Imparipes specus sp. nov., a new scutacarid mite species (Acari, Heterostigmatina: Scutacaridae) associated with the blind cave trapdoor spider Troglodiplura beirutpakbarai Harvey & Rix, 2020 (Araneae, Mygalomorphae: Anamidae)
Baumann, Julia  1
and Shaw, Matthew D.
1
and Shaw, Matthew D.  2
2
1✉ 1Institute of Biology, University of Graz, Universitätsplatz 2, 8010 Graz, Austria.
2Australian Museum Research Institute, Australian Museum, 1 William St, Sydney, NSW 2010, Australia.
2026 - Volume: 66 Issue: 1 pages: 1-13
https://doi.org/10.24349/ofho-icwyZooBank LSID: 0C8D04EC-8E05-45E6-9515-E2FC95E097CF
Original research
Keywords
Abstract
Introduction
Approximately half of the more than 800 (Zhang et al. 2011) described species in the mite family Scutacaridae (Acari: Heterostigmatina) are known to form phoretic associations (for complete information about phoresy in Scutacaridae, see the review by Baumann 2018 and its update Baumann 2024). Although many different arthropods like ants, bees or beetles are known as hosts for scutacarids, associations between Scutacaridae and other arachnids are rarely reported. Notably, Scutacarus talpae (Oudemans, 1913), S. acarorum (Goeze, 1780), and S. deserticolus Mahunka, 1969 have been found attached to mesostigmatid mites (see Baumann 2018). Imparipes tocatlphilus Ebermann & Palacios-Vargas, 1988 was discovered phoretic on the ricinuleid Cryptocellus boneti from Mexico (Ebermann & Palacios-Vargas 1988), although this species is not arachnid-specific and is found in soil as well. Two species, Scutacarus adgregatus Ebermann & Goloboff, 2002 and S. araneophilus Ebermann & Goloboff, 2002, have been reported as phoretic on nemesoid trapdoor spiders (Stenoterommata iguazu, S. platense, and S. uruguai, family Nemesiidae) from Argentina (Ebermann & Goloboff 2002). Based on current understanding of scutacarid habits, it was predicted that permanent burrows made by their associates would likely serve as the primary habitat for these mites (Ebermann & Goloboff 2002). Therefore, the authors hypothesized that these scutacarids are not only phoretic but also inquilines.
Inquilinism is the relationship where a species lives within the nest or burrow of a host, feeding on substrate (like detritus or fungi growing on detritus) encountered inside it and completing its lifecycle there, and it can be encountered in a large number of scutacarid species (Baumann 2018). However, documented cases where a scutacarid is demonstrated to be both phoretic and an inquiline remain rare (Baumann 2024). The issue of inquilinism is especially relevant to the frequent observations of scutacarids associated with ants and bees. Inquilinism is not unexpected in these systems and numerous arthropod families, including representatives of beetles, flies or lepidoptera, are well known to cohabit with social Hymenoptera (e.g. Smith 1886, Yanega & Leschen 1994, Hölldobler & Kwapich 2023). However, inquilinism may warrant closer scrutiny in less well-known host associations, such as those involving mygalomorph spiders.
During surveys of caves in the Australian Nullarbor Plain, a new species of Imparipes was found phoretic on the large blind nemesoid spider Troglodiplura beirutpakbarai Harvey & Rix, 2020 (Marsh et al. 2023). This spider is enigmatic in that its burrows remain unknown. Here, we describe the new species and discuss its possible biology. This is the ninth species of Imparipes recorded from Australia and adds valuable insight about the still poorly explored biodiversity of Australian Scutacaridae (30 species in five genera are currently known; Katlav et al. 2020).
Material and methods
Sediment from two nearby caves from the Nullarbor Plain, both inhabited by Troglodiplura beirutpakbarai (Caves 5N-253 and 5N-6838) and ca 18km apart, were sampled and extracted using desiccating funnels. The study area and the geomorphic characters of the two caves are given in detail in Marsh et al. (2023). To protect the sensitive cave systems, and following the recommendations published by the GBIF Secretariat (Chapman & Wieczorek 2020), no location data of the caves is released publicly. Individual spiders were collected into clean plastic containers filled with either propylene glycol or 100% ethanol. In the laboratory, spiders were examined under a high power dissecting microscope (63x objective), and all fluid contents within the vials were carefully inspected for dislodged mites.
Microscopic slides were prepared using Swan's modified Berlese's fluid as the embedding medium (Swan 1936), and specimens were examined, measured, drawn, and photographed using a Nikon Eclipse Ni-U compound microscope. Illustrations were created using a tablet and the Concepts application (https://concepts.app ![]() ), and subsequently processed with the open-source vector graphics editor Inkscape (https://inkscape.org
), and subsequently processed with the open-source vector graphics editor Inkscape (https://inkscape.org ![]() ).
).
Most specimens suitable for mounting were measured (n = 7), with measurements provided in micrometres (μm), giving first the paratypes range and holotype in parenthesis. When a particular character could not be measured in all specimens, the corresponding sample size is noted in the description. As specimens were collected from two different caves (see below, ''Type Material''), the two groups were compared using univariate t-tests on all measured characters, as well as multivariate PERMANOVA, to assess potential differences. Only characters that could be clearly measured in all specimens were included in these analyses. Although the small sample size limits the strength of statistical inference, preliminary results suggest possible minor differences between the two cave populations and underscore the value of further sampling. Nomenclature of idiosoma and legs setae follow that of Grandjean (1940), with modification used in Lindquist (1986) for Heterostigmata. The number of solenidia in setal formulas is indicated in parentheses.
