Know your campus: salient research potential of prostigmatic soil mite fauna (Acariformes: Prostigmata, Endeostigmata) within university campus area
Laniecki, Ronald1 ; Kaźmierski, Andrzej2 ; Mąkol, Joanna3 ; Laniecka, Izabella4 and Magowski, Wojciech5
1✉ Department of Animal Taxonomy and Ecology, Adam Mickiewicz University in Poznań, Uniwersytetu Poznańskiego 6, PL- 61-714 Poznań, Poland.
2Department of Animal Morphology, Adam Mickiewicz University in Poznań, Uniwersytetu Poznańskiego 6, PL- 61-714 Poznań, Poland.
3Department of Invertebrate Systematics and Ecology, Wrocław University of Environmental and Life Sciences, Kożuchowska 5b, PL- 51-631 Wrocław, Poland.
4Department of Animal Morphology, Adam Mickiewicz University in Poznań, Uniwersytetu Poznańskiego 6, PL- 61-714 Poznań, Poland.
5Department of Animal Taxonomy and Ecology, Adam Mickiewicz University in Poznań, Uniwersytetu Poznańskiego 6, PL- 61-714 Poznań, Poland.
2021 - Volume: 61 Issue: 3 pages: 650-663
https://doi.org/10.24349/TTpf-5Q3lOriginal research
Keywords
Abstract
Introduction
Although the history of the study of prostigmatic and endeostigmatic mites extends over more than two hundred years (Krantz and Walter 2009), the knowledge about these arthropods is still poor. The number of known species represents only a small fraction of their real richness and even less is known about their ecology and behavior. Moreover, when comparing mites (excluding ticks) with spiders, the former seems to have a much higher diversity, than the latter, despite having similar number of described species. Furthermore, scientific literature devoted to all mites and available through the internet is much poorer, than that related to spiders (Walter and Proctor 2010). There are several reasons for this situation, e.g. mites, with special reference to Prostigmata, are numerous and highly diverse, and their identification is not easy. The literature is still scanty and spotty among the members of the suborder. The identification keys, or comprehensive revisions and monographs on many (if not most) groups of Prostigmata are still relatively scarce. Descriptions of many species are often curt and lacking vital diagnostic features, indicating a vast demand for redescriptions. Additionally, the number of competent specialists is limited and their regard in wide academic and scientific communities is meager (Walter and Proctor 2010).
Mites and particularly Prostigmata however, have great potential as model organisms in various research areas of modern biology. For instance, they can serve as models in variety of studies, including ecology (Walter and Proctor 2010) or experimental evolution (Belliure et al. 2010). Thus, the aim of this study was not only to contribute to the checklist of prostigmatic mites in Poland, but also to create a list of species accessible in our immediate vicinity, i.e. surroundings of the university facilities, and make them available for those interested in carrying out the research on these arachnids.
Material and methods
A total of 20 samples of soil litter and plant debris were collected from 10 localities representing 10 different habitats (see Table 1) in western part of Morasko Campus of Adam Mickiewicz University in Poznań (Figure 1). The first sampling was carried out in winter, whereas the second one in spring 2019.



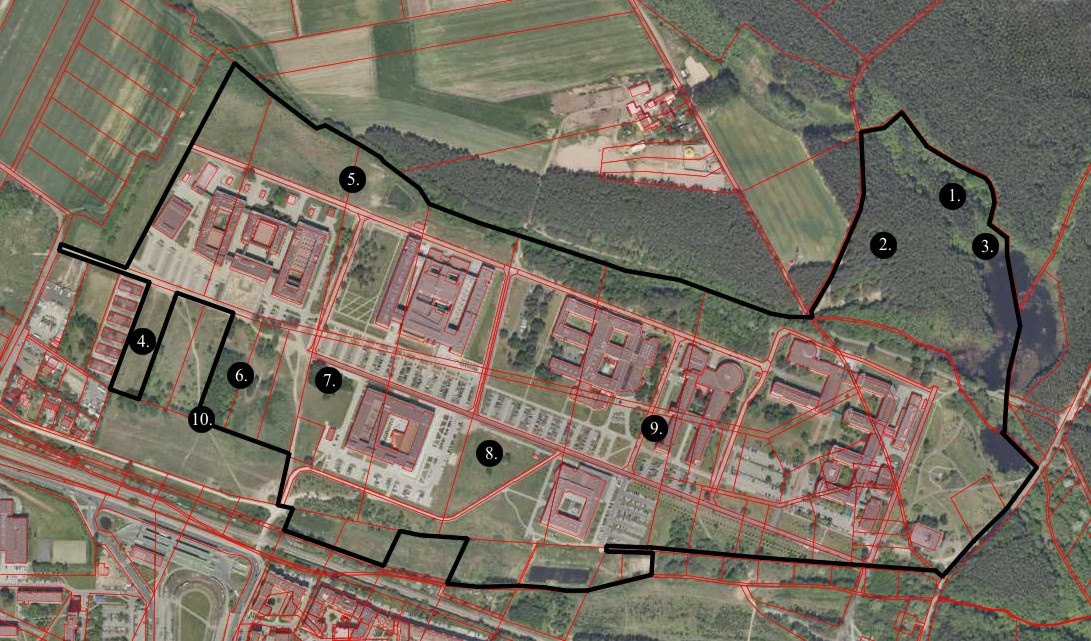


The sampling area is located in lowland in central-western Poland, whose landforms originated during glacial (Pleistocene), and postglacial (Holocene) periods. Most of the Morasko Campus are actually covered by buildings. Except for secondary plant community with Pinus sylvestris and Padus serotina as well as an alder forest, there are mostly regularly mowed lawns with sparsely distributed trees (mainly P. sylvestris and Betula pendula) and separated small tree stands.
