A discussion on the validity of the genus Abalakeus (Acari: Erythraeidae) with a redescription of A. gonabadensis
Saboori, Alireza  1
; Kapankaya, Alperen
1
; Kapankaya, Alperen  2
; Cakmak, Ibrahim
2
; Cakmak, Ibrahim  3
and Hakimitabar, Masoud
3
and Hakimitabar, Masoud  4
4
1✉ Jalal Afshar Zoological Museum, Department of Plant Protection, Faculty of Agriculture, University of Tehran, Karaj, Iran & Department of Plant Protection, Faculty of Agriculture, Aydin Adnan Menderes University, Aydin, Türkiye.
2Department of Plant Protection, Faculty of Agriculture, Aydin Adnan Menderes University, Aydin, Türkiye.
3Department of Plant Protection, Faculty of Agriculture, Aydin Adnan Menderes University, Aydin, Türkiye & Department of Research and Innovation, Saveetha School of Engineering, SIMATS, Thandalam, Chennai-602105, Tamil Nadu, India.
4Department of Plant Protection, Faculty of Agriculture, Shahrood University of Technology, Shahrood, Iran.
2023 - Volume: 63 Issue: 2 pages: 295-305
https://doi.org/10.24349/1icc-70f3Original research
Keywords
Abstract
Introduction
The genus Abalakeus (with type species A. chekei Southcott) was established by Southcott in 1994 based on the larval stage. Since then, a new species (A. bambusae Zhang) from bamboo forests in Fujian, China (Zhang et al. 2000) and three species (A. lorestanicus Saboori and Lachinani; A. jahromiensis Sedghi, Saboori and Hakimitabar; A. gonabadensis Ahmadi, Hajiqanbar and Saboori) from Iran (Saboori and Lachinani 2003; Sedghi et al. 2010; Ahmadi et al. 2012) have been described. Mayoral and Barranco (2006) collected larval specimens of A. lorestanicus Saboori and Lachinani, 2003 in Andalucia, Spain ectoparasitic on Steropleurus andalusius (Rambur, 1838) (Orthoptera: Tettigoniidae) and described its nymphal stage covered with leaf-like setae on idiosoma and legs. Mąkol and Sevsay (2015), obtained heteromorphic larvae, congeneric with the hitherto distinguished Abalakeus, from eggs laid by a ''plume-footed'' female, identified as Eatoniana plumipes from Türkiye and considered Abalakeus a junior synonym of Eatoniana Cambridge, 1898 and synonymized E. jahromiensis (Sedghi, Saboori and Hakimitabar, 2010) with E. plumipes (L. Koch, 1856). New metric data on E. lorestanica (Saboori and Lachinani, 2003) was presented by Noei and Rabieh (2019). Also, they corrected some mistakes in the original description and presented new metric data and a key to species of Eatoniana. Noei and Honarmand (2020) corrected some mistakes in the original description and presented new metric data for E. gonabadensis (Ahmadi, Hajiqanbar and Saboori, 2012). Recently, Xu et al. (2022) described a new species of Eatoniana from China and transferred Erythraeus (Er.) yangshuonicus Haitlinger, 2006 to Eatoniana and presented an updated key to species.
The study of terrestrial Parasitengona from Türkiye is still in progress. During a survey on terrestrial Parasitengona in Aydin province, Türkiye, and Semnan province, Iran, A. gonabadensis was collected and redescribed from type specimens as well as larvae collected ectoparasitic on grasshoppers. There were some mistakes in the original description and figures and they are corrected here.
Material and methods
Type specimens of A. gonabadensis were loaned from Acarological collection, Department of Entomology, Faculty of Agriculture, Tarbiat Modares University and from Acarological collection, Jalal Afshar Zoological Museum, Department of Plant Protection, Faculty of Agriculture, University of Tehran, Karaj, Iran. Also, 25 mite larvae attached to the wings, thoraces and hind legs of Heteracris littoralis (Rambur, 1838), Calliptamus sp. (Orthoptera: Acrididae) and Bucephaloptera sp. (Orthoptera: Tettigoniidae) from Aydin province, Türkiye, and four larvae ectoparasitic on Oedipoda miniata (Pallas, 1771) (Orthoptera: Acrididae) from Semnan province, Iran were detached by an insect pin, preserved in 75% ethanol, cleared in Nesbitt's fluid and finally mounted on microscope slides using Hoyer's or Faure media (Walter and Krantz, 2009). Figures were drawn and measurements were made using an AxioImager A2 (Carl Zeiss, Germany) compound microscope with phase contrast and DIC illumination in Türkiye and an Olympus BX 51 (Olympus, Japan) compound microscope with phase contrast in Iran. Photomicrographs were taken with an AxioCam 506 color (Carl Zeiss, Germany) digital camera. Measurements are given in micrometers (µm). Abbreviations and terminology follow Wohltmann et al. (2007) and Saboori et al. (2009).