Results
From the sediment samples, various predatory and scavenging mites were recovered (Shaw 2022), but no scutacarids were found. However, several specimens of a new species of Imparipes were recovered from Troglodiplura beirutpakbarai spiders. Most were found at the bottom of the collecting vials and could only have been dislodged from the host spider. Additionally, three specimens were found in the intercoxal regions of two large T. beirutpakbarai individuals (see Taxonomic section).
Taxonomic part
Family Scutacaridae
Genus Imparipes Berlese, 1903
Type species Imparipes histricinus Berlese, 1903, by original designation
Imparipes (Imparipes) specus Baumann sp. nov.
ZOOBANK: D74DBF21-9509-48BE-9DED-1FDDD0C86CE8 ![]()
Imparipes ''YALBB sp11'' in Shaw 2022
Imparipes sp. Marsh, Milner, Shaw, Stempel, Harvey & Rix 2023
Type material
Holotype — Adult female mounted on microscope slide, recovered from fluid containing a subadult Troglodiplura beirutpakbarai (voucher SAM NN30801; spider 1, cephalothorax length 9.1 mm, body length 22.1 mm), collected from Eagles Rest Cave (5N-253), Nullarbor Regional Reserve, dark zone, 28 November 2021. Collected by J. Marsh, S. Milner, A. Stempel & M. Shaw.
Paratypes — Three adult females, slide-mounted, same data as holotype; Three adult females mounted on microscope slides, recovered from fluid containing an adult female T. beirutpakbarai (NN30813; Trog 2, cephalothorax length 12.8 mm, body length 26.6 mm), collected from Wedge Cave (5N-6838), Nullarbor Regional Reserve, South Australia, dark zone, 7 March 2022. Ambient conditions: 17.3 °C, 95% RH. Collected by S. Milner & A. Stempel.
Other unmounted material in ethanol — Three female Imparipes specimens were recovered directly from the bodies of two T. beirutpakbarai spiders. These mites were adhered to hairs emerging from the coxal regions of the spiders and were collected using a fine brush. Two specimens were found between coxae III and IV of an adult female spider (NN30806; Cave 5N-253), and one specimen was located on a hair between the retrolateral faces of coxae IV of a subadult spider (NN30808; Cave 5N-253). One of these specimens was slide-mounted and confirmed the co-specificity with the type material, but the slide was