moser†. It is noteworthy that several species formerly assigned to Trichouropoda, such as Oodinychus hirsuta and O. ovalis, have been reassigned following recent taxonomic revisions (e.g., Halliday, 2016; Kontschán et al., 2019). This study adopts these updated designations to maintain nomenclatural consistency with current uropodid systematics All mite specimens were deposited in the Acarological Collection at Colegio de Postgraduados (COLPOS).
Results
A total of 1,713 bark beetles belonging to nine species were examined. From these, 337 mites of the family Trematuridae (genus Trichouropoda) were collected: 178 were found phoretically attached to the insect bodies, 104 were recovered from the galleries, and 55 were retrieved from the alcohol sediments of the collection vials. Eleven species of Trichouropoda were identified.
Download as
D. adjunctus
D. frontalis
D. mexicanus
D. pseudotsugae
D. rhizophagus
D. valens
I. bonanseai
I. calligraphus
H. incomptus
Distribution
O. ovalis
1
-
-
-
19
21
-
-
-
Chih., EMex., Tlax.
T. adjuncti
6
-
-
-
-
4
-
-
-
Chih., Jal. and Ver.
T. australis
-
7
-
-
-
-
24
2
-
EMex, Jal.
T. fallax
-
-
8
-
-
-
-
-
-
Ver.
T. hondurasae
-
5
-
-
-
-
-
-
-
Qro.
T. polytricha
-
2
1
3
19
18
-
8
-
B.C, Chih., Jal., Mich., Pue., Qro., Ver.
T. sp. 1
-
7
-
-
-
2
-
-
-
Tlax., EMex.
T. sp. 2
-
-
-
-
6
-
-
-
-
Chih.
T. sp. 3
-
1
-
-
-
0
-
-
-
Hgo., EMex
T. sp. 4
-
-
-
-
-
-
7
Jal.
T. sp. 5
-
-
-
-
-
-
-
-
7
Ver.
Total Mites
6
22
9
3
25
24
24
10
14
Mite species
2
5
2
1
3
4
1
2
2
Total Bark beetles
237
146
572
83
172
246
117
76
64
Prevalence (%)
2.95
15.1
1.57
3.61
25.58
18.29
20.51
13.15
21.8
Out of the 24 Mexican states sampled, trematurid mites were recorded in ten: Baja California (BC), Chihuahua (Chih.), Estado de México (EMex), Hidalgo (Hgo.), Jalisco (Jal.), Michoacán (Mich.), Puebla (Pue.), Querétaro (Qro.), Tlaxcala (Tlax.), and Veracruz (Ver.) (Table 2).
The distribution of Trichouropoda species across host beetles varied significantly. The highest mite abundance was observed on Dendroctonus rhizophagus (n=44) and D. valens (n=45), which accounted for 25.58% and 18.29% of the total associations, respectively. In contrast, D. mexicanus showed the lowest proportion of associated mites (1.57%) despite having the largest sample size of beetles (n=572).
In terms of host specificity, Trichouropoda polytricha was the most generalist species, associated with six different bark beetle species (D. frontalis, D. mexicanus, D. pseudotsugae, D. rhizophagus, D. valens, and Ips confusus) across eight states. Conversely, species such as T. fallax and T. hondurasae were restricted to a single host and a single state in this study (Veracruz and Querétaro, respectively).
Furthermore, the genus Ips also hosted a considerable number of mites, with I. bonanseai showing a 20.51% association rate, primarily with T. australis (n=24).



Regarding overall species associations, five mite species were collected from two or more bark beetle hosts. In contrast, six species (T. fallax, T. hondurasae, and the undescribed T. sp. 2, T. sp. 3, T. sp. 4, and T. sp. 5) were associated with a single host species, although these were represented by only a few specimens each (Table 2; Figure 1).
Download as
Species
Mites on bark beetles
Fixation body site
Mites in galleries
Gallery type
Mites in alcohol
sediments
O. ovalis
41
Gz, Oe, Ed
27
Ng-H
16
T. adjuncti
10
Oe, Ed, Lg
2
Og-D
0
T. australis
33
As, Pr
14
Ng-H
0
T. fallax
8
Vs
5
Og-H
0
T. hondurasae
5
Oe
-
-
5
T. polytricha
51
Gz, Vs, Ed
40
Og-H
28
T. sp. 1
9
Gz, Pr
-
-
0
T. sp. 2
6
Vs
-
-
0
T. sp. 3
1
Oe
4
Og-H
2
T. sp. 4
7
Oe
12
Og-H
4
T. sp. 5
7
Vs
-
-
0
Total
178
104
55
Trichouropoda polytricha displayed no clear host preference, as was found on seven of the nine beetle species examined. In contrast, the remaining trematurid species exhibited minimal overlaps in host associations within this study; this suggests relative host specificity, despite previous literature reports characterizing them as generalists (Chaires-Grijalva et al., 2019). Most mites were observed on the ventral surface (29%), gular area (22%), abdominal sternites (18%), elytra (17%), elytral declivity (11%), legs (7%), and pronotum (2%) (Figure 2). Overall, mites were primarily detected on the host body and within the bark beetle galleries (Table 3).