Mites were extracted from samples using Berlese-Tullgren funnels (photo-eclectors) for 7-10 days and stored in 75% ethanol. Specimens of the family Nanorchestidae were additionally placed in 10% KOH to dissolve crystals of uric acid accumulated in the body, and then rinsed with distilled water. After that, specimens were mounted in Hoyer's medium on microscopic slides and heated for several days at 55 °C. They were studied with phase-contrast (Olympus BX 41) and interference-contrast (Olympus BX 51) microscopes supplied with a digital photographic camera Olympus E-5, and also with a phase-contrast microscope Nikon Eclipse 80i, equipped with DIC and DS-Fi3 camera system. Mite species were identified using keys and other taxonomic references by following authors: Bizarro et al. (2020), Booth et al. (1985), Fan Q.-H. (2004), Gabryś (2016), Gilyarov et al. (1978), Haitlinger (1986, 2002), Jesionowska (2010), Kaliszewski (1987, 1993), Kaźmierski (1998), Khaustov (2014), Krantz and Walter (2009), Łaydanowicz and Mąkol (2010), Magowski (1997), Mąkol (2010), Mąkol and Wohltmann (2012), Michocka (1987), Rack (1981), Smiley (1992), Stålstedt et al. (2016), Strandtmann (1971), Thor and Willmann (1941), Uusitalo (2010), Wohltmann et al. (2007), and Zacharda (1980).
The species found are divided into three categories, namely: (a) fully identified species, (b) those resembling known species but its identity uncertain (hence marked ''cf.''), and (c) those whose identity could not be ascertained for technical reasons (bad position, missing diagnostic features) but not closely resembling known species (hence marked ''sp. 1'', ''sp. 2'', etc.). Species omitted from the count are individuals damaged beyond species recognition. Only fully named species are counted as new for Polish or European faunas. Genera Echinoeupodes, Neoprotereunetes, Archidispus, and Pygmodispus are also listed as new for respective faunas (for Poland or Europe) even though their representing species were not identified and therefore only indicated as ''sp.'' (thus in abovementioned category c). In the list hereunder of recorded taxa the abbreviations ''NP'' and ''NE'' refer to taxa new to Polish and European faunas, respectively. Localities and seasons of sampling (W – winter, S – spring) following the number of the locality are marked according to the Table 1. The following symbols refer to the instars: female – ♀, male – ♂, tritonymph – tn, deutonymph – dn, protonymph – pn, larva – lv, unidentified nymph – n, unidentified instar – ?.
The collection of samples and specimens is deposited in the Department of Animal Taxonomy and Ecology, Adam Mickiewicz University in Poznań, except for Erythraeidae, which are deposited at the Department of Invertebrate Systematics and Ecology, Wrocław University of Environmental and Life Sciences.
Results
In the course of our study 868 individuals of 21 families belonging to 58 genera and 139 species were found. Selected representatives of the suborder Prostigmata are shown in Figure 2. The list of family level taxa is provided in systematic order following Krantz and Walter (2009); the list of genera and species within particular family – in alphabetical order.



Superorder: Acariformes (=Actinotrichida)
- Order: Trombidiformes
- Suborder: Prostigmata (=Actinedida)
- Supercohort: Eupodides
- Superfamily: Bdelloidea Dugès, 1834
- Family: Bdellidae Dugès, 1834
- Subfamily: Bdellinae Dugès, 1834
- Genus: Bdella Latreille, 1795
- Bdella dispar C.L. Koch, 1839. Locality: 10W: 1 lv
- Bdella semiscutata Thor, 1931. Locality: 7W: 5 ♀♀
- Bdella dispar C.L. Koch, 1839. Locality: 10W: 1 lv
- Genus: Biscirus Thor, 1913
- Biscirus silvaticus (Kramer, 1881). Localities: 5S: 1 n 2 lv; 6S: 2 n; 7S: 5 n; 8S: 2 lv
- Biscirus silvaticus (Kramer, 1881). Localities: 5S: 1 n 2 lv; 6S: 2 n; 7S: 5 n; 8S: 2 lv
- Genus: Bdella Latreille, 1795
- Subfamily: Cytinae Grandjean, 1938
- Genus: Cyta von Heyden, 1804
- Cyta latirostris (Hermann, 1804)
Localities: 3S: 3 ♀♀; 4W: 2 lv; 6W: 3 n, 6S: 3 ♀♀; 7S: 2 ♀♀ 1 lv; 10S: 3 n