accidently damaged beyond repair. The other two specimens remain preserved in ethanol. In addition, other Imparipes specimens were recovered from vials containing individual T. beirutpakbarai spiders. These specimens, particularly those from caves other than the type locality, require further investigation to determine their taxonomic status. Notably, all Imparipes specimens recovered so far were associated with large subadult or adult spiders. None were found on the bodies of, or in vials containing, juvenile Troglodiplura specimens.
Deposition of type material: Holotype and two paratypes to South Australian Museum (Adelaide); two paratypes to Natural History Museum Vienna, Austria; two paratypes to Australian Museum (Sydney).
Description
Female (Figs. 1–5)
Body dimensions — idiosoma length 227–279 (279), width measured on broadest part of tergite C 179–230 (218).



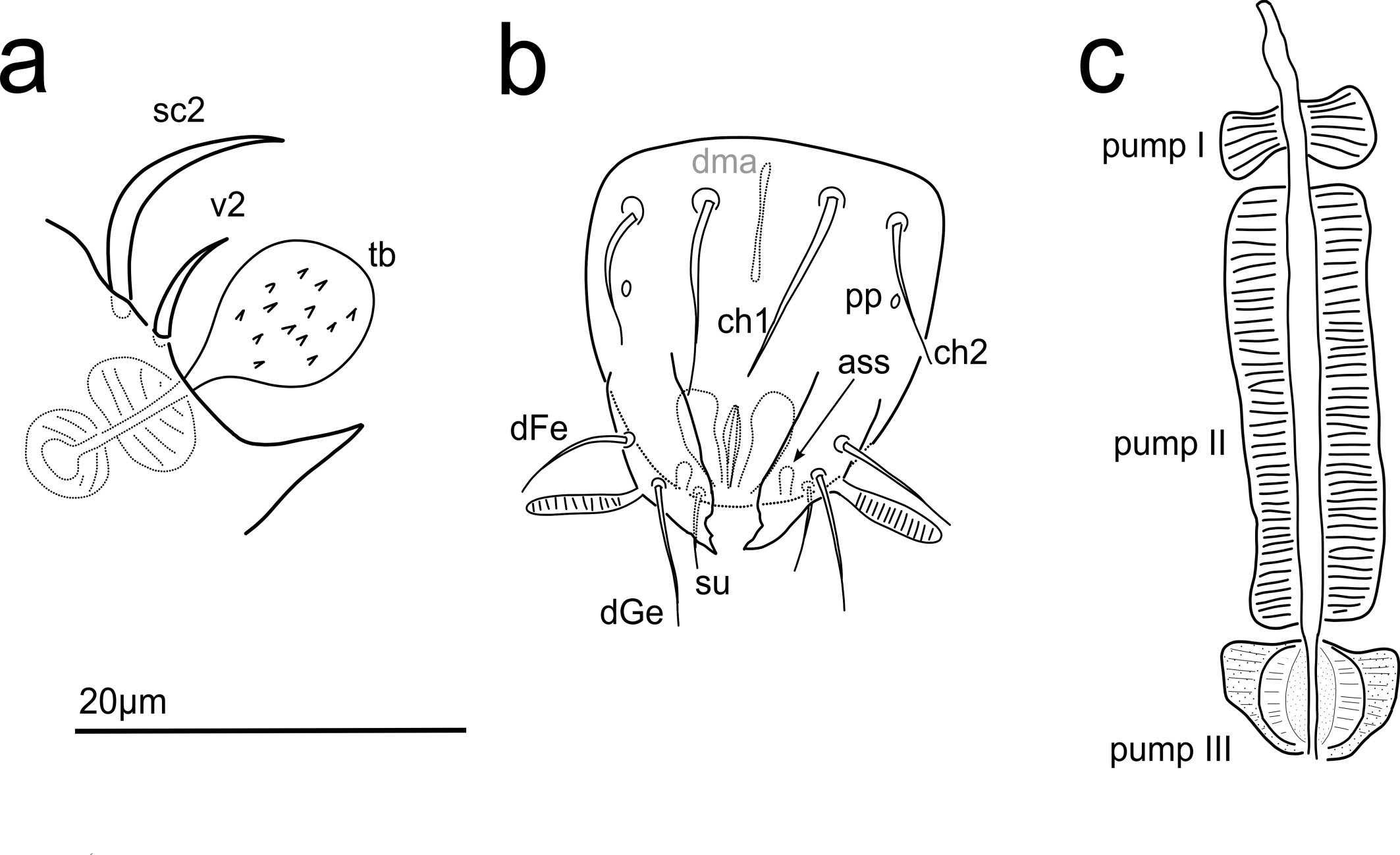
Dorsum — (Fig. 1). The entire surface of the body stippled with very fine dimples. Free margin of tergite C with fine radiating striae, posterior parts of tergites C, D and EF with fine imbricate furrows. Setae sc2, v2 smooth (Fig. 2a), all other setae barbed, c1 , c2 , e and f only sparsely. Seta c2 with long, thick-walled alveolar canal, 33–43 (43). Setae f and h1 blunt-ended, all other setae pointed. Trichobothrium (tb; Fig. 2a) 20–23 (22), club-shaped, stemmed, weakly barbed. Seta sc2 12–17 (17), v2 6–8 (6). Cupules ia on tergite D roundish, situated anterolaterally to bases of setae d; cupules ih on tergite H roundish, situated anteromedially to bases of setae h2.


Length of dorsal setae c1 53–68 (60), c2 55–70 (67), d 62–76 (73), e 32–45 (44), f 54–68 (66), h1 57–77 (74), h2 31–40 (40). Distance between dorsal setae: c1–c1 44–56 (51), c2–c2 142–185 (165), d–d 117–160 (140), e–e 132–176 (155), f–f 56–76 (65), h1–h1 35–47 (44), h2–h2 98–130 (118).
Venter — (Fig. 3). Sternal plates stippled with very small dimples. Apodeme (ap)1, ap2 clearly sclerotized; ap2 fused with well developed prosternal ap (appr); appr joined with sejugal ap (apsej); secondary transverse ap (sta) well developed; ap3 weakly developed, ap4 well developed and joined with poststernal apodeme (appo); ap5 developed only at base of leg IV.
