A new genus and species of bat chiggers (Acariformes: Trombiculidae) from Kenya
Stekolnikov, Alexandr A.  1
1
1✉ Laboratory of Parasitic Arthropods, Zoological Institute of the Russian Academy of Sciences, Universitetskaya embankment 1, St. Petersburg 199034, Russia.
2022 - Volume: 62 Issue: 2 pages: 418-425
https://doi.org/10.24349/5n37-k5b9ZooBank LSID: E08F06DE-62D8-45C9-9C89-6BE882BD92E0
Original research
Keywords
Abstract
Introduction
According to Zajkowska et al. (2018), 397 species belonging to 72 genera of Trombiculidae were recorded on bat hosts and 18 genera of chiggers are considered as specific parasites of Chiroptera. Three of these 18 genera — Audytrombicula Vercammen-Grandjean, 1963, Tectumpilosum Feider, 1983, and Vergrandia Yunker & Jones, 1961 — are monotypic. Two new monotypic genera of bat chiggers were described recently — Bramkeria Bassini-Silva, Jacinavicius & Ochoa, 2021 and Batmanacarus Bassini-Silva, Jacinavicius & Ochoa, 2021. In general, chiggers specific to bat hosts include a set of forms with unclear taxonomic position (Daniel & Stekolnikov 2003a, b; Stekolnikov & Quetglas 2019). It is difficult to establish, which of their morphological characters suggest a monophyly rather than a parallel evolution. It concerns, for example, a reduction of the posterior scutal angles resulting in an extrascutal insertion of the posterolateral setae (peniscutum), reduction of eyes, and the number of palpal tarsal setae (from four to seven). An attempt of Vercammen-Grandjean (1968) to unite a series of different forms under the name Myotrombicula Womersley & Heaslip, 1943 seems improper; probably, a fractional classification would be more adequate.
During my revision of the personal collection of Vercammen-Grandjean deposited in Muséum d'histoire naturelle de Genève (MHNG, Geneva, Switzerland) I found a provisional type series (holotype and six paratypes) of a species labeled as Trombicula (T.) mombasa (nomen nudum). In the catalogue of types (Stekolnikov 2019), I placed it presumably in the genus Grandjeana Koçak & Kemal, 2009. However, a thorough study revealed that this species could not be placed in any of the known chigger genera. Therefore, I describe it here as a new genus and species.
These specimens were stored among the materials collected from different hosts in Mombasa (Kenya) by Minter, which included a series of chigger species with provisional names given by Vercammen-Grandjean — Arabacarus mombasai (nomen nudum), Helenicula lionoti (nomen nudum), Microtrombicula stenaspida (nomen nudum), Myotrombicula cor (nomen nudum), Myotrombicula tricoxalae (nomen nudum), and Sasatrombicula cardioderma (nomen nudum). With the exception of first two species, these chiggers were collected from bat hosts (Stekolnikov 2019). Their further examination is required to describe them as new species or to establish their identity with previously described species.
Material and methods
I examined the microscope slides in MHNG under a microscope Zeiss Axioscop (Carl Zeiss AG, Oberkochen, Germany) with a differential interference contrast. Drawings were made using a drawing tube; measurements were taken by an ocular micrometer. Calibration was done with the use of a stage micrometer. The label of the holotype was photographed using a Canon EOS 600D camera (Canon Inc., Tokyo, Japan). The terminology, abbreviations, and diagnostic formulas are used in the present work following Goff et al. (1982).
Minteracarus n. gen.