Mites were found in diverse microhabitats associated with their hosts. Of the total specimens collected, 178 were found directly on the bark beetles, 104 within the galleries, and 55 in alcohol sediments. Regarding phoretic attachment, specific body sites were preferred depending on the mite species. For instance, Trichouropoda polytricha and Oodinychus ovalis (the most abundant species) were frequently located in the gular zone (Gz) and on the elytra (Oe). Other species, such as T. australis, showed a preference for abdominal sternites (As) and the pronotum (Pr). Additionally, mites were recovered from both new (Ng) and old (Og) galleries, primarily under humid (H) conditions.
Organisms examined
The specimens analyzed in this study were obtained from 24 states across Mexico between 2009 and 2023. A total of 1,713 bark beetles were examined, from which phoretic trematurid mites were recovered (Figure 3). For each species listed below, the data includes the specific locality, geographic coordinates (where available), collection date, host insect species, and the number of mite individuals (deutonymphs) collected. All specimens were collected by the authors and have been deposited in the Acarological Collection of the Colegio de Postgraduados (COLPOS). The taxonomic arrangement follows recent nomenclature, including the reassignment of certain species to the genus Oodinychus.
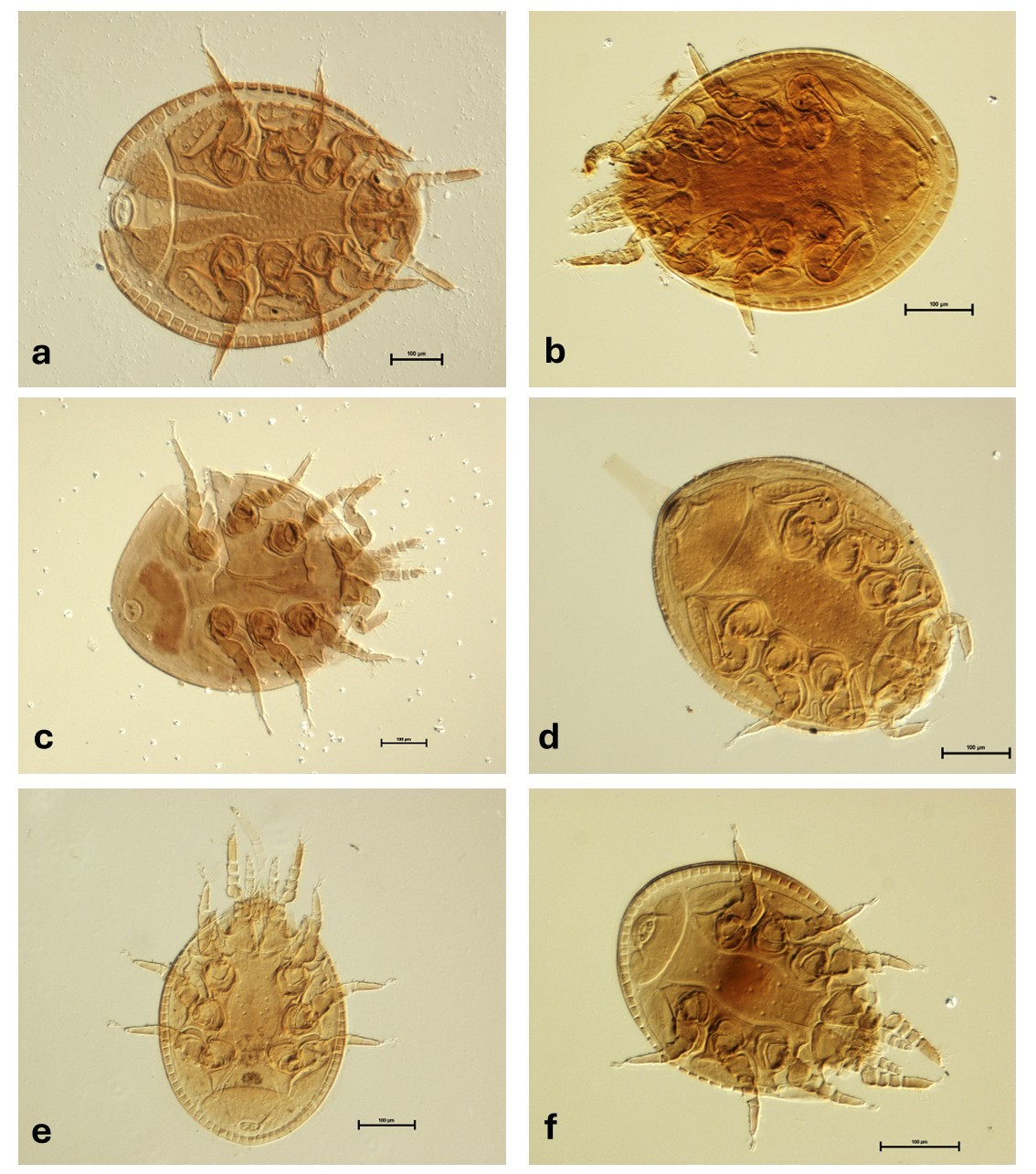


Oodynichus ovalis (Koch, 1939)
Notaspis ovalis Koch, 1839
Chihuahua: La Laja, Bocoyna, Pinus arizonica, 2452 masl, 27.931916 N, 107.5985 W; Jun. and Jul. 2008 to 2011, 19 DN ex. on gular zone of D. rhizophagus, 9 DN ex. in new and humid galleries of D. rhizophagus; Cuesta Prieta, Ejido San Juanito, Bocoyna, Pinus arizonica, 2450 masl, 27.930555 N, 107.595999 W, 16 DN ex. on gular zone of D. valens, 18 DN ex. galleries under bark. Estado de México: Experimental Station Zoquiapan, Universidad Autónoma de Chapingo, Ixtapaluca, P. hartwegii, 3600 masl. 19.282225 N, 98.670701 W, May 2006, 1 DN ex. on elytra of D. adjunctus. Tlaxcala: Highway Texcoco-Calpulalpan, 2842 masl., 19.5601027 N., 98.6944138 W., Jun. 16th, 2012. 5 DN ex. on elytral declivity of D. valens. colls. M.P. Chaires-Grijalva,
Trichouropoda adjuncti Wisniewski & Hirschmann, 1988
Chihuahua: Mesa de Parra, Municipality Madera, Pinus engelmannii, 2 481 masl, 29.204424 N, 108.184462 W, 1DN ex. elytral declive Dendroctonus valens. Jalisco: Rancho los Zuno, Gómez Farias, 2 200 msnm, 19.886659 N, 103.409291 W.; 3DN ex. on elytral declive of D. valens. Veracruz: Ejido Forestal Los Pescados, Municipality Perote; 3 128 masl. 19.528888 N, 97.123333 W; May 2004 to Apr. 2005; 4 DN ex. on elytra of D. adjunctus, 2 DN on legs and 2 DN ex. old and dry galleries of D. adjunctus. colls. M.P. Chaires-Grijalva, A. Rodríguez.
Trichouropoda australis Hirschmann, 1972