- Cyta sp.. Localities: 9W: 1 ♀
- Cyta latirostris (Hermann, 1804)
- Genus: Cyta von Heyden, 1804
- Family: Cunaxidae Thor, 1902
- Subfamily: Bonziinae Den Heyer, 1978
- Genus: Bonzia Oudemans, 1927
- Bonzia halacaroides Oudemans, 1927. Locality: 3W: 1 ♀
- Bonzia halacaroides Oudemans, 1927. Locality: 3W: 1 ♀
- Genus: Bonzia Oudemans, 1927
- Subfamily: Cunaxinae Thor, 1902
- Genus: Armascirus Den Heyer, 1978 NP
- Armascirus bakeri Smiley, 1992 NP. Localities: 5S: 1 lv; 6S: 2 lv; 10W: 1 lv
- Armascirus bakeri Smiley, 1992 NP. Localities: 5S: 1 lv; 6S: 2 lv; 10W: 1 lv
- Genus: Cunaxa von Heyden, 1826
- Cunaxa setirostris (Hermann, 1804). Localities: 1W: 1 lv; 3S: 2 n 1 lv; 4W: 1 ♀; 5W: 1n, S: 1 ♀; 6W: 4 ♀♀, 6S: 1n; 7S: 2 lv; 8W: 1 ♀
- Cunaxa setirostris (Hermann, 1804). Localities: 1W: 1 lv; 3S: 2 n 1 lv; 4W: 1 ♀; 5W: 1n, S: 1 ♀; 6W: 4 ♀♀, 6S: 1n; 7S: 2 lv; 8W: 1 ♀
- Genus: Dactyloscirus Berlese, 1916
- Dactyloscirus inermis (Trägårdh, 1905). Locality: 2S: 1 ♀ 1 n 4 lv
- Dactyloscirus inermis (Trägårdh, 1905). Locality: 2S: 1 ♀ 1 n 4 lv
- Genus: Armascirus Den Heyer, 1978 NP
- Subfamily: Cunaxoidinae Den Heyer, 1978
- Genus: Cunaxoides Baker et Hoffmann, 1948
- Cunaxoides kielczewskii Michocka, 1982. Localities: 4W: 1 lv; 5W: 1 dn 2 pn 2 lv, 5S: 1 ♀; 6S: 2 ♀♀; 8S: 2 ♀; 10S: 1 ♀
- Cunaxoides kielczewskii Michocka, 1982. Localities: 4W: 1 lv; 5W: 1 dn 2 pn 2 lv, 5S: 1 ♀; 6S: 2 ♀♀; 8S: 2 ♀; 10S: 1 ♀
- Genus: Cunaxoides Baker et Hoffmann, 1948
- Superfamily: Eupodoidea C.L. Koch, 1842
- Family: Cocceupodidae Jesionowska, 2010
- Genus: Cocceupodes Thor, 1934
- Cocceupodes mollicellus (C.L. Koch, 1838). Localities: 1W: 7 ♀♀ 2 ♂♂, 1S: 1 ♀ 1 pn 1 lv; 2W: 1 pn, 2S: 1 ♂; 3W: 4 ♀♀ 2 dn 2 pn; 5W: 1 ♀
- Cocceupodes mollicellus (C.L. Koch, 1838). Localities: 1W: 7 ♀♀ 2 ♂♂, 1S: 1 ♀ 1 pn 1 lv; 2W: 1 pn, 2S: 1 ♂; 3W: 4 ♀♀ 2 dn 2 pn; 5W: 1 ♀
- Genus: Filieupodes Jesionowska, 2010
- Filieupodes filiformis Jesionowska, 2010. Locality: 1W: 1 ♀ 4 ?; 1S: 1 ♀
- Filieupodes filistellatus Jesionowska, 2010. Localities: 7S: 4 ♀♀; 8W: 4 ♀♀ 2 ♂♂; 9S: 5 ♀♀; 10S: 1 ♀
- Filieupodes shepardi (Strandtmann, 1971) NP NE. Locality: 7W: 6 ♀♀
- Filieupodes sp. 1. Locality: 3S: 2 dn
- Filieupodes sp. 2. Locality: 8S: 1 ♀ 1 tn
- Filieupodes filiformis Jesionowska, 2010. Locality: 1W: 1 ♀ 4 ?; 1S: 1 ♀
- Genus: Linopodes C.L. Koch, 1835
- Linopodes sp.. Localities: 2W: 1 ♀ 1 ♂ 2 tn; 3S: 3 ?; 5W: 1 ♀
- Linopodes sp.. Localities: 2W: 1 ♀ 1 ♂ 2 tn; 3S: 3 ?; 5W: 1 ♀
- Genus: Cocceupodes Thor, 1934
- Family: Eupodidae C.L. Koch, 1842
- Subfamily: Benoinyssinae Fain, 1958
- Genus: Benoinyssus Fain, 1958
- Benoinyssus cf. occonori Bizarro, Wurlitzer et Da Silva, 2020. Localities: 4W: 1 ?; 7S: 2 ♀♀
- Benoinyssus cf. serratus Olivier et Theron, 1997. Localities: 3S: 3 ♀♀ 1 tn; 4S: 1 ♀; 6S: 2 ♀♀ 1 ?; 9S: 1 tn
- Benoinyssus sp.. Localities: 8S: 1 ♀
- Benoinyssus cf. occonori Bizarro, Wurlitzer et Da Silva, 2020. Localities: 4W: 1 ?; 7S: 2 ♀♀
- Genus: Benoinyssus Fain, 1958
- Subfamily: Eupodinae C.L. Koch, 1842
- Genus: Caleupodes Baker, 1987
- Caleupodes reticulatus Baker, 1987. Locality: 2W: 4 ♀♀
- Caleupodes reticulatus Baker, 1987. Locality: 2W: 4 ♀♀
- Genus: Echinoeupodes Khaustov, 2017 NP
- Echinoeupodes sp.. Locality: 5S: 1 dn
- Echinoeupodes sp.. Locality: 5S: 1 dn
- Genus: Eupodes C.L. Koch, 1835
- Eupodes voxencollinus Thor, 1934. Localities: 1W: 1 ♀, 1S: 2 ♀♀ 3 ♂♂; 2W: 6 ♀♀ 1 ♂; 3S: 4 ♀♀; 5W: 4 ♀♀ 1 ♂, 5S: ♀ 1 ♂; 6W: 3 ♀♀ 2 ♂♂, 6S: 3 ♀♀ 3 ♂♂ 1 ?; 7S: 1 ♀; 8S: 1 ♀ 1 tn; 10W: 5 ♀♀ 1 tn
- Eupodes cf. voxencollinus Thor, 1934. Locality: 4S: 4 ♀♀; 7S: 1 dn; 10S: 1 ♀
- Eupodes voxencollinus Thor, 1934. Localities: 1W: 1 ♀, 1S: 2 ♀♀ 3 ♂♂; 2W: 6 ♀♀ 1 ♂; 3S: 4 ♀♀; 5W: 4 ♀♀ 1 ♂, 5S: ♀ 1 ♂; 6W: 3 ♀♀ 2 ♂♂, 6S: 3 ♀♀ 3 ♂♂ 1 ?; 7S: 1 ♀; 8S: 1 ♀ 1 tn; 10W: 5 ♀♀ 1 tn
- Genus: Neoprotereunetes Fain et Camerik, 1994 NP
- Neoprotereunetes sp. 1. Locality: 1W: 2 lv, 1S: 2 lv
- Neoprotereunetes sp. 2. Locality: 4W: 1 lv
- Neoprotereunetes sp. 1. Locality: 1W: 2 lv, 1S: 2 lv
- Genus: Pseudoeupodes Khaustov, 2014 NP
- Pseudoeupodes porosus Khaustov, 2014 NP. Locality: 4W: 1 pn; 8W: 6 ♀♀