ZOOBANK: BFBE06BC-8911-4A5B-8D74-E80B3DFA7ABC ![]()
Diagnosis
SIF = 5B1N-N-2-3111.0000; fsp = 7.7.7; fCx = 1.1.1; fSt = 2.2; fPp = B/B/NBB. Idiosoma slightly contracted medially in engorged specimens; eyes absent; first two rows of dorsal idiosomal setae double or triple, total number of idiosomal setae exceeds 150. Cheliceral blade with tricuspid cap; galeal (deutorostral) seta nude; palpal claw (odontus) slender, with two small apical prongs; palpal tarsus with heavily branched dorsal seta of about same width and length as palpal claw, four setae bearing few branches, one nude seta, and large curved basal tarsala (ω) longer than palpal tarsus. Scutum small, bell-shaped, with puncta and prominent anterolateral shoulders; PL setae extrascutal; sensilla flagelliform, branched. Legs I and II with one pretarsala (ζ); subterminala (ζ) and nude parasubterminala (z) present; tarsalae I and II (ω) of about equal shape and length; famulus I (ε) distal to tarsala I; famulus II (ε) near and slightly proximal to tarsala II; microgenuala and microtibiala I (κ) long and thick, setiform; tibialae I (φ) very thick and long (longer than tarsala I), curved, striated; three long genualae I (σ); two tibialae II (φ), one genuala II (σ), one tibiala III (φ) and one genuala III (σ), mastisetae absent.
Type species
Minteracarus mombasa n. sp., designated here.
Etymology
The genus is named in honour of Dr. Donald M. Minter (The London School of Hygiene and Tropical Medicine) who collected the material of the new genus and species in Kenya in 1963.
Differential diagnosis
The new genus seems unique within the family Trombiculidae by the shape of both tibialae I (φ), which are very thick and long, longer than tarsala I (ω), and clearly striated. The presence of very long and thick microgenuala and microtibiala I (κ), comparable by their size with usual tibialae (φ) and genualae (σ), is also a rare character.
Minteracarus is similar to the monotypic genus of bat chiggers from Jamaica Bramkeria Bassini-Silva, Jacinavicius & Ochoa, 2021 by the extrascutal PLs, absence of eyes, presence of anterolateral shoulders of scutum, by the setation of legs, presence of nude setae on palpal tarsus, slender palpal claw with small apical prongs, and by a large number of idiosomal setae, with double or triple first two rows clearly separated from posterior dorsal idiosomal setae. However, Minteracarus differs from Bramkeria by the presence of six non-specialized palpal tarsal setae including dorsal seta of about same width as palpal claw, whereas other setae are much thinner, vs. seven palpal tarsal setae of about same width, all clearly thinner than the palpal claw; by two-pronged palpal claw vs. three-pronged; branched sensilla vs. nude; most of palpal setae branched vs. nude (fPp = B/B/NBB vs. N/N/NNN); and by the shape of scutum, with rounded posterior margin and AM placed at level of ALs, vs. concave posterior margin and AM placed anterior to ALs.
The new genus is also similar to the monotypic genus of bat chiggers from Cuba Tectumpilosum Feider, 1983, from which it differs by the presence of extrascutal PLs, six palpal tarsal setae vs. seven, two-pronged palpal claw vs. three-pronged, and by the rounded posterior scutal margin vs. slightly concave. The first row of dorsal idiosomal setae in Tectumpilosum is double, but all other rows are simple and distinct, with the following numbers of setae: 8-6-4-2-2; NDV = 82.
Minteracarus mombasa n. sp.
ZOOBANK: 969EC25B-FAA0-4F94-B996-7C0E8F97E9BF ![]()
(Figures 1, 2)
Grandjeana (?) mombasa (nomen nudum): Stekolnikov 2019: 36.
Diagnosis
SIF = 5B1N-N-2-3111.0000; fCx = 1.1.1; fSt = 2.2; fPp = B/B/NBB; DS = 100 – 119, V = 63 – 81, NDV = 166 – 194; Ip = 681 – 763; PL ≥ AM ≥ AL; eyes absent; scutum bell-shaped, with moderate puncta, with prominent anterolateral shoulders; PLs situated off scutum; sensilla flagelliform, with 3 – 4 long branches in distal half. Measurements given in Tables 1 and 2.