Estado de México: Experimental Station Zoquiapan, Universidad Autónoma de Chapingo, Municipality Ixtapaluca, P. hartwegii, 3600 masl. 19.282225 N, 98.670701 W; 7DN ex. on abdominals sternites of D. frontalis, 2 DN ex. new and humid galleries of D. frontalis; 24 DN ex. abdominal sternites of Ips bonanseai, 1♀, 12 DN ex. galleries under bark. Jalisco: Rancho los Zuno, Gómez Farias, 2200 msnm, 19.886659, 103.409291; 2DN ex. on abdominals sternites of Ips calligraphus, coll. M.P. Chaires-Grijalva.
Trichouropoda fallax (Vitzthum, 1926)
Trichoobscura fallax Hirschmann & Zirngiebl-Nicol, 1961
Uropoda fallax Vitzthum, 1926
Estado de México: Experimental Station Zoquiapan, Universidad Autónoma de Chapingo, Municipality Ixtapaluca, P. hartwegii, 3600 masl. 19.282225 N, 98.670701 W, May 2006, 2♀ ex. old and humid galleries of D. adjunctus. Veracruz: Ejido Forestal Los Pescados, Municipality Perote; P. hartwegii, 3128 masl. 19.528888 N, 97.123333 W; May 2004 to Apr. 2005; 8 DN ex. on elytra of D. mexicanus, 3 DN ex. galleries of D. mexicanus.
Trichouropoda hondurasae (Hirschmann & Wisniewski, 1986)
Ipiduropoda hondurasae Hirschmann & Wisniewski, 1986
Querétaro: Rio Blanco, Municipio de Peñamiller, 1720 masl, 21.208056 N, 99.737500 W; Nov 26th, 2010; 5 DN ex. on elytra of D. frontalis.
Trichouropoda polytricha (Vitzthum, 1923)
Ipiduropoda polytricha Hirschmann 1986
Uropoda polytricha Vitzthum, 1923
Baja California: Parque Nacional Sierra de San Pedro Mártir, Ensenada. Pinus jeffreyi, 2830 masl, 31.027794 N, 115.467846 W., Sept. 2006, 3 DN ex. ventral sternites of D. pseudotsuga. Chihuahua: La Laja, Bocoyna, P. arizonica, 2452 masl, 27.931916 N, 107.5985 W.; Jun. and jul. 2008 to 2011, 8 DN ex. on ventral sternites of D. rhizophagus; Cuesta Prieta, Ejido San Juanito, Bocoyna, P. arizonica, 2450 masl, 27.930555 N, 107.595999 W., 5 DN ex. on gular zone of D. rhizophagus; Mesa de Parra, Madera, P. arizonica, 2300 masl., 29.187027, -108.171007, 6 DN ex. on ventral sternites of D. rhizophagus. Jalisco: Parque Nacional Nevado de Colima, San Gabriel, Pinus hartwegii, 3300 masl, 19.626806, 103.562139, 8 DN ex. on elytral declivity of Ips calligraphus. Michoacán: Zirahuén Lake, Salvador Escalante, Pinus patula, 2187 masl, 19.466111 N, 101.736336 W, Jan. 14th, 2010, 13 DN ex. on ventral sternites of D. valens, 3♀, 1♂, 27 DN, 9 PN ex. old and humid galleries of D. valens. Puebla: El puerto, Tetela de Ocampo, P. patula, 2120 masl, 19.805075 N, 97.779233 W., 1 DN ex. on ventral sternites of D. frontalis. Querétaro: Rio Blanco, Peñamiller, P. patula, 1720 masl, 21.208056 N, 99.737500 W; Nov 26th 2010; 1 DN ex. on ventral sternites of D. mexicanus. Veracruz: Ejido Forestal Los Pescados, Perote; P. hartwegii, 3128 masl. 19.528888 N, 97.123333 W; May 2004 to Apr. 2005; 1 DN ex. on ventral sternites of D. frontalis, 5 DN ex. on ventral sternites of D. valens. colls. M.P. Chaires-Grijalva, A. Rodríguez.