- Pseudoeupodes porosus Khaustov, 2014 NP. Locality: 4W: 1 pn; 8W: 6 ♀♀
- Genus: Caleupodes Baker, 1987
- Family: Penthaleidae Oudemans, 1931
- Genus: Penthaleus C.L. Koch, 1836
- Penthaleus cf. major (Dugès, 1834). Localities: 4W: 5 ♀♀, 4S: 1 ?; 8W: 1 ♀; 9S: 4 ♀♀ 1 ♂; 10W: 1 ♀
- Penthaleus cf. major (Dugès, 1834). Localities: 4W: 5 ♀♀, 4S: 1 ?; 8W: 1 ♀; 9S: 4 ♀♀ 1 ♂; 10W: 1 ♀
- Genus: Penthaleus C.L. Koch, 1836
- Family: Rhagidiidae Oudemans, 1922
- Genus: Coccorhagidia, Thor, 1934
- Coccorhagidia clavifrons (Canestrini, 1886). Locality: 8S: 1 ♀
- Coccorhagidia clavifrons (Canestrini, 1886). Locality: 8S: 1 ♀
- Genus: Crassocheles Zacharda, 1980 NP
- Crassocheles virgo Zacharda, 1980 NP. Locality: 4W: 1 ♀; 7W: 1 ♀
- Crassocheles virgo Zacharda, 1980 NP. Locality: 4W: 1 ♀; 7W: 1 ♀
- Genus: Latoempodia Zacharda, 1980 NP
- Latoempodia similis Zacharda, 1980 NP. Locality: 4W: 2 ♀♀; 7W: 1 ♀
- Latoempodia similis Zacharda, 1980 NP. Locality: 4W: 2 ♀♀; 7W: 1 ♀
- Genus: Parallelorhagidia Zacharda, 1980
- Parallelorhagidia sp.. Locality: 7S: 1 pn
- Parallelorhagidia sp.. Locality: 7S: 1 pn
- Genus: Poecilophysis Cambridge, 1876
- Poecilophysis (Dentocheles) pratensis (C.L. Koch, 1835). Locality: 1S: 1 dn
- Poecilophysis (Procerocheles) faeroensis (Trägårdh, 1931) NP*** Locality: 2W: 1 tn 1 dn
- Poecilophysis (Dentocheles) pratensis (C.L. Koch, 1835). Locality: 1S: 1 dn
- Genus: Shibaia Zacharda, 1980
- Shibaia longisensilla (Shiba, 1969). Locality: 5S: 1 dn
- Shibaia longisensilla (Shiba, 1969). Locality: 5S: 1 dn
- Genus: Thoria Zacharda, 1980 NP
- Thoria brevisensilla Zacharda, 1980 NP. Locality: 4W: 1 tn
- Thoria uniseta (Thor, 1909) NP. Localities: 6S: 1 ♀ 1 tn; 7S: 4 ♀♀ 2 ?
- Thoria cf. uniseta (Thor, 1909). Locality: 7W: 2 pn
- Thoria brevisensilla Zacharda, 1980 NP. Locality: 4W: 1 tn
- Genus: Coccorhagidia, Thor, 1934
- Superfamily: Tydeoidea Kramer, 1877
- Family: Ereynetidae Oudemans, 1931
- Subfamily: Ereynetinae Oudemans, 1931
- Genus: Ereynetes Berlese, 1883
- Ereynetes sp.. Locality: 1W: 1 ?
- Ereynetes sp.. Locality: 1W: 1 ?
- Genus: Ereynetes Berlese, 1883
- Family: Iolinidae Pritchard, 1956
- Subfamily: Tydaeolinae André, 1979
- Genus: Coccalicus Willmann, 1952
- Coccalicus lukoschusi (André, 1980). Locality: 7S: 1 ♀
- Coccalicus lukoschusi (André, 1980). Locality: 7S: 1 ♀
- Genus: Microtydeus Thor, 1931
- Microtydeus beltrani Baker, 1944. Localities: 1W: 1 ♀; 3S: 2 ♀♀ 1 tn; 4S: 3 ♀♀; 5W: 1 ♀; 7S: 2 ♀♀; 9S: 3 ♀♀ 1 tn
- Microtydeus subterraneus Wood, 1965 NP. Localities: 6S: 3 ♀♀ 1 tn 1 lv; 7S: 2 ♀♀
- Microtydeus subtilis (C.L. Koch, 1838). Localities: 3S: 1 ♀; 7S: 1 ♀
- Microtydeus beltrani Baker, 1944. Localities: 1W: 1 ♀; 3S: 2 ♀♀ 1 tn; 4S: 3 ♀♀; 5W: 1 ♀; 7S: 2 ♀♀; 9S: 3 ♀♀ 1 tn
- Genus: Tydaeolus Berlese, 1910
- Tydaeolus frequens (Grandjean, 1938). Locality: 2W: 1 ♀
- Tydaeolus sphaeroclaviger Kuznetzov, 1972 . Localities: 4S: 2 ♀♀; 8S: 4 ♀♀
- Tydaeolus frequens (Grandjean, 1938). Locality: 2W: 1 ♀
- Genus: Coccalicus Willmann, 1952
- Family: Triophtydeidae André, 1979
- Subfamily: Triophtydeinae André, 1979
- Genus: Triophtydeus Thor, 1932
- Triophtydeus immanis Kuznetzov, 1973. Localities: 3W: 3 ♀♀ 1 dn 5 ?; 7S: 1 ♀
- Triophtydeus sp.. Locality: 2S: 2 dn
- Triophtydeus immanis Kuznetzov, 1973. Localities: 3W: 3 ♀♀ 1 dn 5 ?; 7S: 1 ♀
- Genus: Triophtydeus Thor, 1932
- Family: Tydeidae Kramer, 1877
- Subfamily: Tydeinae Kramer, 1877
- Genus: Lorryia Oudemans, 1925
- Lorryia arkadiensis Panou et Emmanouel, 1995. Locality: 10W: 2 ♀♀
- Lorryia brevicula (C.L. Koch, 1838). Locality: 1W: 4 ♀♀
- Lorryia cf. danielsoni (Momen et Lindquist, 1995). Localities: 2W: 2 ♂♂ 3 tn; 3S: 2 ♀♀ 1 ♂; 4S: 1 ♀; 5W: 1 tn; 6S: 1 ♀
- Lorryia devexa (Kuznetzov, 1973). Locality: 2W: 2 ♀♀ 1 tn, 2S: 1 tn
- Lorryia grandiinsignia Kaźmierski, 1991. Localities: 4S: 3 ♀♀ 1 tn; 6W: 1 ♀
- Lorryia maga (Kuznetzov, 1973). Locality: 1W: 1 n
- Lorryia matura (Livshitz, 1973). Localities: 4S: 1 dn; 9W: 2 ♀♀ 1 ♂ 1 tn, 9S: 1 ♀
- Lorryia obnoxia (Kuznetzov et Zapletina, 1972) NP. Locality: 6S: 1 ?