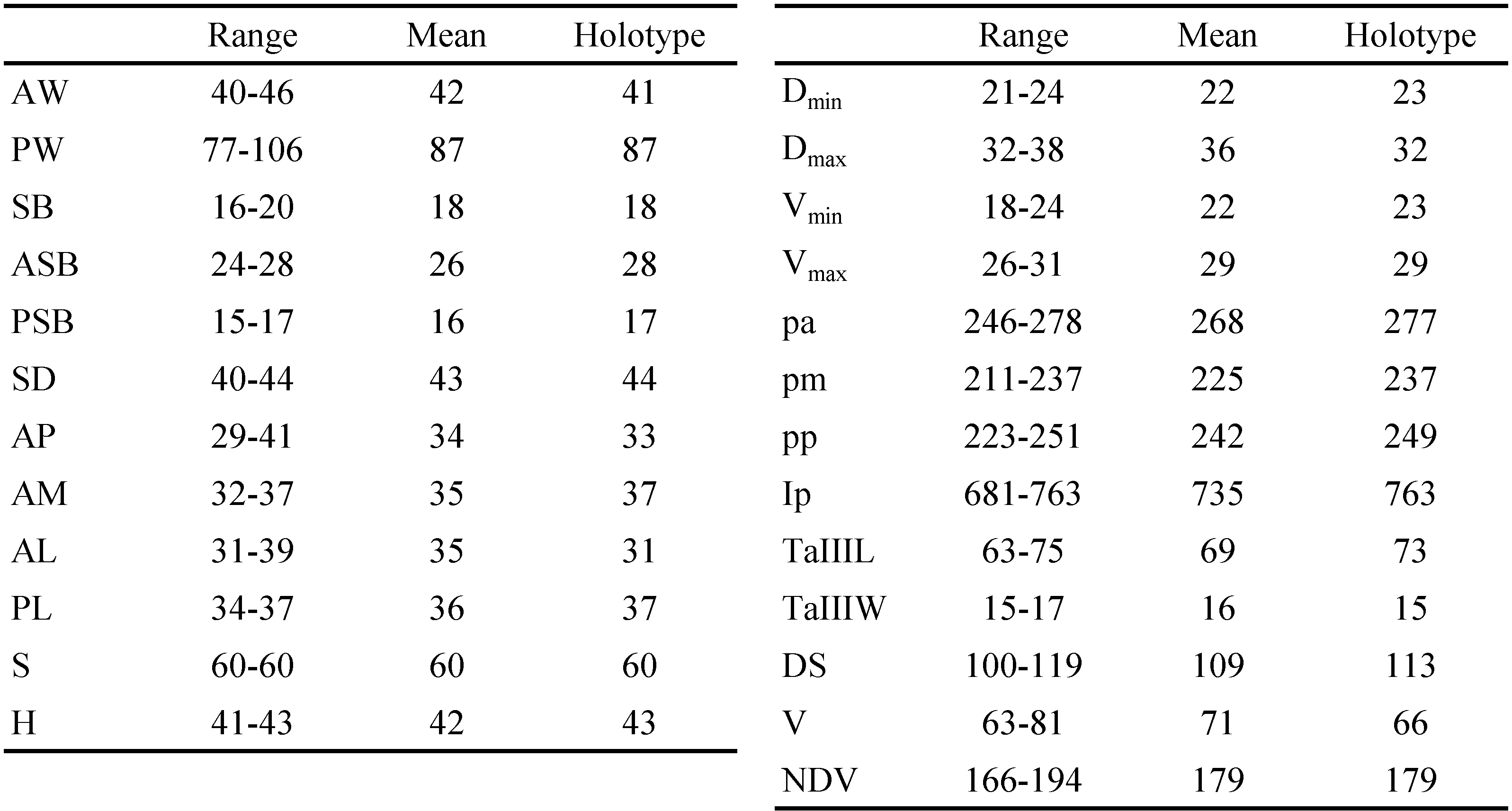





Description of larva




Idiosoma — (Figure 1) — Slightly contracted medially in engorged specimens; eyes absent; two pairs of humeral setae densely covered with long barbs; 1st posthumeral row (C excluding humeral setae) triple, with 35 – 41 setae; 2nd posthumeral row (D) double, sometimes triple in medial part, with 23 – 30 setae; distribution of other 36 – 47 setae by rows unclear; 1st and 2nd rows clearly isolated from each other and from posterior setae; total 100 – 119 short, heavily barbed dorsal idiosomal setae; four sternal and 63 – 81 ventral idiosomal setae, NDV = 166 – 194.