Trichouropoda sp. 1
Estado de México: Experimental Station Zoquiapan, Universidad Autónoma de Chapingo, Ixtapaluca, P. patula, 3600 masl. 19.282225 N, 98.670701 W; 7 DN ex. on elytra of D. frontalis. Tlaxcala: Highway Texcoco-Calpulalpan, 2842 masl., 19.5601027 N., 98.6944138 W., Jun. 16th, 2012, 2 DN ex. on elytral declivity of D. valens.
Trichouropoda sp 2
Chihuahua: La Laja, Bocoyna, P. arizonica, 2452 masl, 27.931916 N, 107.5985 W.; Jun. and Jul. 2008 to 2011, 6 DN ex. on ventral sternites of D. rhizophagus*.
Trichouropoda sp 3
Estado de México: Experimental Station Zoquiapan, Universidad Autónoma de Chapingo, Ixtapaluca, P. patula, 3600 masl. 19.282225 N, 98.670701 W, May 2006, 1 DN ex. on elytra of D. frontalis. Hidalgo: Presa el Tejocotal, Acaxochitlan, 2260 masl., 20.133055 N, 98.129749 W, Oct 10th, 2022; 4 DN ex. old and humid galleries of D. valens.
Trichouropoda sp 4
Jalisco: Rancho los Zuno, Gómez Farias, 2200 msnm, 19.886659 N, 103.409291W, Nov 13th, 2023; 12 DN ex. in old and humid galleries of Ips calligraphus, coll. M.P. Chaires-Grijalva; 7 DN ex. on elytra of Hylurgops incomptus.
Trichouropoda sp 5
Veracruz: Ejido Forestal Los Pescados, Perote; P. hartwegii, 3128 masl. 19.528888 N, 97.123333 W; May 2004 to Apr. 2005, 7 DN ex. on ventral surface of Hylurgops incomptus.
Discussion
Biogeography and host plasticity. Six of the eleven mite species identified in this study are also present in Europe and North America, suggesting a high capacity for co-invasion with their host insects. This distribution pattern likely stems from the native status of both beetles and mites across these continents. For instance, Trichouropoda australis has been documented on 17 species of Dendroctonus and Ips across the United States and Canada (Hofstetter, 2008; Knee et al., 2012), while T. fallax is known to associate with seven species of Dendroctonus, Hylastes, and Hylurgops in Europe and North America (Hirschmann and Wiśniewski, 1989; Knee et al., 2012).
Notably, these mites are not obligate associates of bark beetles. O. ovalis has been recorded in diverse habitats, including bird nests in Poland and Scandinavia (Błoszyk et al., 2006), reflecting broad ecological plasticity. Similarly, T. polytricha (the most generalist species in our study, found on seven different bark beetle species) is a cosmopolitan mite previously recorded in Mexico (Moser et al., 1989). Its success across eight Mexican states suggests that a lack of host-sex preference and high host plasticity broaden the range of available carriers, increasing the probability of successful dispersal and colonization of new habitats (Bajerlein et al., 2023).
Mechanisms of phoresy and attachment sites. The selection of specific attachment sites by phoretic deutonymphs is a strategic adaptation to minimize active eviction by the host or passive dislodgment during flight (Chaires-Grijalva, 2013). Unlike most mites, uropodines utilize a flexible and resistant anal pedicel for anchorage, significantly reducing the likelihood of dislodgment (Bajerlein et al., 2013). Our results show a preference for the ventral surface (29%) and the gular area (22%), consistent with findings by Hunter (1993), who noted that uropodids typically attach to areas posterior to coxa III.
These sites likely offer protection from the beetle's grooming behavior. However, when primary sites become saturated, deutonymphs adhere to other areas, such as the elytra or pronotum. We observed that uropodine deutonymphs tend to occupy setae-uncoated body parts not yet colonized by other mite taxonomic groups (Bajerlein et al., 2013). It is worth noting that some mites found in alcohol sediments likely originated from these sites but were dislodged during sampling, a common limitation in phoretic studies.
The gallery as a micro-ecosystem. Mites found within bark beetle galleries are integral components of the associated fauna. Each gallery represents a complex micro-ecosystem where mites find essential resources, including nematodes, fungi, and bark beetle eggs or larvae (Moser, 1975; Kontschán et al., 2019). Humid, newly formed galleries support the mites' entire biological cycle by providing stable temperatures and refuge from natural enemies (Chaires-Grijalva et al., 2013).