- Lorryia cf. obnoxia (Kuznetzov et Zapletina, 1972). Locality: 7S: 1 ♀
- Lorryia cf. obstinata (Livshitz, 1973). Locality: 6W: 4 tn 1 dn
- Lorryia opifera (Kuznetzov, 1973). Locality: 7S: 1 ♀ 1 dn
- Lorryia oregonensis (Baker, 1970). Locality: 1W: 1 ♀ 1 ♂
- Lorryia reticuloinsignia Kaźmierski et Panou, 1997 NP. Locality: 4W: 2 ♀♀ 3 ♂♂ 1 dn, 4S: 1 ♂
- Lorryia sororcula Kuznetzov, 1975 NP. Locality: 6W: 5 ♀♀ 1 ♂, 6S: 3 ♀♀
- Lorryia cf. tridactyla (Weis-Fogh, 1947). Locality: 5W: 3 tn
- Lorryia unigena (Livshitz, 1973). Localities: 8W: 1 ♀ 1 tn; 10W: 4 ♀♀
- Lorryia varsoviensis Kaźmierski, 1978. Localities: 9S: 6 ♀♀; 10W: 1 ♀
- Lorryia volgini (Kuznetzov, 1973). Locality: 10S: 3 ♀♀
- Lorryia arkadiensis Panou et Emmanouel, 1995. Locality: 10W: 2 ♀♀
- Genus: Metalorryia André, 1980
- Metalorryia delicata (Kuznetzov, 1971). Locality: 5W: 2 tn
- Metalorryia delicata (Kuznetzov, 1971). Locality: 5W: 2 tn
- Genus: Tydeus C. L. Koch, 1835
- Tydeus californicus (Banks, 1904) sensu Baker 1970. Locality: 7S: 1 ♀
- Tydeus kochi (Oudemans, 1928) sensu Baker 1970. Localities: 4S: 1 dn; 5S: 2 ♀♀ 1 ♂ 1 tn 1 dn; 6S: 1 ♀ 2 ♂♂; 7S: 3 ♀♀ 1 ♂ 1 tn; 9S: 1 ♂
- Tydeus lindquisti (Marshall, 1970). Localities: 7W: 2 ♀♀ 4 ♂♂; 8W: 3 ♀♀ 3 ♂♂, S: 4 ♀♀ 1 dn; 10W: 2 ♀♀ 1 ♂, 10S: 2 ♀♀ 7 ♂♂
- Tydeus sp.. Locality: 2S: 2 pn
- Tydeus californicus (Banks, 1904) sensu Baker 1970. Locality: 7S: 1 ♀
- Genus: Lorryia Oudemans, 1925
- Supercohort: Anystides
- Cohort: Parasitengonina
- Subcohort: Erythraiae
- Superfamily: Erythraeoidea Robineau-Desvoidy, 1828
- Family: Erythraeidae Robineau-Desvoidy, 1828
- Subfamily: Balaustiinae Grandjean, 1947
- Genus: Balaustium von Heyden, 1826
- Balaustium cf. nikae Haitlinger, 1996. Localities: 4S: 2 dn 4 lv; 7S: 1 dn; 8S: 3 dn 2 lv; 9S: 3 dn 4 lv; 10S: 3 dn
- Balaustium sp.. Locality: 9W: 1 lv
- Balaustium cf. nikae Haitlinger, 1996. Localities: 4S: 2 dn 4 lv; 7S: 1 dn; 8S: 3 dn 2 lv; 9S: 3 dn 4 lv; 10S: 3 dn
- Genus: Balaustium von Heyden, 1826
- Subfamily: Callidosomatinae Southcott, 1957
- Genus: Abrolophus Berlese, 1891
- Abrolophus artemisiae (Schrank, 1803). Localities: 3W: 1 ♀ 1 dn; 4S: 1 ♀; 10W: 2 ♀♀, 10S: 2 ♀♀ 2 ♂♂
- Abrolophus cf. aitapensis (Southcott, 1948). Localities: 5S: 1 lv; 8W: 3 lv, 8S: 3 lv
- Abrolophus miniatus (Hermann, 1804). Localities: 3S: 1 dn; 6S: 2 ♀♀ 5 dn; 7S: 2 dn
- Abrolophus rudaensis (Haitlinger, 1986). Locality: 4W: 3 lv
- Abrolophus rhopalicus (C.L. Koch, 1837). Locality: 8W: 1 ♀ 2 ♂♂
- Abrolophus artemisiae (Schrank, 1803). Localities: 3W: 1 ♀ 1 dn; 4S: 1 ♀; 10W: 2 ♀♀, 10S: 2 ♀♀ 2 ♂♂
- Genus: Abrolophus Berlese, 1891
- Subfamily: Erythraeinae Robineau-Desvoidy, 1828
- Genus: Erythraeus Latreille, 1806
- Erythraeus phalangoides (De Geer, 1778). Locality: 10S: 1 dn
- Erythraeus phalangoides (De Geer, 1778). Locality: 10S: 1 dn
- Genus: Erythraeus Latreille, 1806
- Subfamily: Leptinae Billberg, 1820
- Genus: Leptus Latreille, 1796
- Leptus molochinus (C.L. Koch, 1837). Locality: 5W: 3 dn
- Leptus molochinus (C.L. Koch, 1837). Locality: 5W: 3 dn
- Genus: Leptus Latreille, 1796
- Supercohort: Eleutherengonides
- Cohort: Raphignathina
- Superfamily: Raphignathoidea Kramer, 1877
- Family: Caligonellidae Grandjean, 1944
- Genus: Caligonella Berlese, 1910
- Caligonella humilis (C.L. Koch, 1838). Locality: 10W: 1 ♀, 10S: 3 ♀♀ 1 dn
- Caligonella humilis (C.L. Koch, 1838). Locality: 10W: 1 ♀, 10S: 3 ♀♀ 1 dn
- Genus: Caligonella Berlese, 1910
- Family: Eupalopsellidae Willmann, 1952 NP
- Genus: Eupalopsellus Sellnick, 1950 NP
- Eupalopsellus trudis Summers, 1960 NP. Localities: 3S: 2 ♀♀; 4S: 2 ♀♀; 5W: 2 ♀♀; 7S: 2 ♀♀
- Eupalopsellus trudis Summers, 1960 NP. Localities: 3S: 2 ♀♀; 4S: 2 ♀♀; 5W: 2 ♀♀; 7S: 2 ♀♀
- Genus: Eupalopsellus Sellnick, 1950 NP
- Family: Raphignathidae Kramer, 1877
- Genus: Raphignathus Dugès, 1833
- Raphignathus gracilis (Rack, 1962). Localities: 4W: 6 ♀♀; 9W: 4 ♀♀ 2 ♂♂, 9S: 3 ♀♀
- Raphignathus gracilis (Rack, 1962). Localities: 4W: 6 ♀♀; 9W: 4 ♀♀ 2 ♂♂, 9S: 3 ♀♀
- Genus: Raphignathus Dugès, 1833
- Family: Stigmaeidae Oudemans, 1931
- Genus: Agistemus Summers, 1960 NP
- Agistemus gratus Kuznetzov et Vainshtein, 1977 NP. Locality: 5W: 1 ♀
- Agistemus gratus Kuznetzov et Vainshtein, 1977 NP. Locality: 5W: 1 ♀
- Genus: Eustigmaeus Berlese, 1910
- Eustigmaeus coronaria (Kuznetzov, 1976) NP. Locality: 8S: 1 ♀
- Eustigmaeus cf. formosus Kaźmierski et Dończyk, 2003. Locality: 8S: 3 ♀♀
- Eustigmaeus pinnatus (Kuznetzov, 1977). Locality: 10W: 1 ?