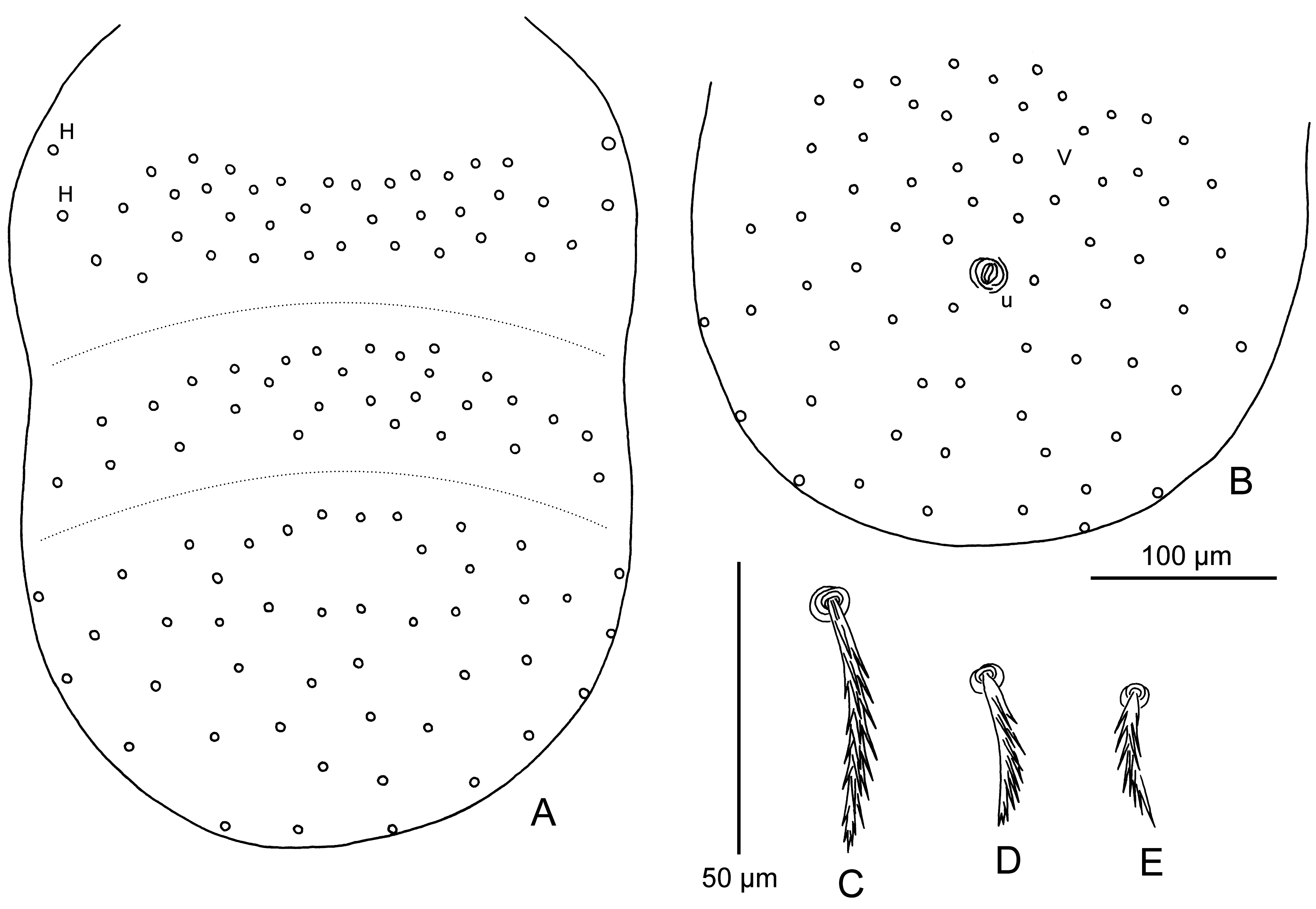
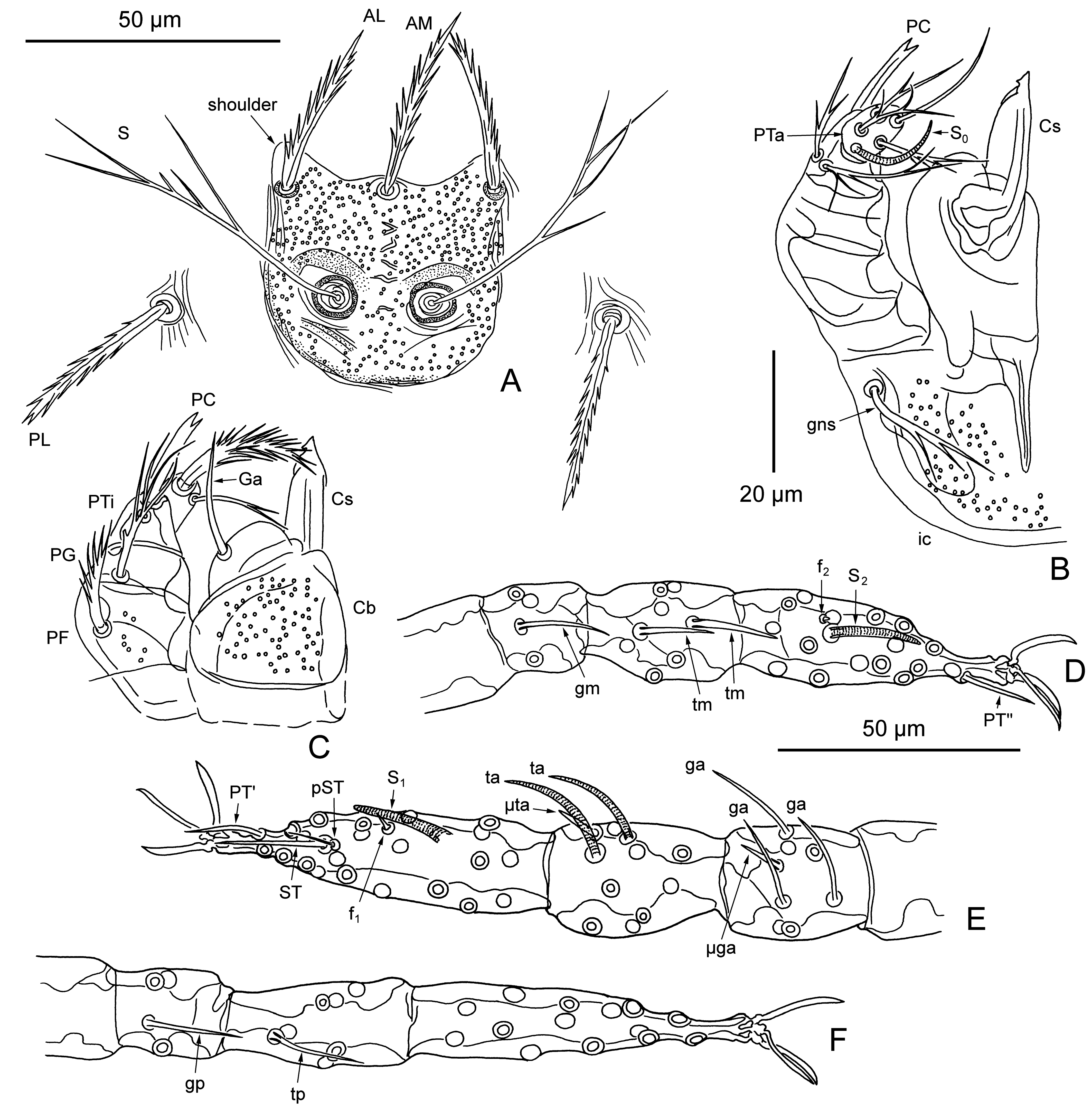
Gnathosoma — (Figure 2B, C) — Cheliceral base with sparse puncta; cheliceral blade with tricuspid cap; gnathobase (infracapitulum) with sparse puncta and one pair of branched gnathocoxal (tritorostral) setae; galeal (deutorostral) seta thick and nude; palpal claw (odontus) slender, with two short subequal prongs; palpal femur with few puncta and strong branched seta; palpal genu with branched seta; dorsal palpal tibial seta nude, lateral and ventral palpal tibial setae branched; palpal tarsus with 6 non-specialized setae, including 1 very thick, heavily barbed dorsal seta and 5 thin setae (4 bearing few branches and 1 nude), and long curved basal tarsala (ω).
Scutum — (Figure 2A) — Small, bell-shaped, with moderate puncta, with prominent anterolateral shoulders; anterior scutal margin sinuous, lateral margins with superimposed cuticular striations, posterior margin rounded; AM at level of ALs; PLs