Abalakeus Southcott, 1994
Definition of larva (after Southcott 1994, updated)
Scutum with two normal setae on each side (2 + 2); coxal setal formula: 1, 1, 1; basifemural setal formula: 2, 2, 1; TFe I with one solenidion; Ge I with one solenidion on the proximal half of the segment, Ge II with one solenidion, anterior tarsal claw hook-like, posterior tarsal claw pulvilliform with numerous ventral onychotrichs and without hook element; with two sternalae II (2a), bases of posterior sensillary (PSens) setae well-separated; palp femur and palp genu, each with one seta, odontus bifurcate at termination.
Also, leg normal setal formula is as follows: fn Cx = 1-1-1, fn Tr = 1-1-1, fn BFe = 2-2-1, fn TFe = 5-5-5, fn Ge = 8-8-8, fn Ti = 14-15-15, fn Ta = 24–28, 22–24, 22–24 (Table 1). We checked A. lorestanicus and A. gonabadensis and fn Ta for both species is 26-23-23 and here is corrected for both species. Also, number of normal setae on Ge II of A. checkei was checked and it was 8, so fn Ge will be 8-8-8 in this species too. Southcott (1994) showed only 7 normal setae in Figure 3.



Corrections in the description of A. lorestanicus
Saboori and Lachinani (2003) mistook companion seta (z) with dorsal eupathidia as a famulus for A. lorestanicus. They did not consider dorsal eupathidium with a Cp by mistake. No famulus was seen in Abalakeus.
Discussion on Abalakeus and its species
After the description of Abalakeus bambusae (now Eatoniana bambusae) by Zhang et al. (2000), the presence of solenidion on TFe I and shape of posterior tarsal claw were considered by authors as intraspecific variations. Mayoral and Barranco (2006) reared Abalakeus lorestanicus and got a deutonymph that had only leaf-like setae, and without any plume-like setae on legs. Deutonymph is similar to the adult in morphology and a few characters may be different in terrestrial Parasitengona e.g. number of acetabula. Mąkol and Sevsay (2015) reared a plume-footed female of Eatoniana plumipes and obtained larvae without solenidion on TFe I. They assumed the presence of solenidion on TFe I is an intraspecific variation and considered it as an optional character in the diagnosis. Also, they did not consider tarsal claws as an important character. We added more characters in the diagnosis of Abalakeus which helps to separate it from Eatoniana in the larval stage. Mayoral and Barranco (2006) did not present a diagnosis for post-larval forms of Abalakeus and only considered leaf-like setae as the most important character of the genus. For a better separation of these two genera at larval stage, we present an updated diagnosis of Eatoniana.
Diagnosis Eatoniana (larva) (updated from Mąkol and Sevsay 2015)
Scutum with two normal setae on each side (2 + 2); coxal setal formula: 1, 1, 1; basifemural setal formula: 2, 2, 1; TFe I without solenidion; Ge I with one solenidion on the distal half of the segment, Ge II without solenidion, anterior and posterior tarsal claw feather-like; without sternalae II (2a), bases of posterior sensillary setae close together (similar to ASens bases); palp femur and palp genu, each with one seta, odontus bifurcate at termination.