- Eustigmaeus cf. pinnatus (Kuznetzov, 1977). Locality: 10S: 1 ♀
- Eustigmaeus cf. frigida (Habeeb, 1958). Locality: 5S: 2 ♀♀
- Eustigmaeus segnis (C.L. Koch, 1836). Locality: 7S: 1 ♀; 9S: 2 ♀♀ 1 dn
- Eustigmaeus coronaria (Kuznetzov, 1976) NP. Locality: 8S: 1 ♀
- Genus: Cheylostigmaeus Willmann, 1951
- Cheylostigmaeus sp.. Locality: 5S: 2 ♀♀
- Cheylostigmaeus sp.. Locality: 5S: 2 ♀♀
- Genus: Stigmaeus C.L. Koch, 1836
- Stigmaeus corticeus Kuznetzov et Vainshtein, 1977. Localities: 2W: 2 ♀♀; 4W: 5 ♀♀; 6W: 6 ♀♀ 1 dn, 6S: 1 ?; 8W: 6 ♀♀; 10W: 6 ♀♀
- Stigmaeus fusus Summers, 1962 NP. Localities: 8S: 7 ♀♀; 9S: 6 ♀♀; 10S: 6 ♀♀
- Stigmaeus glypticus Summers, 1962 NP. Locality: 8W: 6 ♀♀
- Stigmaeus longipilis (Canestrini, 1889) NP. Localities: 1S: 5 ♀♀; 4S: 5 ♀♀
- Stigmaeus cf. longisetis Wood, 1967. Locality: 1W: 1 ♀
- Stigmaeus pilatus Kuznetzov, 1978. Locality: 7S: 1 ♀
- Stigmaeus cf. pulchellus Kuznetzov, 1978. Locality: 10S: 1 ♀
- Stigmaeus rhodomelas Berlese, 1910 NP. Locality: 5S: 3 ♀♀
- Stigmaeus sphagneti (Hull, 1918). Locality: 5S: 1♀
- Stigmaeus corticeus Kuznetzov et Vainshtein, 1977. Localities: 2W: 2 ♀♀; 4W: 5 ♀♀; 6W: 6 ♀♀ 1 dn, 6S: 1 ?; 8W: 6 ♀♀; 10W: 6 ♀♀
- Genus: Agistemus Summers, 1960 NP
- Cohort: Heterostigmatina
- Superfamily: Scutacaroidea Oudemans, 1916
- Family: Scutacaridae Oudemans, 1916
- Genus: Archidispus Karafiat, 1959 NP
- Archidispus sp.. Locality: 1S: 1 ♀
- Archidispus sp.. Locality: 1S: 1 ♀
- Genus: Diversipes Berlese, 1903
- Diversipes sp.. Locality: 1S: 1 ♀
- Diversipes sp.. Locality: 1S: 1 ♀
- Genus: Imparipes Berlese, 1903
- Imparipes sp. 1. Locality: 2W: 1 ♀
- Imparipes sp. 2. Locality: 10S: 3 ♀♀
- Imparipes sp. 1. Locality: 2W: 1 ♀
- Genus: Pygmodispus Paoli, 1911 NP
- Pygmodispus sp.. Locality: 2S: 1 ♀
- Pygmodispus sp.. Locality: 2S: 1 ♀
- Genus: Scutacarus Gross, 1845
- Scutacarus plumosus (Paoli, 1911) NP. Locality: 7S: 1 ♀; 10S: 3 ♀♀
- Scutacarus sp. 1 . Locality: 1S: 1 ♀; 2W: 1 ♀
- Scutacarus sp. 2. Locality: 8S: 4 ♀♀
- Scutacarus plumosus (Paoli, 1911) NP. Locality: 7S: 1 ♀; 10S: 3 ♀♀
- Genus: Archidispus Karafiat, 1959 NP
- Superfamily: Pygmephoroidea Cross, 1965
- Family: Microdispidae Cross, 1965
- Genus: Microdispus Paoli, 1911
- Microdispus sp.. Locality: 5S: 1 ♀
- Microdispus sp.. Locality: 5S: 1 ♀
- Genus: Microdispus Paoli, 1911
- Family: Pygmephoridae Cross, 1965
- Genus: Bakerdania Sasa, 1961
- Bakerdania sp.. Locality: 10S: 1 ♀
- Bakerdania sp.. Locality: 10S: 1 ♀
- Genus: Pediculaster Vitzthum, 1929
- Pediculaster sp. 1 . Localities: 6W: 6 ♀♀; 7W: 4 ♀♀
- Pediculaster sp. 2 . Localities: 5W: 1 ♀; 8W: 4 ♀♀
- Pediculaster sp. 1 . Localities: 6W: 6 ♀♀; 7W: 4 ♀♀
- Genus: Siteroptes Amerling, 1861
- Siteroptes avenae (Mueller, 1905). Localities: 2S: 2 ♀♀; 5W: 1 ♀, 5S: 2 ♀♀; 10W: 1 ♀
- Siteroptes avenae (Mueller, 1905). Localities: 2S: 2 ♀♀; 5W: 1 ♀, 5S: 2 ♀♀; 10W: 1 ♀
- Genus: Bakerdania Sasa, 1961
- Superfamily: Tarsonemoidea Canestrini et Fanzago, 1877
- Family: Tarsonemidae Canestrini et Fanzago, 1877
- Genus: Neotarsonemoides Kaliszewski, 1984
- Neotarsonemoides lucifer Schaarschmidt, 1959. Localities: 2S: 1 ♀; 7S: 10 ♀♀
- Neotarsonemoides sp. 1. Locality: 2W: 3 ♀♀
- Neotarsonemoides sp. 2. Locality: 2W: 1 ♀
- Neotarsonemoides lucifer Schaarschmidt, 1959. Localities: 2S: 1 ♀; 7S: 10 ♀♀