The high prevalence observed in Dendroctonus rhizophagus (25.58%) compared to D. mexicanus (1.57%) is particularly striking. This disparity may be linked to the specialized biology of D. rhizophagus, which attacks the roots and base of seedlings; the increased humidity and relative stability of the subterranean environment may favor higher mite densities and more successful phoretic transfers compared to the more exposed subcortical environments of trunk-attacking species like D. mexicanus. According to Wilson's (1980) association/predation index, a closer phoretic relationship often correlates with a reduced predatory threat to the host (Moser, 1995). Consequently, the high abundance of certain Trichouropoda species suggests a well-established symbiotic equilibrium within the Mexican conifer forests.
Conclusions
The present study reveals a significant diversity of trematurid mites associated with bark beetles in Mexico, identifying eleven species within the genera Trichouropoda and Oodinychus. The presence of six species previously recorded in Europe and North America highlights a broad biogeographic distribution and suggests a robust capacity for co-invasion alongside their host insects.
Our findings demonstrate two distinct ecological strategies among the identified mites:
- Host Plasticity: T. polytricha stands out as a highly successful generalist, exhibiting no clear host preference and a wide geographic range across ten Mexican states.
- Specialization and microhabitat: Most species showed restricted host associations and specific phoretic attachment sites, particularly in protected areas like the gular zone and ventral surface. These locations are likely selected to minimize dislodgment and avoid host grooming behaviors.
Furthermore, the high prevalence observed in D. rhizophagus (25.58%) underscores the importance of the host's specific ecology—such as its subterranean habitat—in determining mite population densities. The consistent presence of these mites within both new and old galleries confirms that these structures serve as essential micro-ecosystems, providing the humidity, stable temperature, and food resources (nematodes, fungi, and larvae) necessary for the mites' entire biological cycle.
In conclusion, this research provides a fundamental baseline for understanding the symbiotic networks within Mexico's coniferous forests. Future studies should focus on the functional role of these mites—whether as commensals, predators, or competitors—to better assess their impact on bark beetle population dynamics and forest health.
Acknowledgements
To Dr. Guillermo Sánchez Martínez (INIFAP Campus Pabellón) and, Dr. Gerardo Zúñiga Bermúdez (IPN ENCB) for their help in providing the biological material for this study.
References
- Ahadiyat A., Ostovan H. 2006. Observations on the biology of Winterschmidtia hamadryas (Vitzthum) (Acari: Astigmata: Winterschmidtiidae) associated with the almond bark beetle, Scolytus amygdali Guerin-Meneville (Coleoptera: Scolytidae) in Iran. Belg. J. Entomol., 8 (2):153-164.
- Ahadiyat A., Ostovan H., Saboori A. 2004. Mites associated with Scolytus amygdali Guerin-Meneville, 1847 in Karaj region. 16th Iranian Plant Protection Congress, Tabriz, Iran p. 251.
- Athias-Binche F. 1984. La phorésie chez les Acariens Uropodides (Anactinotriches), une stratégie écologique originale. Acta Oecologica, 5:119-133.
- Bajerlein D., Błoszyk J., Halliday B., Konwerski S. 2023. Hitchhiking through life: a review of phoresy in Uropodina mites (Parasitiformes: Mesostigmata). Eur. Zool. J., 91(1): 31-63. https://doi.org/10.1080/24750263.2023.2288847
- Bajerlein D., Witaliński W., Adamski Z. 2013. Morphological diversity of pedicels in phoretic deutonymphs of Uropodina mites (Acari: Mesostigmata). Arthropod Struct. Dev., 42(3), 185-196. https://doi.org/10.1016/j.asd.2013.02.002
- Błoszyk J. 1999. Geographical and ecological variability of mites of the cohort Uropodina (Acari: Mesostigmata) in Poland. I. Uropodina mites of oak-hornbeam forests (Carpinion betuli). Wydawnictwo Kontekst, Poznań, Poland. 245 pp.
- Błoszyk J., Bajerlein D., Gwiazdowicz D.J., Halliday R.B., Dylewska M. 2006. Uropodine mite communities (Acari: Mesostigmata) in birds' nests in Poland. Belg. J. Zool.,136: 145-153.