So, Abalakeus can be distinguished from Eatoniana in the larval stage with the following key:
1. TFe I and Ge II each with one solenidion, anterior tarsal claw hook-like, with two sternalae II (2a), bases of posterior sensillary (PSens) setae well-separated
...... Abalakeus
— TFe I and Ge II without solenidion, anterior tarsal claw feather-like, without sternalae II (2a), bases of posterior sensillary (PSens) setae close together
...... Eatoniana
Abalakeus gonabadensis Ahmadi, Hajiqanbar & Saboori, 2012
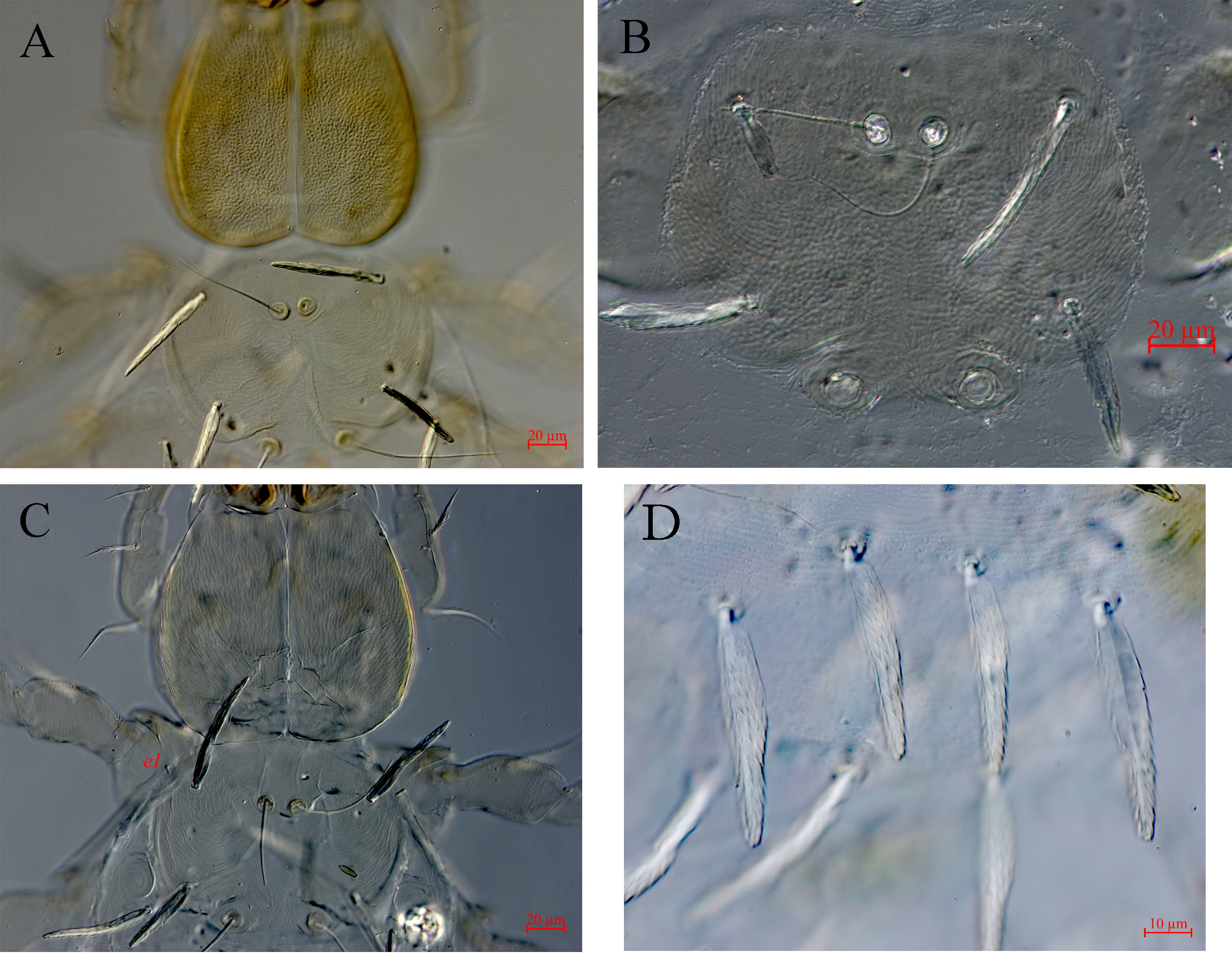
(Figures 1–4)











Diagnosis
Idiosomal setae leaf-like, some normal setae on legs blunt-ended, with two pairs of sternalae III; one solenidion on Ge III; fD = 36–44, fV = 8–12; Ti I 121–162, Ti III 118–227.
Redescription (N = 33)
Color red when alive. Idiosoma oval with 36–44 leaf-like dorsal setae with short barbs (Figures 1A and 4D). Scutum semi-oval (Figure 1A) or circular (Figure 4A) or semi-quadrangle (Figure 4B) in outline wider than long with slightly concave anterior border, and convex other borders; AL longer than PL, both leaf-like with short barbs. Anterior pair of sensilla (ASens) shorter than posterior one (PSens), both barbed in distal ¼. Scutum punctate and finely striated; distance between the SBp bases greater than SBa; ISD greater than AP (Fig. 4).