situated off scutum; all scutal setae covered with strong pointed barbs; sensilla flagelliform, with 3 – 4 long branches in distal half; arched crests present anterior to sensillary bases.
Legs — (Figure 2D – F) — All 7-segmented (with divided femur), with 1 pair of claws and claw-like empodium. Leg I: coxa with 1 branched setae (1B); trochanter 1B; basifemur 1B; telofemur 5B; genu 4B, 3 genualae (σ), long setiform microgenuala (κ); tibia 8B, 2 very long, curved, striated tibialae (φ), long setiform microtibiala (κ); tarsus 21B, tarsala (ω), famulus (ε) distal to tarsala, subterminala (ζ), nude parasubterminala (z), pretarsala (ζ). Leg II: coxa 1B; trochanter 1B; basifemur 2B; telofemur 4B; genu 3B, genuala (σ); tibia 6B, 2 tibialae (φ) in tandem; tarsus 16B, tarsala II (ω), famulus (ε) near and slightly proximal to tarsala, pretarsala (ζ). Leg III: coxa 1B; trochanter 1B; basifemur 2B; telofemur 3B; genu 3B, genuala (σ); tibia 6B, tibiala (φ); tarsus 14B.
Type material
Larval holotype (MHNG) (Figure 3), L:7263/1, ex ear of Cardioderma cor (Peters) (Chiroptera: Megadermatidae), KENYA, Mombasa, collected 7 Feb. 1963 by D.M. Minter, identified in 1972 by P.H. Vercammen-Grandjean; six paratypes (MHNG), L:7263/2 – 7, with same data as holotype.



Etymology
I retain the name given this species by Vercammen-Grandjean, which refers to the type locality, the city of Mombasa.
Remarks
The new species resembles an Ethiopian bat chigger Grandjeana kanuchi Kalúz & Ševčík, 2015 by the shape of scutum; therefore, Stekolnikov (2019) putatively placed it to the genus Grandjeana. However, Minteracarus differs from Grandjeana by the presence of six non-specialized setae on palpal tarsus vs. five, presence of subterminala and parasubterminala on leg tarsus I (absent in Grandjeana), and the presence of one pretarsala on leg tarsus I (vs. two in Grandjeana).
The date of collection was reconstructed by me from the slide number given by Vercammen-Grandjean (Stekolnikov 2019).