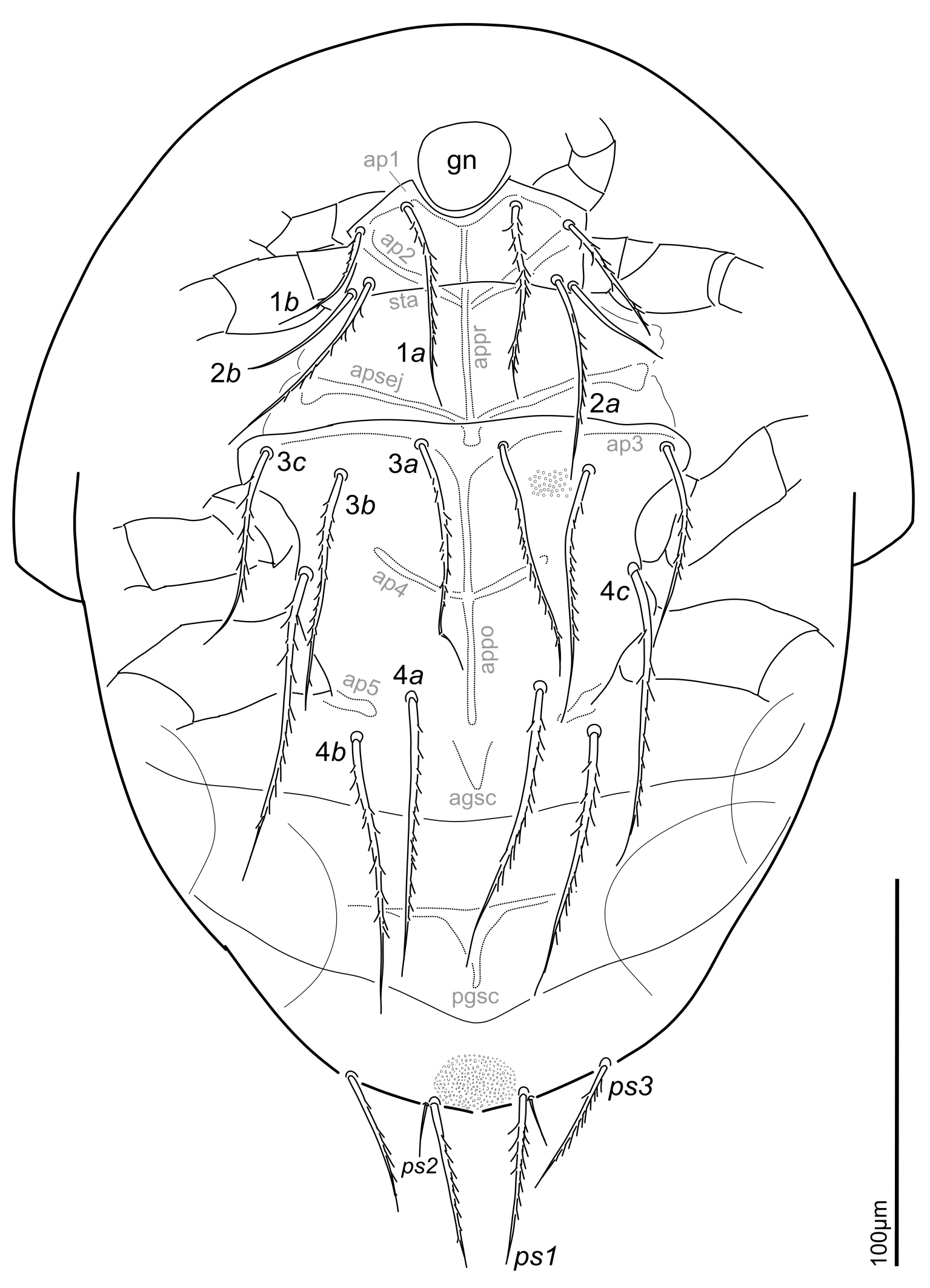


Length of ventral setae 1a 39–54 (50), 1b 28–48 (29; n = 6), 2a 41–54 (54), 2b 24–32 (32), 3a 51–76 (67), 3b 52–69 (66), 3c 42–58 (58; n = 6), 4a 60–80 (72), 4b 59–72 (72), 4c 60–87 (78), ps1 39–47 (46), ps2 10–15 (15), ps3 26–42 (39). Distance between ventral setae 1a–1a 25–28 (27), 1b–1b 46–55 (52), 2a–2a 42–53 (47), 2b–2b 58–60 (55), 3a–3a 19–23 (21), 3b–3b 53–68 (62), 3c–3c 85–107 (101), 4a–4a 28–37 (33), 4b–4b 52–67 (60), 4c–4c 71–90 (82), ps1 –ps1 20–26 (23), ps2–ps2 24–30 (27), ps3–ps3 52–73 (65).
Setae 2b and ps2 smooth, all other setae barbed, seta 2b very slightly dagger-like, ps2 short, thin. Setae 2b and all ps setae rather blunt, all other setae pointed.
Anterior genital sclerite (agsc), posterior genital sclerite (pgsc) triangular, pgsc broader than long; width 18–25 (23), length 19–21 (20).
Gnathosoma — (gn; Fig. 2b, 3). Roundish, length 20–30 (26; n = 6), width 17–21 (19); surface finely stippled; dorsal setae ch1 12–17 (15; n = 6) and ch2 9–15 (14; n = 4) both thin, smooth; postpalpal setae pp very thin and rather short, hardly discernable; ventral setae su 5–7 (7; n = 4). No pits visible. Each palp with thin, smooth setae dGe 8–10 (10; n = 4) and dFe 7–12 (10; n = 5), one solenidion 5–8 (8; n = 6) and piston-like accessory setigerous structure (ass). Dorsal median apodeme (dma) visible. Pharyngeal pumps (Fig. 2c) tripartite, situated on long esophagus, separated from each other. Pump I bow-shaped, pump II long, striated, pump III 1/5 of pump II, subtriangular in shape with internal circular structure, only very weakly striated. Esophagus narrowing abruptly when reaching pump III.



Leg I — (Fig. 4a). Setal formula: Tr 1, Fe 3, Ge 4, TiTa 16(4). Solenidion ω1 14–18 (17), ω2 9–11 (11), φ1 11–14 (14), φ2 9–13 (11); ω1 thick, with acute tip, φ2 and ω2 baculiform, φ1 clavate with acute tip. TiTa with a large claw with tapering tip. Setae v′Tr, dFe, l′Fe, v″Fe and kTiTa smooth, all other setae (except eupathidia) barbed. Seta dFe bifurcate, seta v″Fe capitate, seta k blunt; all other setae (except eupathidia) pointed. Eupathidia tc″ and ft″ arise from long cuticular setal base (pinnaculum).