- Cardoza Y.J., Moser J.C., Klepzig K.D., Raffa K.F. 2008. Multipartite symbioses among fungi, mites, nematodes, and the spruce beetle, Dendroctonus rufipennis. Environ. Entomol., 37(4): 956-963. https://doi.org/10.1093/ee/37.4.956
- Čejka M., Holuša J. 2014. Phoretic mites (Mesostigmata) on double-spined spruce bark beetle Ips duplicatus (Coleoptera: Curculionidae: Scolytinae) in recent oubreak area in the central Europe. Lesn. Čas. For. J., 60: 240-243. https://doi.org/10.1515/forj-2015-0006
- Chaires-Grijalva M.P. 2013. Mesostigmados (Acari: Mesostigmata) asociados a Scolytinae (Coleóptera: Curculionidae) de importancia forestal en México. [Phd Thesis] Colegio de Postgraduados. Campus Montecillo. Pp. 249. Avalaible in: http://colposdigital.colpos.mx:8080/jspui/bitstream/handle/10521/2158/Chaires_Grijalva_MP_DC_Entomologia_Acarologia_2013.pdf
- Chaires-Grijalva M.P., Estrada-Venegas E.G., Equihua-Martínez A., Moser J.C., Blomquist S.R. 2016. Trophic habits of mesostigmatid mites associated with bark beetles in Mexico. Journal of Acarology Society of Japan 25(S1): 161-167. https://doi.org/10.2300/acari.25.Suppl_161
- Chaires-Grijalva M.P., Estrada-Venegas E.G., Quiroz-Ibáñez I.F., Equihua-Martínez A., Moser J.C., Blomquist S.R. 2019. Acarine biodiversity associated with bark beetles in Mexico. Acarological Studies, 1(2): 152-160. https://izlik.org/JA64EL47MN
- Chaires-Grijalva M.P., Estrada-Venegas E.G., Equihua-Martínez A., Moser J. C., Sánchez-Martínez G., Vázquez-Rojas I.M., Otero-Colina G., Romero-Nápoles J. 2013. Mesostigmados (Acari: Mesostigmata) asociados con Dendroctonus rhizophagus de Chihuahua, México. Rev. Mex. Biodivers., 84(4): 1235-1242. https://doi.org/10.7550/rmb.35723
- Cibrián T.D., Méndez M.J.T., Bolaños R.C., Yates, III H.O., Lara J.F. 1995. Insectos Forestales de México. Universidad Autónoma Chapingo. México. Pp.453.
- Cilbircioğlu C., Kovač M., Pernek M. 2021. Associations of phoretic mites on bark beetles of the genus Ips in the black sea mountains of Turkey. Forests, 12(5), 516. https://doi.org/10.3390/f12050516
- Evans G.O. 1972. Leg chaetotaxy and the classification of the Uropodina (Acari: Mesostigmata). J. Zool., 167(2): 193-206. https://doi.org/10.1111/j.1469-7998.1972.tb01729.x
- Fernández M., Diez J., Moraza M.L. 2013. Acarofauna associated with Ips sexdentatus in northwest Spain. Scand. J. For. Res., 28(4): 358-362. https://doi.org/10.1080/02827581.2012.745897
- Gwiazdowicz D.J., Błoszyk J., Gdula A.K. 2015. Alpha diversity of mesostigmatid mites associated with the bark beetle Ips typographus (L.) in Poland. Insect Conserv. Divers., 8(5): 448-455. https://doi.org/10.1111/icad.12122
- Gwiazdowicz D.J., Kamczyc J., Błoszyk J. 2011. The diversity of phoretic Mesostigmata on Ips typographus (Coleoptera: Scolytinae) caught in the Karkonosze forest. Eur. J. Entomol., 108: 489-491. https://doi.org/10.14411/eje.2011.063
- Gwiazdowicz D.J., Kamczyc J., Teodorowicz E., Błoszyk J. 2012. Mite communities (Acari, Mesostigmata) associated with Ips typographus (Coleoptera, Scolytidae) in managed and natural Norway spruce stands in Central Europe. Cent. Eur. J. Biol., 7(5): 910-916. https://doi.org/10.2478/s11535-012-0070-z
- Halliday R.B. 2016. Catalogue of families and their type genera in the mite suborder Uropodina (Acari: Mesostigmata). Zootaxa, 4061(4), 347-366. https://doi.org/10.11646/zootaxa.4061.4.2
- Halliday R.B. 2019. The enemy of my parasite is my friend: the possible role of predatory mites as biological control agents of pest beetles in soil. Int. J. Acarol., 45(4): 189- 196. https://doi.org/10.1080/01647954.2019.1574895
- Hirschmann W., Wiśniewski J. 1989. Gangsystematik der Parasitiformes Teil 507-508: Weltweite Revision der Gattung Trichouropoda Berlese 1916 Teil IV. Acarologie, 36: 1-72.
- Hofstetter R.W. 2008. Information and images of mites associated with bark beetles and their predators. ( http://oak.ucc.nau.edu/rh245/MiteImages.htm)
- Hunter P.E. 1993. Mites associated with new world passalid beetles (Coleoptera: Passalidae). Instituto de Ecología. Acta Zool. Mex. (N.S.), 58: 1-37. https://doi.org/10.21829/azm.1993.55581766
- Karg W. 1989. Acari (Acarina), Milben Unterordnung Parasitiformes (Anactinochaeta) Uropodina Kramer, Scheldkrötenmilben. Fischer G., Verlag J. (Eds). Pp. 203.
- Karg W. 1993. Acari (Acarina), Milben. Parasitiformes (Anactinochaeta), Cohors Gamasina Leach, Raubmilben. In: Fischer G., Verlag J. (Eds). Second revised ed. Die Tierwelt Deutschlands, Vol 59. Raubmilben, Germany:
- Khaustov A.A., Klimov P.B., Trach V.A., Bobylev A.N., Salavatulin V.M., Khaustov V.A., Tolstikov A.V. 2018. Review of mites (acari) associated with the European spruce bark beetle, Ips typographus (Coleoptera: Curculionidae: Scolytinae) in Asian Russia. Acarina, 26 (1): 3-79. https://doi.org/10.21684/0132-8077-2018-26-1-3-79
- Kiełczewski B., Wiśniewski J. 1983. Bark Beetle acarofauna in different types of forest habitat. I, II. Introduction and Mesostigmata. Folia For. Pol. A. Leśnictwo, 25: 129-162.