Discussion
Many of the traits characterizing Minteracarus n. gen. are scattered among the taxa of chiggers specific to bats and could be connected with the parasitism on these hosts. Thus, extrascutal PLs (peniscutum) are characteristic of the genera Trisetica Traub & Evans, 1950, Audytrombicula Vercammen-Grandjean, 1963, Nycterinastes Brennan & Reed, 1973, Octasternala Brown, 1990, and Bramkeria. Peniscutum is also present in some species of Trombigastia Vercammen-Grandjean & Brennan, 1957 (Vercammen-Grandjean & Fain 1958), in Trombicula (Anomalaspis) rhinopomae Vercammen-Grandjean, 1963 (species incertae sedis), and in Chiroptella (Neosomia) audyi (Vercammen-Grandjean & Nadchatram, 1965). In Grandjeana, different stages of ''peniscutalization'' are observed, including asymmetrical intermediate variants (Vercammen-Grandjean & Nadchatram 1963; Kalúz & Ševčík 2014, 2015). The absence of eyes is a characteristic of Alexfainia Yunker & Jones, 1961, Tectumpilosum, Bramkeria, and most Perates Brennan & Dalmat, 1960. A slender palpal claw with small apical prongs is present in the genera Trisetica (Stekolnikov & Quetglas 2019) and Bramkeria, in some species of Myotrombicula, Perates (Vercammen-Grandjean 1968), Trombigastia (Vercammen-Grandjean & Fain 1958), and Chiroptella Vercammen-Grandjean, 1960 (Nadchatram 1966).
Myotrombicula dusbabeki Daniel & Stekolnikov, 2003 shows the following characters common with Minteracarus: (1) absence of eyes, (2) slender palpal claw, (3) presence of multiple idiosomal setae (NDV = 243) with first two rows double and separated from other setae, (4) presence of one heavily branched dorsal seta on the palpal tarsus, almost as thick as palpal claw, whereas other palpal tarsal setae are much thinner, nude or having a few branches, (5) tibialae I longer than tarsala I (although much thinner). First three of these traits are also characteristic of Bramkeria and probably do not suggest an evolutionary closeness that might be assumed for the last two characters.
The setation of legs (not taking into account the shape of setae) in Minteracarus is usual for chigger mites (probably, plesiomorphic) and lets to separate this genus from the genera of bat chiggers having specific (autapomorphic) formulas of leg setation, such as Grandjeana (characterized by the presence of two pretarsalae I and the absence of leg subterminala), Sasatrombicula Vercammen-Grandjean, 1960 and Chiroptella Vercammen-Grandjean, 1960 (presence of additional genuala or mastigenuala III).
The shape of scutum is one of the most useful characters for the discrimination of chigger genera. Excluding from consideration the peniscutalization, we get such traits as the presence/absence of anterolateral shoulders (projections of the scutum anterior to ALs), the insertion of AM at level, anterior or posterior to AL, and the shape of posterior scutal margin (concave, straight, convex, angulate, rounded). By the shape of scutum, Minteracarus is most similar to Myotrombicula (Perates) macrozota (Brennan & Jones, 1960), which has prominent, acute anterolateral shoulders, AM situated at level of ALs, and convex, rounded posterior scutal margin (Vercammen-Grandjean 1968). Tectumpilosum also has prominent shoulders and AM situated at level of ALs, but its posterior scutal margin is slightly concave (Daniel & Stekolnikov 2003a); Bramkeria has rather small shoulders, AM situated anterior to ALs, and concave posterior scutal margin (Bassini-Silva et al. 2021). Sasatrombicula and Trombigastia have no shoulders (ALs are inserted in the scutal angles).
Therefore, the new genus seems most close to Myotrombicula in the sense of Vercammen-Grandjean (1968), i.e. including Perates as a subgenus. However, its status as a separate monotypic genus is undoubted.
Acknowledgements
I am grateful to Peter Schwendinger, curator of the collection of Arachnida at Muséum d'histoire naturelle de Genève (Geneva, Switzerland) for his valuable help during my visit to MHNG. I thank Stanislav Kalúz (Institute of Zoology, Slovak Academy of Sciences, Bratislava, Slovak Republic) and two anonymous reviewers for useful comments. This work was supported by the Ministry of Science and Higher Education of the Russian Federation (Grant No. 1021051603202-7).