Two eyes posterolateral to scutum on each side (Figure 1A). Eye plate absent. Anterior lens 16–25 and posterior one 10–20 in diameter. Ventral surface of idiosoma bearing 8–12 leaf-like barbed setae posterior to coxae III shorter and narrower than dorsal ones (Figure 1B). Sternalae (1a, 2a, 3a) leaf-like with short barbs. There are two pairs of sternalae III (3a). Coxae I-III each with one leaf-like seta; coxalae 1b longer than other coxalae, all coxalae with fine barbs (Figure 1B). Coxal fields punctate. Supracoxal seta of leg I (eI) small, peg-like and 3–9 long (Figure 4B). Tarsi with median claw-like empodium, falciform anterior claw; posterior claw a rod which is spoon-like, without hook element but with numerous onychotrichs. NDV= [36–44] + [8–12] = [48–54].
Subcapitulum with striation and punctation. Palpal trochanter, femur and genu with striations and punctations. Galealae (cs) smooth or with 1–2 short barbs, (31–56) long, hypostomalae (bs) smooth or with 1–2 short barbs, hypostomalae (as) spine-like and minute. Palp trochanter without seta, palpal femur and genu each with one barbed seta, tibia with three barbed setae; palpal tibial claw bifid. Tarsus with six setae including a solenidion and an eupathidium (Fig. 1C). fPp = 0-B-B-BBB2-4Nωζ. Cheliceral bases punctate and finely striate. Supracoxal seta of palp (eP), minute, peg-like, 4–10 long.
Leg setal formula: Leg I (Figure 2A–B): Ta- 1ω, 2ζ, 1z, 24–28n; Ti- 2φ, 1z, 1κ, 14n; Ge- 1σ, 1κ, 8n; TFe- 1θ, 5n; BFe- 2n; Tr- 1n. Leg II (Figure 2C–D): Ta- 1ω, 1ζ, 22–24n; Ti- 2φ, 15n; Ge- 1σ, 1κ, 8n; TFe- 5n; BFe- 2n; Tr- 1n. Leg III (Figure 3): Ta- 1ζ, 20–24n; Ti- 1φ, 15n; Ge-1σ, 8n; TFe- 5n; BFe- 1n; Tr-1n. Abnormalities are considered in the leg setal formula. Leg segments punctate, all leg setae with small barbs. Some normal setae on leg segments are blunt-ended and others pointed. Meristic data is given in Table 1 for the holotype and Turkish specimens.
Measurements are given in Table 2.



Remarks on scutum and cheliceral bases punctations and striations
Punctions and striations on the scutum and cheliceral bases depend on the sclerotization of the body at the time of sampling. When the sclerotization is not completed and the body is soft (newly emerged larva), punctations cannot be seen or are weak whereas striations can be seen easily on scutum and cheliceral bases (Figure 4C). When sclerotization of the cheliceral bases is completed, then punctations can be seen easily but striations will be difficult to see and the same time scutum sclerotization may not be completed and striations can be seen easily but punctations may be too weak or cannot be seen (Figure 4A) while punctations can be seen easily on scutum and cheliceral bases in completely sclerotized specimens and striations are difficult to be seen. These observations suggests that punctions and striations on the scutum and cheliceral bases may not be a good character for comparison of species and may not be good taxonomic characters.