- Kinn D.N., Linit M.J. 1989. A key to phoretic mites commonly found on long-horned beetles emerging from southern pines. United States Department of Agriculture, Forest Service, Southern Forest Experiment Station, 357: 1-8. https://doi.org/10.2737/SO-RN-357
- Kinn D.N. 1976. Key to mites commonly associated with the southern pine beetle. Note SO-214. New Orleans, LA. U.S. Department of Agriculture, Forest Service, Southern Forest Experiment Station. pp. 11.
- Knee W., Beaulieu F., Skevington J.H, Kelso S., Forbes M.R. 2012b. Cryptic species of mites (Uropodoidea: Uroobovella spp.) associated with burying beetles (Silphidae: Nicrophorus): The collapse of a host generalist revealed by molecular and morphological analyses. Mol. Phylogenetics Evol., 65(1):276-286. https://doi.org/10.1016/j.ympev.2012.06.013
- Knee W., Beaulieu F., Skevington J.H., Kelso S., Cognato A.I., Forbes M.R., Shaw P. 2012a. Species boundaries and host range of tortoise mites (Uropodoidea) phoretic on bark beetles (Scolytinae), using morphometric and molecular markers. PloS One 7(10):e47243. https://doi.org/10.1371/journal.pone.0047243
- Knee W., Forbes M.R., Beaulieu F. 2013. Diversity and host use of mites (Acari: Mesostigmata, Oribatida) phoretic on bark beetles (Coleoptera: Scolytinae): Global generalists, local specialists? Ann. Entomol. Soc. Am.,106(3):339-350. https://doi.org/10.1603/AN12092
- Knee W., Hartzenberg T., Forbes M.R., Beaulieu F. 2012c. The natural history of mites (Acari: Mesostigmata) associated with the white-spotted sawyer beetle (Coleoptera: Cerambycidae): Diversity, phenology, host attachment, and sex bias. Can. Entomol., 144(5):711-719. https://doi.org/10.4039/tce.2012.57
- Kofler A., Schmölzer K. 2000. Zur Kenntnis phoretischer Milbenund ihrer Tragwirte in Österreich (Acarina: Gamasina, Uropodina). Ber. Naturwiss.-med. Ver. Innsb., 87:133-157
- Kontschán J., Szőcs G., Kiss B., Khaustov A.A. 2019. Bark beetle associated trematurid mites (Acari: Uropodina: Trematuridae) from Asian Russia with description of a new species. Syst. Appl. Acarol., 24(9): 1592-1603. http://doi.org/10.11158/saa.24.9.2 https://doi.org/10.11158/saa.24.9.2
- Manu M., Poliză D., Onete M. 2017. Comparative analysis of the phoretic mites communities (Acari: Mesostigmata) associated with Ips typographus from natural and planted Norway spruce stands - Romania. Rom. Biotechnol. Lett.,10: 1-9.
- Mašán P., Krištofík J. 1995. Mesostigmatid mites (Acarina: Mesostigmata) in the nests of penduline tit (Remiz pendulinus). Biologia, 50 (5): 481-485.
- Mašán P. 2001. Mites of the Cohort Uropodina (Acarina, Mesostigmata) in Slovakia. Annot. Zool. Bot., 223: 1-321.
- Milosavljević M., Tabaković-Tošić M., Pernek M., Rakonjac L., Lučić A., Eremija S., Rindos M. 2022. Mites associated with the european spruce bark beetle Ips typographus (Linnaeus, 1758) in europe, with new evidence for the fauna of serbia. Forests, 13(10): 1586. https://doi.org/10.3390/f13101586
- Moraza M.L., Fernández M., Jurc M. 2013. Phoretic mites of the six-spined engraver beetle, Ips sexdentatus (Böerner, 1776) (Coleoptera, Scolytinae), on Pinus halepensis in Slovenia. Int. J. Acarol., 39(8): 597-599. https://doi.org/10.1080/01647954.2013.867900
- Moser J.C. 1975. Mite predators of the southern pine beetle. Ann. Entomol. Soc. Am., 68(6):1163-1116. https://doi.org/10.1093/aesa/68.6.1113
- Moser J.C. 1976. Phoretic carrying capacity of flying southern pine beetles (Coleoptera: Scolytidae). Can. Entomol., 108(8): 807-808. https://doi.org/10.4039/Ent108807-8
- Moser J.C. 1995. Mites associated with forest insects. Willamette Institute for Biological Control, Inc. Monroe, Oregon. 52 p.