Leg II — (Fig. 4b). Setal formula: Tr 1, Fe 3, Ge 3, Ti 4(1), Ta 6(1); Ta with 2 claws and large empodium. Solenidion ω 13–16 (14), clavate with acute tip, φ 6–9 (9), finger-shaped, pressed against surface of segment. Setae v′Tr, dFe, l′Fe, v″Fe, l′Ti, pl″, u, tc′ and tc″ smooth, all other setae barbed. Seta dFe blunt, all other setae pointed.
Leg III — (Fig. 4c). Setal formula: Tr 1, Fe 2, Ge 2, Ti 4(1), Ta 6, Ta with 2 claws and large empodium. Solenidion φ 5–7 (6; n = 5), finger-shaped, pressed against surface of segment. All setae pointed and barbed, some only sparsely.



Leg IV — (Fig. 5). Setal formula: Tr 1, Fe 2, Ge 1, Ti 3(1), Ta 6, PrTa 0. Tr rather thin and long. Ta longer than PrTa, Ta 56–72 (66), PrTa 34–48 (43), PrTa terminating with two tiny claws and small empodium. Solenidion φ 7–10 (8), finger-shaped. Seta u′ and tc″ smooth, all other setae barbed, tc′ only sparsely. Seta dGe blunt, all other setae pointed.
Like the whole surface of the body, the segments of the legs are stippled with very fine dimples (not depicted in the figures).
Male and larva
Not known.
Etymology
The species name specus is derived from the Latin word specus (f.), meaning ''cave'', in reference to the cave habitat of the new species.
Differential diagnosis
The new species is characterized by a long and thin leg IV with Tr thin and Ta > PrTa, by setae e and h2 being shorter than the remaining dorsal setae, and by a long alveolar channel of seta c2 , although the latter character is quite common in the genus and additionally might not be reliable (see below).
Of all other Imparipes species with a pronounced alveolar channel of c2 , only I. hajiqanbari Rahiminejad, 2022 (Rahiminejad et al. 2022) has a similarly shaped leg IV, but setae e and h2 are very long in this species and setae c1 are very short. Moreover, I. bengalensis Radhakrishnan & Ramaraju, 2017 and I. comatus Mahunka, 1970a also have rather thin legs IV, but again in both species setae e and h2 are clearly longer than in the new species.
There are ten other Imparipes species which possess similarly shaped legs IV as the new species, but lack alveolar channels of c2 : I. cavernophilus Sevastianov, 1974, I. chacoensis Mahunka & Mahunka-Papp, 1982, I. ethiopicus Mahunka, 1981, I. heterotrichus Mahunka, 1963a, I. latispinus Mahunka, 1970b, I. longisetosus Willmann, 1951, I. mongolicus Mahunka, 1967, I. penicillatus Mahunka, 1967, I. rafalskii Dastych, 1978 and I. risafiensis Sevastianov & Al Douri, 1991. In fact, several (mostly older) descriptions of Imparipes species don′t mention the alveolar channel of c2 . The channel may indeed have been absent, but it may have been ignored or overlooked due to limitations in optical equipment. Therefore, the species given above were also compared with our new species. All, except I. cavernophilus, are clearly distinguishable from I. specus sp. nov. based on dorsal setation, particularly the relatively longer setae e and h2. Imparipes cavernophilus differs more subtly, with only slightly longer e and h2, but has distinctly shorter h1 setae and dagger-like ventral setae 2b (versus more setiform in I. specus sp. nov.).
Statistical comparison
Univariate t-tests of all measured characters revealed no significant differences between specimens from the two caves 5N-6838 and 5N-253 except for the lengths of the dorsal seta h2 and the small pseudanal seta ps2 (Table 1). Both setae were significantly (p < 0.05) longer in individuals from cave 5N-253 than in individuals from cave N-6838. Multivariate PERMANOVA revealed no significant differences.