References
- Bassini-Silva R., Huang-Bastos M., Welbourn C., Ochoa R., Barros-Battesti D.M., Jacinavicius F.C. 2021. A new genus of chiggers (Trombidiformes: Trombiculidae) from bats in Jamaica. Acta Parasitol., 66: 837-843. https://doi.org/10.1007/s11686-021-00336-0
- Daniel M., Stekolnikov A.A. 2003a. Chigger mites (Acari: Trombiculidae) new to the fauna of Cuba, with the description of two new species. Folia Parasitol., 50: 143-150. https://doi.org/10.14411/fp.2003.025
- Daniel M., Stekolnikov A.A. 2003b. To the fauna of chigger mites (Acari: Trombiculidae) parasitize bats in Cuba. Acarologia, 43: 29-37.
- Goff M.L., Loomis R.B., Welbourn W.C., Wrenn W.J. 1982. A glossary of chigger terminology (Acari: Trombiculidae). J. Med. Entomol., 19, 221-238. https://doi.org/10.1093/jmedent/19.3.221
- Kalúz S., Ševčík M. 2014. A new species of the genus Grandjeana (Koçak & Kemal, 2009) (Acari: Trombiculidae) from Mauritanian bat with a key to species of the genus. Int. J. Acarol., 40: 31-36. https://doi.org/10.1080/01647954.2013.875063
- Kalúz S., Ševčík M. 2015. A new species of Grandjeana (Acari: Trombiculidae) from heart-nosed bat (Chiroptera: Megadermatidae) in Ethiopia (Africa) with notes to biogeography of this genus. Biologia, 70: 380-385. https://doi.org/10.1515/biolog-2015-0043
- Nadchatram M. 1966. Revision of the bat-infesting chiggers of Chiroptella Vercammen-Grandjean (Acarina: Trombiculidae), with descriptions of two new larval species and nymph. J. Med. Entomol., 3: 19-28. https://doi.org/10.1093/jmedent/3.1.19
- Stekolnikov A.A. 2019. A catalogue of the holotypes of chigger mites (Acariformes: Trombiculidae) at the Natural History Museum of Geneva. Zootaxa, 4620: 1-71. https://doi.org/10.11646/zootaxa.4620.1.1
- Stekolnikov A.A., Quetglas J. 2019. Bat-infesting chiggers (Acariformes: Trombiculidae) of the Balearic Islands and new data on the genus Trisetica Traub et Evans, 1950. Folia Parasitol., 66: 017. https://doi.org/10.14411/fp.2019.017
- Vercammen-Grandjean P.H. 1968. Revision of the genus Myotrombicula Womersley and Heaslip, 1943 (Trombiculidae: Acarina). Acarologia, 10: 65-85.
- Vercammen-Grandjean P.H., Fain A. 1958. Les Trombiculidae parasites de Chiroptères. Révision du genre Trombigastia, description d'un ? Myotrombicula. Ann. Parasitol. Hum. Comp., 33: 5-35.
- Vercammen-Grandjean P.H., Nadchatram M. 1963. Le genre Trombicula et ses sous-genres (Trombiculidae-Acarina). Acarologia, 5: 384-393.
- Zajkowska P., Moniuszko H., Mąkol J. 2018. Host-parasite associations between bats (Mammalia: Chiroptera) and chiggers (Trombidiformes: Trombiculidae) - a review and checklist. Ann. Zool., 68: 97-178. https://doi.org/10.3161/00034541ANZ2018.68.1.006



222-02-12
Date accepted:
2022-04-12
Date published:
2022-04-13
Edited by:
Mąkol, Joanna

This work is licensed under a Creative Commons Attribution 4.0 International License
2022 Stekolnikov, Alexandr A.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