Material studied
Holotype and three paratypes as in the original paper. Four larvae (ARS-20221001-1a, 1b, 1i, 1j), Faculty of Agriculture, Aydin Adnan Menderes University, Aydin, Türkiye, 37°4536.0″N; 27°4514.6″E, 120 m.a.s.l, Cihan Aslı and Mesut Sayan col., ectoparasitic on Heteracris littoralis (Rambur, 1838) (Orthoptera: Acrididae: Eyprepocnemidinae), 11 October 2022; five larvae (ARS-20221001-1c, 1f, 1g, 1h, , 1k), the same data except collected ectoparasitic on Calliptamus sp. (Orthoptera: Acrididae: Calliptaminae); two larvae (ARS-20221001-1d, 1q), the same as latter except collected on 10 October 2022 and 4 August 2022, respectively; 13 larvae (ARS-20221001-1l to 1y), the same except collected on 26 August 2022; and one larva (ARS-20221001-1e), Kazan pond, Muğla, Türkiye, 37°1735.1″N; 28°1250.8″E, 480 m a.s.l, Alperen Kapankaya col., ectoparasitic on Bucephaloptera sp. (Orthoptera: Tettigoniidae: Tettigoniinae) on 6 September 2022. Four specimens, Mayamey mountains, Mayamey city, Semnan province, Iran, 36°2149.39″N, 55°4058.04″E, 1753 m a.s.l., Masoud Hakimitabar col., ectoparastitic on Oedipoda miniata (Orthoptera: Acrididae: Oedipodinae), 4 June 2018. Ten specimens (ARS-20221001-1a to 1j) are deposited in the Acarological Collection, Jalal Afshar Zoological Museum, Department of Plant Protection, Faculty of Agriculture, University of Tehran, Karaj, Iran, and 10 specimens (ARS-20221001-1k to 1o & 1u to 1y) are deposited in the Acarological Collection, Acarological Society of Iran, Department of Plant Protection, Faculty of Agriculture, University of Tehran, Karaj, Iran, five specimens (ARS-20221001-1p to 1t) and more than 20 extra specimens in the Acarological Collection, Department of Plant Protection, Faculty of Agriculture, Aydin Adnan Menderes University, Aydin, Türkiye; Iranian specimens will be deposited in the Acarological Collection, Acarological Society of Iran, Department of Plant Protection, Faculty of Agriculture, University of Tehran, Karaj, Iran.
Corrections in the descriptions of A. gonabadensis
Ahmadi et al. (2012) did not consider dorsal eupathidium with a Cp by mistake. We checked holotype of A. gonabadensis and confirm Noei and Honarmand (2020) which Ge I has only 8 normal setae and there are four sternalae 3a. Ahmadi et al. (2012) mentioned 9 normal setae on Ge I by mistake. Also, we confirm that there is a minute spine-like anterior hypostomal seta as in both species which is not reported in the original descriptions. Noei and Rabieh (2019) pointed the presence of spine-like anterior hypostomal seta as and showed its shape in Fig. 1 of their paper. Noei and Honarmand (2020) pointed out that there is anterior hypostomal seta as but nothing written about its shape. Moreover, leaf-like setae on dorsal idiosoma and blunt-ended setae on leg segments are not pointed out in the original description. Ahmadi et al. (2012) showed that solenidion on TFe I placed on 1/3 distal end of the segment whereas we checked, and it is placed on the middle of the segment. Also, there are three barbed setae on palpal tibia as pointed out by Noei and Honarmand (2020). Number of dorsal and ventral idiosomal setae are 42 and 12 whereas it was written 50 and 11 in the original description respectively.
Remarks
Abalakeus gonabadensis is unique among all species of Abalakeus by having leaf-like setae on idiosoma and blunt-ended normal setae on legs. Also, it differs from A. chekei in the number of solenidia on Ta I (1 vs. 2), Ge III (1 vs. 0), number of sternalae III (2 pairs vs. 1 pair), fD (36–44 vs. ~ 52), fV (8–12 vs. 17); and from A. lorestanicus in the shorter TFe I (80–107 vs. 116–131), Ge I (105–147 vs. 177–199), Ti I (120–162 vs. 192–213), TFe II (68–94 vs. 102–109), Ge II (81–120 vs. 138–155), Ti II (102–133 vs. 172–187), TFe III (79–117 vs. 128–138), Ge III (89–121 vs. 133–145), Ti III (118–227 vs. 262–301).
Key to species of the Abalakeus of the world (larva)
1. One solenidion on Ge III
...... 2
— Without solenidion on Ge III
...... A. checkei Southcott, 1994
2. Dorsal idiosomal setae leaf-like, some normal setae on leg segments truncate, Ti III 118–227, Ti I 120–162
...... A. gonabadensis Ahmadi et al., 2012
— Dorsal idiosomal setae not leaf-like, all normal setae on leg segment pointed, Ti III 262–301, Ti I 177–213
...... A. lorestanicus Saboori & Lachinani, 2003
Acknowledgements
This study was supported by Aydin Adnan Menderes University Research Foundation (ZRF-22006). The senior author was supported by the University of Tehran for a one-year sabbatical leave which is greatly appreciated. We are very grateful to Dr. Anne Baker (British Museum of Natural History, UK) for confirming number of normal setae on Ge II of all type specimens of A. chekei and sending its pictures. The authors would like to thank Cihan Aslı and Mesut Sayan for their valuable contributions to the collection of insect samples and separation of the mite specimens from their hosts. We have special thanks to Dr. Jaime Mayoral and Dr. Javad Noei for their useful comments which highly improved the quality of the paper.