- Genus: Steneotarsonemus Beer, 1954
- Steneotarsonemus sp.. Locality: 1W: 1 ♀
- Steneotarsonemus sp.. Locality: 1W: 1 ♀
- Genus: Tarsonemus Canestrini et Fanzago, 1876
- Subgenus: Tarsonemus Canestrini et Fanzago, 1876
- Tarsonemus annotatus Livshitz, Mitrofanov et Sharonov, 1979 NP. Locality: 8S: 1 ♀
- Tarsonemus bachmaieri Cooreman, 1958 NP. Localities: 5W: 1 ♀, 5S: 10 ♀♀
- Tarsonemus bognari Nemestothy et Mahunka, 1981 NP . Locality: 6S: 1 ♀
- Tarsonemus cf. confusus Ewing, 1922 . Locality: 8W: 10 ♀♀
- Tarsonemus fusarii Cooreman, 1941. Localities: 7W: 7 ♀♀; 10S: 1 ♀
- Tarsonemus hermes Suski, 1966. Locality: 3W: 3 ♀♀
- Tarsonemus lenticulatus Gheblealivand, Haddad et Magowski, 2018 NP NE. Localities: 1W: 1 ♀; 5W: 2 ♀♀, 5S: 1 ♀; 9W: 2 ♀♀
- Tarsonemus limitatus Kaliszewski, 1993. Localities: 7W: 1 ♀, S: 1 ♀; 8W: 3 ♀♀, 8S: 1 ♀; 10S: 1 ♀
- Tarsonemus mixtus Kaliszewski, 1993. Localities: 1W: 9 ♀♀, 1S: 1 ♀; 2S: 1 ♀; 3W: 2 ♀♀
- Tarsonemus monoporus Weis-Fogh, 1947 NP. Localities: 3S: 1 ♀; 5W: 4 ♀♀, 5S: 4 ♀♀
- Tarsonemus myceliophagus Hussey, 1963. Locality: 8S: 1 ♀
- Tarsonemus peltatus Kaliszewski, 1993. Localities: 1S: 8 ♀♀; 2S: 8 ♀♀; 3W: 6 ♀♀
- Tarsonemus preaterius Kaliszewski, 1993. Localities: 7S: 1 ♀; 8W: 11 ♀♀, S: 4 ♀♀; 9W: 13 ♀♀, 9S: 10 ♀♀
- Tarsonemus regularis Kaliszewski, 1993. Locality: 5S: 1 ♀
- Tarsonemus schaarschmidti Mahunka, 1969. Locality: 9W: 2 ♀♀
- Tarsonemus violae Schaarschmidt, 1960. Locality: 2W: 1 ♀
- Tarsonemus waitei Banks, 1912. Localities: 4S: 4 ♀♀; 5S: 2 ♀♀; 6S: 1 ♀; 7S: 3 ♀♀; 9W: 1 ♀
- Tarsonemus sp. 1 . Locality: 8W: 4 ♀♀
- Tarsonemus sp. 2 . Locality: 9W: 1 ♂
- Tarsonemus annotatus Livshitz, Mitrofanov et Sharonov, 1979 NP. Locality: 8S: 1 ♀
- Genus: Neotarsonemoides Kaliszewski, 1984
- Order: Sarcoptiformes
- Suborder: Endeostigmata
- Cohort: Alycina
- Superfamily: Alycoidea Canestrini et Fanzago, 1877
- Family: Nanorchestidae Grandjean, 1937
- Genus: Nanorchestes Topsent et Trouessart, 1890
- Nanorchestes cf. llanoi Strandtmann, 1982 . Localities: 1W: 3 ♀♀ 1 ♂; 10W: 1 ♀
- Nanorchestes cf. antarcticus Strandtmann, 1963 . Localities: 2W: 1 ♀ 1 ♂; 3W: 1♀, 3S: 1 ♀ 1 ♂ 1 tn; 4W: 4 ♀♀ 2 n, 4S: 1 ♀; 6W: 2 ♀♀ 3 ♂♂; 7W: 3 n; 10W: 2 ♀♀ 2 ♂♂, 10S: 1 ♂
- Nanorchestes cf. llanoi Strandtmann, 1982 . Localities: 1W: 3 ♀♀ 1 ♂; 10W: 1 ♀
- Genus: Speleorchestes Trägårdh, 1909
- Speleorchestes cf. pratensis Willmann, 1936. Localities: 4W: 5 ♀♀ 4S: 4 ♀♀; 5S: 1 ♀
- Speleorchestes cf. pratensis Willmann, 1936. Localities: 4W: 5 ♀♀ 4S: 4 ♀♀; 5S: 1 ♀
- Genus: Nanorchestes Topsent et Trouessart, 1890
- Cohort: Terpnacarina
- Superfamily: Terpnacaroidea Grandjean, 1939
- Family: Terpnacaridae Grandjean, 1939
- Genus: Terpnacarus Grandjean, 1939
- Terpnacarus subterraneus Weis-Fogh, 1948 NP. Locality: 6W: 1 ♀
- Terpnacarus subterraneus Weis-Fogh, 1948 NP. Locality: 6W: 1 ♀
- Genus: Terpnacarus Grandjean, 1939
Discussion
With reference to previous data summarized in Bogdanowicz et al. (2008), Kaźmierski et al. (2018), and Mąkol and Wohltmann (2012), one family, 11 genera and 27 species recorded during present study are new to the fauna of Poland and two species are also new to the fauna of Europe (André 2004; Baker 2004; Silva et al. 2016; Magowski 2004; Uusitalo, and Judson 2004).