- Moser J.C., Roton L.M. 1971. Mites associated with southern pine bark beetles in Allen Parish, Louisiana. Can. Entomol., 103(12): 1775-1798. https://doi.org/10.4039/Ent1031775-12
- Moser J.C., Konrad H., Kirisits T., Carta L.K. 2005. Phoretic mites and nematode associates of Scolytus multistriatus and Scolytus pygmaeus (Coleoptera: Scolytidae) in Austria. Agric. For. Entomol., 7: 169-177. https://doi.org/10.1111/j.1461-9555.2005.00261.x
- Moser J.C., Wilkinson R., Clark E.W. 1974. Mites associated with Dendroctonus frontalis Zimmerman (Scolytidae: Coleoptera) in Central America and Mexico. Turridbiz, 24(4): 373-381. https://repositorio.catie.ac.cr/handle/11554/12975
- Moser J.C., Bogenschutz H. 1984. A key to the mites associated with flying Ips typographus in South Germany. Z. Angew. Entomol., 97: 437-450. https://doi.org/10.1111/j.1439-0418.1984.tb03774.x
- Moser J.C., Perry T. J., Solheim H. 1989. Ascospores hyperphoretic on mites associated with Ips typographus. Mycol. Res., 93(4): 513-517. https://doi.org/10.1016/S0953-7562(89)80045-0
- Ostovan H., Kamali K. 1997. Biodiversity of mites (Acari) associated with elm bark beetle Scolytus multistriatus (Marsh.) (Col.: Scolytidae) in Parks of Tehran. J. Agric. Sci., 3 (11y 12): 3-48.
- Paraschiv M., Isaia G. 2020. Disparity of phoresy in mesostigmatid mites upon their specific carrier Ips typographus (Coleoptera: Scolytinae). Insects, 11(11), 771. https://doi.org/10.3390/insects11110771
- Penttinen R., Viiri H., Moser J.C. 2013. The mites (Acari) associated with bark beetles in the Koli National Park in Finland. Acarologia, 53(1): 3-15. https://doi.org/10.1051/acarologia/20132074
- Pernek M., Hrasovec B., Matosevic D., Pilas I., Kirisits T., Moser J.C. 2008. Phoretic mites of three bark beetles (Pityokteines spp.) on silver fir. J. Pestic. Sci., 81: 35-42. https://doi.org/10.1007/s10340-007-0182-9
- Pernek M., Wirth S., Blomquist S.R., Avtzis D.N., Moser J.C. 2012. New associations of phoretic mites on Pityokteines curvidens (Coleoptera, Curculionidae, Scolytinae). Cent. Eur. J. Biol., 7: 63-68. https://doi.org/10.2478/s11535-011-0096-7
- Pfammatter J. A., Raffa K. F. 2015. Do phoretic mites influence the reproductive success of Ips grandicollis (Coleoptera: Curculionidae)? Environ. Entomol., 44(6): 1498-1511. https://doi.org/10.1093/ee/nvv125
- Pfammatter J. A., Coyle D.R., Gandhi K.J.K., Hernandez N., Hofstetter R.W., Moser J.C., Raffa K.F. 2015. Structure of phoretic mite assemblages across subcortical beetle species at a regional scale. Environ. Entomol., 1-13. https://doi.org/10.1093/ee/nvv150
- Quiroz-Ibáñez I.F., Estrada-Venegas E.G., Campos-Bolaños R., Chaires- Grijalva M.P. 2016. Ácaros foréticos asociados a Dendroctonus frontalis Zimmerman, 1868 (Coleoptera: Curculionidae: Scolytinae) colectados con trampas multiembudo Tipo Lindgren en Tlaxco, Hidalgo. Entomologia mexicana, 3: 110-114.
- Walter D.E., Proctor H.C. 2013. Mites: ecology, evolution and behavior. Life at a microscale (2nd ed.), Springer, New York. https://doi.org/10.1007/978-94-007-7164-2
- Wilson D.S. 1980. The natural selection of populations and communities. Benjamin/Cummins, Menlo Park, California.
- Wiśniewski J., Hirschmann W. 1993. Gangsystematik der Parasitiformes, Teil 548. Katalog der Ganggattungen, Untergattungen, Gruppen und Artender Uropodiden der Erde. Acarologie. Schriftenreihe für Verglichende Milbenkunde, 40:1-220.
- Wiśniewski J. 1993. Die Uropodiden der Erde nach Zoogeographischen Regionen und Subregionen geordnet (Mit Angabe der Lande). Acarologie, 40:221- 291.
- Wood S.L. 2007. Bark and ambrosia beetles of South America (Coleoptera: Scolytidae). Monte L. Bean Sci. Mus., Provo, Utah. pp.900.
- Zach P., Kršiak B., Kulfan J., Parák M., Kontschán J. 2016. Mites Trichouropoda and Uroobovella spp. (Uropodoidea) phoretic on bark beetles (Scolytinae): a comparison from a declining mountain spruce forest in Central Europe. Int. J. Acarol., 42(4):1-6. https://doi.org/10.1080/01647954.2016.1154107



2026-02-19
Date accepted:
2026-04-15
Date published:
2026-04-29
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Chaires-Grijalva, Martha Patricia; Estrada-Venegas, Edith Guadalupe; Equihua-Martínez, Armando; Moser, John C.; Blomquist, Stacy R.; Pérez-Silva, Mauricio; Arriola-Padilla, Víctor Javier; Rodríguez-Ortega, Alejandro and Kontschán, Jenő
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