Download as * indicates significant (p < 0.05) differences revealed by t-test
5N-6838
5N-253
n = 3
n = 4
length
238–276
275–279
width
179–230
184–218
sc2
13–16
12–17
v2
6–8
6–7
c1
58–68
53–67
c2
59–70
55–67
d
63–76
62–73
e
32–45
33–44
f
54–68
55–68
h1
57–73
62–77
h2
31–36*
39–40*
distance c1-c1
47–49
44–56
distance c2-c2
143–185
142–170
distance d-d
129–160
117–151
distance e-e
134–176
132–166
distance f-f
60–76
56–71
distance h1-h1
35–47
37–46
distance h2-h2
100–130
98–126
width of gnathosoma
17–21
19–20
1a
42–54
39–50
2a
41–47
45–54
2b
27–32
24–32
3a
58–76
51–74
3b
59–69
52–69
4a
65–80
60–75
4b
67–72
59–72
4c
68–87
60–82
trichobothrium
20–23
20–22
distance 1a-1a
25–27
25–28
distance 1b-1b
46–55
46–52
distance 2a-2a
45–53
42–52
distance 2b-2b
53–60
48–58
distance 3a-3a
20–23
19–21
distance 3b-3b
57–68
53–65
distance 3c-3c
89–107
85–102
distance 4a-4a
32–36
28–37
distance 4b-4b
58–67
52–63
distance 4c-4c
75–90
71–87
ps1
39–45
42–47
ps2
10–11*
11–15*
ps3
26–42
35–41
distance ps1-ps1
21–25
20–26
distance ps2-ps2
25–29
24–30
distance ps3-ps3
57–73
52–72
length of posterior genital sclerite
19–20
19–21
width of posterior genital sclerite
21–25
18–23
solenidion ω1 of leg 1
14–18
15–17
solenidion ω2 of leg 1
10–11
9–11
solenidion φ1 of leg 1
12–14
11–14
solenidion φ2 of leg 1
11–13
9–11
solenidion ω on leg 2
13–16
13–15
solenidion φ on leg 2
6–9
6–9
solenidion φ on leg 4
7–10
8–9
canal of seta c2
33–43
38–43
Musculature



Musculature is typically not discernible in permanent microscopic slides of scutacarid mites, as all known embedding media tend to digest all soft internal tissues in these small-bodied mites. In the present species, however, muscle strands were visible both in the idiosomal region and within segments of the legs (Fig. 6). This suggests either unusually robust muscle fiber integrity in Imparipes specus sp. nov., or a fortuitous interaction between the specimen's preservation state and the embedding medium used. As is typical for mites (Evans 1992), all observed musculature was striated. The idiosomal musculature likely represents coxal muscles, as the strands extend from the apodemata to the bases of the leg trochanters. The muscle arrangement appears to form two distinct layers, presumably corresponding to the elevator and depressor muscles of the legs.
Discussion
Cave environments offer a variety of niches, supporting surprisingly diverse biodiversity, including mites from various orders (Palacios-Vargas 1996). Ducarme et al. (2004) noted a higher proportion of phoretic forms in two caves compared to mineral soil. Despite this, reports of scutacarid mites from caves remain sparse: Imparipes cavernophilus, collected from bat guano from caves in Azerbaijan (Sevastianov 1974), I. tocatlphilus found in both soil and guano, as well as phoretic on ricinuleids in Mexican caves and also from litter in Brazil (Ebermann and Palacios-Vargas 1988), and Reductacarus singularis Mahunka, 1963b recorded from soil at the entrance of a Greek cave (Mahunka 1974), but also from rodents' nests (Mahunka 1963b) and from soil in Mongolia (Mahunka 1969). Other reports provide limited taxonomic resolution: for instance, Cokendolpher and Polyak (1996) identified a Scutacarus species from the dark zone of a New Mexico cave; Moseley et al. (2012) recorded two scutacarid species in Malaysia's Batu caves; and Żbikowska-Zdun et al. (2009) reported abundant Scutacaridae in bat guano from a Polish cave. These records suggest that further surveys of cave acarofauna would likely uncover additional troglobiotic scutacarid species, particularly in caves rich in organic matter that supports saprophagous communities.
To date, only two scutacarid species have been found in association with spiders: Scutacarus adgregatus and S. araneophilus, both phoretic on nemesiid spiders (Ebermann & Goloboff 2002). Like Imparipes specus sp. nov., these mites were found predominantly on adult spiders, though also occasionally on juveniles. Ebermann and Goloboff (2002) suggested that these mites develop within spider burrows, a hypothesis we find plausible. If true, these mites would qualify as inquilines. Prey remnants and spider exuviae accumulate at the bottom of the burrows, where they most likely are overgrown by fungi and create a suitable habitat for the mites. Interestingly, intercoxal areas have been consistently observed as the microhabitat of attachment in these spider-associated scutacarids. This consistency supports the notion of the mites exhibiting specific microhabitat preferences on the host.
Immatures of the host species of the new Imparipes, T. beirutpakbarai, sometimes retreat into natural holes or crevices when disturbed. However, adult Troglodiplura have not yet been shown to produce or use holes or burrows (Marsh et al. 2023). This is unusual for this family of spiders. These questions about Troglodiplura burrow use are further highlighted by the remarkable positioning of male exoskeletons, and sometimes mummified females, which have been found outside crevices in open areas within caves (Marsh et al. 2023). The Nullarbor caves, where these observations were made and I. specus sp. nov. was found, sometimes exhibit high humidity and large amounts of arthropod remains. Saprophagous and predatory arthropods are abundant, although their presence varies across different zones of Cave 5N-253. It is reasonable to assume that fungal hyphae, growing on arthropod remains, could provide a food source for Imparipes. However, neither Imparipes nor other scutacarids have been found in the sediment of the Nullarbor caves so far. This makes the developmental habitat of I. specus sp. nov. unclear. If future studies find concentrations of I. specus sp. nov. in crevices or holes used by Troglodiplura, and particularly if the still uncollected male mites are discovered there, this would align with a broader pattern observed among many scutacarids, especially Imparipes species, which often inhabit nests or burrows where they attach to their hosts. Or put another way, while I. specus sp. nov. is clearly phoretic, can it also be considered an inquiline? The relatively stable environmental conditions of cave systems may provide an ideal context for studying phoresy and host associations in scutacarid mites.