References
- Ahmadi S., Hajiqanbar H., Saboori A. 2012. A new species of the genus Abalakeus (Acari, Erythraeidae) from Iran. Acta zool. Acad. Sci. Hung., 58(2): 169-176.
- Mąkol J., Sevsay, S. 2015. Abalakeus Southcott, 1994 is a junior synonym of ''plume-footed'' Eatoniana Cambridge, 1898 (Trombidiformes, Erythraeidae)-evidence from experimental rearing. Zootaxa, 3918(1): 92-112. https://doi.org/10.11646/zootaxa.3918.1.4
- Mayoral J.G., Barranco, P. 2006. The first report of the nymphal stage of the genus Abalakeus Southcott, 1994 from Spain. Syst. Appl. Acarol., 11: 125-128. https://doi.org/10.11158/saa.11.1.13
- Noei J., Rabieh M.M. 2019. New data on Nothrotrombidium, Southcottella and Eatoniana larvae (Acari: Trombellidae, Neothrombiidae, Erythraeidae) from Iran. Persian J. Acarol., 8(3): 179-187.
- Noei J., Honarmand A. 2020. Records of terrestrial parasitengone mites (Acari: Erythraeidae, Microtrombidiidae) ectoparasitic on grasshoppers from Khorasan Razavi province with new morphological data. Plant Pest Res. 10(4): 15-26 [In Persian with English summary].
- Saboori A., Khaustov A., Hakimitabar M., Hajiqanbar H. 2009. A new genus and species of larval Erythraeinae (Acari: Prostigmata: Erythraeidae) from Ukraine and the taxonomic state of Zhangiella. Zootaxa, 2203: 22-30. https://doi.org/10.11646/zootaxa.2203.1.2
- Saboori A., Lachinani P. 2003. Discovery of the genus Abalakeus Southcott (Acari: Erythraeidae) in Iran and description of a new species. Biologia, 58: 155-159.
- Sedghi A., Saboori A., Akrami M., Hakimitabar M. 2010. Second Iranian species of the genus Abalakeus Southcott (Acari: Erythraeidae). Internat. J. Acarol., 36: 431-436. https://doi.org/10.1080/01647951003733749
- Southcott R.V. 1994. A new larval Erythraeine mite (Acarina: Erythraeidae) from West Africa. Internat. J. Acarol., 20: 81-85. https://doi.org/10.1080/01647959408684005
- Walter D.E., Krantz G.W. 2009. Collection, rearing and preparing specimens. In: Krantz G.W., Walter, D.E. (Eds). A manual of Acarology, 3rd ed. Texas Tech University Press, pp. 83-97.
- Wohltmann A., Gabryś G., Mąkol J. 2007. Terrestrial Parasitengona inhabiting transient biotopes. In: Gerecke R. (Ed). Süßwasserfauna von Mitteleuropa. Vol. 7/2-1, Chelicerata, Araneae, Acari I. Munich, Germany: Elsevier GmbH, Spektrum Akademisher Verlag, pp. 158-240. https://doi.org/10.1007/978-3-662-55958-1_6
- Xu S.-Y., Yi T.-C., Guo J.-J., Jin D.-C. 2022. Two new species and a new combination of the subfamily Erythraeinae based on larval stage (Acari: Trombidiformes: Erythraeidae) from China. Insects, 13: 706. https://doi.org/10.3390/insects13080706
- Zhang Z.-Q., Zhang Y., Lin J. (2000) Discovery of the genus Abalakeus (Acari: Erythraeidae) in China and description of a new species from bamboo forests in Fujian, China. Syst. Appl. Acarol. Spec. Pub., 4: 69-80. https://doi.org/10.11158/saasp.4.1.8



2022-12-23
Date accepted:
2023-03-09
Date published:
2023-03-17
Edited by:
Mąkol, Joanna

This work is licensed under a Creative Commons Attribution 4.0 International License
2023 Saboori, Alireza; Kapankaya, Alperen; Cakmak, Ibrahim and Hakimitabar, Masoud
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