Acknowledgments
We offer sincere thanks to the Traditional Owners and Indigenous Rangers from the Far West Coast Aboriginal Corporation and the Yalata Anangu community, on the lands on which we conducted our surveys. We thank Frank Kutsche and the Department of Environment and Water for administering a complex permit (M27023-2) in a restricted timeframe, and to the Australian Speleological Foundation for sharing data that helped select and locate the caves used in these surveys. The fieldwork component of this research was supported and funded by Bush Blitz, the species-discovery program run by the Australian Government in partnership with Broken Hill Proprietary Company Limited (BHP) and EarthWatch Australia, during the Yalata Bush Blitz Expedition 2021. M. Shaw thanks the Australian Museum Foundation and the Anne McIntosh Trust via Perpetual Guardian for support via the Micro-Monsters project. Access to these caves was only possible due to the careful sustained efforts and material support of cavers, Steve Milner and Andrew Stempel.
References
- Baumann J. 2018. Tiny mites on a great journey- a review on scutacarid mites as phoronts and inquilines (Heterostigmatina, Pygmephoroidea, Scutacaridae). Acarologia, 58(1): 192-251. https://doi.org/10.24349/acarologia/20184238
- Baumann, J. 2024. An update on the knowledge about scutacarid mites as phoronts and inquilines. Acarologia, 64(1): 3-17. https://doi.org/10.24349/sy16-8iqn
- Chapman, A.D., Wieczorek, J.R. 2020. Georeferencing Best Practices. Copenhagen: GBIF Secretariat. https://doi.org/10.15468/doc-gg7h-s853
- Cokendolpher, J. C., Polyak, V. J. 1996. Biology of the caves at Sinkhole Flat, Eddy County, New Mexico. J. Cave Karst Stud., 58(3):181−192.
- Dastych, H. 1978. Imparipes (Imparipes) rafalskii sp. nov. (Acari, Tarsonemina) a New Species of Scutacarid Mite from Poland. Bull. Acad. Pol. Sci., 26(4): 251−255.
- Ducarme, X., Wauthy, G., André H. M., Lebrun P. 2004. Survey of mites in caves and deep soil and evolution of mites in these habitats. Can. J. Zool., 82 (6): 841−850. https://doi.org/10.1139/z04-053
- Ebermann E., Goloboff P.A. 2002. Association between neotropical burrowing spiders (Araneae: Nemesiidae) and mites (Acari: Heterostigmata, Scutacaridae). Acarologia, 52 (2):173−184.
- Ebermann E., Palacios-Vargas J.C. 1988. Imparipes (Imparipes) tocatlphilus n.sp. (Acari, Tarsonemina, Scutacaridae) from Mexico and Brazil: First record of ricinuleids as phoresy hosts for scutacarid mites. Acarologia, 29: 347−354.
- Evans, G.O. 1992. Principles of Acarology. C.A.B., Wallingford. 563 pp. https://doi.org/10.1079/9780851988221.0000
- Goeze, J.A.E. 1780. Herrn Pastor Gözens neuentdeckte Theile an einigen Insekten. Naturforscher, 14: 93-102.
- Grandjean, F. 1940. Les poils et les organes sensitifs portés par les pattes et le palpe chez les Oribates. Deuxième Partie Bull. Soc. Zool. Fr., 65: 32-44.
- Harvey, M. S., Rix, M. G., Hillyer, M. J., & Huey, J. A. 2020. The systematics and phylogenetic position of the troglobitic Australian spider genus Troglodiplura (Araneae: Mygalomorphae), with a new classification for Anamidae. Invertebr. Syst., 34(8): 799−822. https://doi.org/10.1071/IS20034
- Hölldobler, B., Kwapich, C. 2023. Die Gäste der Ameisen. Wie Myrmecophile mit ihren Wirten interagieren. Springer Berlin, Heidelberg. 487 pp. https://doi.org/10.1007/978-3-662-66526-8
- Katlav, A., Riegler, M., Seeman, O.D. 2020. Tiny hitchhikers and parasites: a review of Australian heterostigmatic mites (Acari: Prostigmata) associated with insects, with description of three new species. Austral Entomol., 59 (3): 401−421. https://doi.org/10.1111/aen.12484
- Lindquist, E. 1986. The world genera of Tarsonemidae (Acari: Heterostigmata): a morphological, phylogenetic, and systematic revision, with a reclassification of family-group taxa in Heterostigmata. Mem. Entomol. Soc. Can., 136: 1–517 https://doi.org/10.4039/entm118136fv
- Mahunka, S. 1963a. The Zoological Result of Gy. Topál's Collectings in South Argentina. Ann. Hist.-Nat. Mus. Natl. Hung., 55: 501−507.
- Mahunka, S. 1963b. Beiträge zur Kenntnis der Milbenfauna (Acari) von Säugetiernestern. Acta Zool. Acad, Sci. Hung., 9: 355−372.
- Mahunka, S. 1967. 83. Acari: Pyemotidae und Scutacaridae. Ergebnisse der zoologischen Forschungen von Dr. Kaszab in der Mongolei. Reichenbachia, 9 (1): 1−196913.
- Mahunka, S. 1969. Pyemotidae und Scutacaridae IV. Ergebnisse der zoologischen Forschungen von Dr. Kaszab in der Mongolei (Acari). Reichenbachia, 12 (10): 83−112.
- Mahunka, S. 1970a. Dudichiana foveolata gen. nov., sp. nov., sowie einige andere neue und interessante Milbenarten aus Ameisenhaufen (Acari: Tarsonemina). Opusc. Zool. Budapest, 10:143-149.
- Mahunka, S. 1970b. Two New Scutacarid Mites (Acari, Tarsonemina) from Pogonmyrmex occidentalis (Hymenoptera) in the United States of America. Parasit. Hung, 3: 87-96.
- Mahunka, S. 1974. Neue und interessante Milben aus dem Genfer Museum VIII Tarsonemina-Arten (Acari) aus Griechenland. Biol. Gallo-Hellenica 5 (2), 209-225.
- Mahunka, S. 1981. Tarsonemiden aus Äthiopien (Acari: Tarsonemina). Fol. Entomol. Hung., 42(34), 2: 101-121.
- Mahunka, S., Mahunka-Papp, L. 1982. Neue und interessante Milben aus dem Genfer Museum L. Weiterer Beitrag zur Kenntnis der Tarsonemiden-Fauna (Acari) von Paraguay. Rev. Suissse Zool., 89 (3): 595-605. https://doi.org/10.5962/bhl.part.82462
- Marsh J.R., Milner S.J., Shaw M., Stempel A.J., Harvey M.S., Rix M.G. 2023. A case for below-ground dispersal? Insights into the biology, ecology and conservation of blind cave spiders in the genus Troglodiplura (Mygalomorphae: Anamidae). Insects, 14: 449. https://doi.org/10.3390/insects14050449
- Moseley, M., Lim, T.W., Lim T.T. 2012. Fauna reported from Batu caves, Selangor, Malaysia: annotated checklist and bibliography. Cave Karst Sci., 39(2): 77−92.
- Oudemans, A.C. 1913. Acarologisches aus Maulwurfsnestern. Arch. Naturg., 79(9): 68−136.
- Palacios-Vargas, J. G. 1996. Why, where and when are mites abundant in caves? Mundos Subterráneos, 7: 11−19.
- Rahiminejad, V., Nadimi, A., Seyedein, S. 2022. Contribution to the knowledge of the genus Imparipes Berlese (Acari: Heterostigmata: Scutacaridae) associated with ants in Iran. Zootaxa, 5133(4): 585−593. https://doi.org/10.11646/zootaxa.5133.4.9
- Radhakrishnan, V., Ramaraju, K. 2017. First record on Imparipes bengalensis sp. nov.(Acari: Scutacaridae) on Labidura bengalensis Dohrn (Forficulidae: Dermaptera) from Tamil Nadu, India. J. Entomol. Zool. Stud, 5(6): 1606−1611.
- Sevastianov, V.D. 1974. [New species of the family Scutacaridae (Trombidiformes)]. Vestn.
- Zool., 6: 18-22. [in Russian]
- Sevastianov, V.D., Al Douri, Z. 1991. [A new genus and species of mites of cohort Tarsonemina (Trombidiformes) of Central Iraq fauna]. Zool. Zhu., 70 (4): 137-140. [in Russian]
- Shaw, M.D. 2022. Mites and ticks from the Yalata Bush Blitz. Report to Bush Blitz Program, Canberra. 18pp https://bushblitz.org.au/wp-content/uploads/2023/06/Mites-and-ticks.pdf
- Smith, J. B. 1886. Ants' nests and their inhabitants. Am. Nat., 20(8): 679-687. https://doi.org/10.1086/274306
- Swan, D.C. 1936. Berlese's fluid: remarks upon its preparation and use as a mounting medium. Bull. Entomol. Res., 27: 389-391. https://doi.org/10.1017/S0007485300058259
- Willmann , C. 1951. Untersuchungen über die terrestrische Milbenfauna im pannonischen Klimagebiet Österreichs. Sitzungs. Öst. Akad. Wissensch. Math.-Naturwiss. Abt. I , 160: 91-176 .
- Yanega, D., Leschen, R. A. 1994. Beetles associated with bee nests (Hymenoptera: Apidae) in Chiapas, Mexico, with descriptions of the immature stages of Vanonus balteatus Werner (Coleoptera: Aderidae, Endomychidae, Meloidae). Col. Bull., 48(4): 355−360.
- Żbikowska-Zdun, K., Piksa, K., Smaczyńska A. 2009. Variation of selected morphological characters of the cave mite Oribella cavatica Kunst, 1962 (Acari, Oribatida). Biol. Lett., 46 (2): 123−127. https://doi.org/10.2478/v10120-009-0007-z
- Zhang Z.-Q., Pesic V., Smit H., Bochkov A.V., Khaustov A.A., Baker A., Wohltmann A., Wen T.-H., Amrine J.W., Beron P., Lin J.-Z., Gabrys G., Husband R. 2011. Order Trombidiformes Reuter, 1909. In: Zhang Z.-Q. (Ed.). Animal biodiversity: an outline of higher-level classification and survey of taxonomic richness. Zootaxa, 3148: 129-138. https://doi.org/10.11646/zootaxa.3148.1.24



2025-10-28
Date accepted:
2025-12-22
Date published:
2026-01-09
Edited by:
Pfingstl, Tobias

This work is licensed under a Creative Commons Attribution 4.0 International License
2026 Baumann, Julia and Shaw, Matthew D.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