Majority of recorded species were found in one habitat only, whereas the most frequently occurring species were Cunaxa setirostris, Eupodes voxencollinus, Microtydeus beltrani and Nanorchestes cf. antarcticus, each found in more than five habitats. The latter may indicate that these four species display relatively low habitat selectivity.
A comparison of our results with those of other similar studies is difficult due to the scarcity of surveys on Prostigmata soil faunas carried out in limited spatial areas. Such studies usually deal with highly distinct habitats (e.g. subterranean in Skubała et al. 2013, marches in Dogan et al. 2015) or refer to narrower taxonomic scope. One of the few similar projects to ours, though covering a larger area, was carried out by Niedbała et al. (1982). During our investigation, 139 species were found with 868 examined individuals, whereas Niedbała et al. (1982) recorded 321 species represented by 8169 individuals. The latter work involved also Oribatida (Acariformes) and Gamasida (Parasitiformes), which were not included in our research. This means that in our study the no. of species per no. of specimens is four times higher than in Niedbała et al. (1982), despite involving narrower spectrum of systematic groups. The disproportion can be tentatively explained by a wider variety of habitats examined in our survey. Moreover, in our study only 20 samples were collected, while as many as 400 were examined by Niedbała et al. (1982). Thus, the species no./sample index amounts to 7.0 in our study, while it is 0.8 in Niedbała et al. (1982). Unfortunately, no exact number of samples per each particular mite group is given in the latter paper, rendering such comparisons hardly informative. When compiling the comparable aspects of these two datasets, the cohort Heterostigmatina is represented by 71 species in Niedbała et al. (1982) and by 36 in our study. The remaining Prostigmata, however, are represented by 50 species in Niedbała et al. (1982) and 99 in this study, testifying to nearly a twice higher diagnostic resolution in our approach, despite ca. 20 times lower number of samples examined. The number of species of the suborder Endeostigmata recorded in both studies was comparable and rather low.
No doubts, the regular sampling, even limited in space contributes to species richness. Łaydanowicz and Mąkol (2010) recorded as many as 13 species, representing four families and constituting almost nine percent of the Polish fauna of terrestrial Parasitengona mites, within a studied area of ca. 200 m2, located in the vicinity of the university campus in Wrocław.
Some semi-aquatic or aquatic taxa i.e., representatives of the Hydrachnidia (water mites), as well as plant-associated mites, i.e., Tetranychidae (spider mites) were recorded in our material. Presence of the former can be explained by the proximity of the pond to the locality no. 2, whereas the latter finding is regarded accidental. Nevertheless, both should be considered as accidental in the typically soil habitats.
As mentioned before, our aim was not only to provide a local checklist of Prostigmata and Endeostigmata, but also to make the data on the presence of those mites in AMU Campus accessible to other biologists, who need material for their laboratory studies. This objective has been already met, since some of our early results were recently used for a molecular phylogenetic assay on Eupodoidea by Szudarek-Trepto et al. (2020). Further studies based on our findings are underway.
Systematic evaluation and comments on the mite material presented herewith will be a subject of a separate study.
Acknowledgements
We would like to thank the staff members of the Department of Plant Ecology and Conservation Biology, Adam Mickiewicz University in Poznań for identification of plant communities in sampling areas. We are grateful to the reviewers for their valuable comments.
References
- André H.M. 2004. Fauna Europaea Database. Ereynetidae, Iolinidae, Meyerellidae, Tydeidae (Acari: Prostigmata). [Internet]. [14 April 2021]. Available from: http://www.faunaeur.org

- Baker A.S. 2004. Fauna Europaea Database. Eupodidae, Penthaleidae, Penthalodidae, Rhagidiidae (Acari: Prostigmata). [Internet]. [14 April 2021]. Available from: url{http://www.faunaeur.org}
- Belliure B., Montserrat M., Magalhães, S. 2010. Mites as models for experimental evolution studies. Acarologia, 50(4): 513-529. https://doi.org/10.1051/acarologia/20101985
- Bizarro G.L., Wurlitzer W.B., Britto E.P.J., Johann L., Ferla N.J., de Moraes G.J., da Silva G.L. 2020. Two new species (Acari: Tydeidae: Eupodidae) from Mato Grosso, Brazil. Int. J. Acarology, 46(7): 538-543. https://doi.org/10.1080/01647954.2020.1825527
- Bogdanowicz W., Chudzicka E., Pilipiuk I. 2008. Fauna Polski. Charakterystyka i wykaz gatunków. Tom III. Muzeum i Instytut Zoologii PAN, Warszawa, 603 pp. (In Polish).
- Booth R.G., Edwards M., Usher M.B. 1985. Mites of the genus Eupodes (Acari, Prostigmata) from maritime Antarctica: a biometrical and taxonomic study. J. Zool., London, (A), 207: 381-406. https://doi.org/10.1111/j.1469-7998.1985.tb04939.x
- Doğan S., Sevsay S., Ayyildiz N., Özbek H.H., Dılkaraoğlu S., Erman O., Aksoy H. 2015. The mite fauna of Ekşisu Marshes in Erzincan (Turkey). Turk. J. Zool., 39: 571-579. https://doi.org/10.3906/zoo-1407-3
- Fan Q.-H. 2004. A catalogue of the genus Eupalopsellus Sellnick (Acari: Prostigmata, Eupalopsellidae) with the description of a new species from China. Biologia, Bratislava, 59(5): 533-545. Available from: http://biologia.savba.sk/59_5_04/Fan_QH.pdf

- Gabryś G. 2016. A key to postlarval Erythraeidae (Acari, Actinotrichida) of Poland. Rocznik Muzeum Górnośląskiego w Bytomiu, Przyroda, 22: 1-22. Available from: http://muzeum.bytom.pl/wp-content/uploads/2016/12/Przyroda_22online007.pdf

- Gilyarov M.S., Vainshtein B.A., Volgin V.I., Krivolutski D.A., Kuznetsov N.N., Livshits I.Z., Mitrofanov V.I., Sevastyanov V.D., Sosnina E.F. 1978. Opredelitel' obitayushchikh v pochve kleshchei. Trombidiformes. Nauka, Moscow, 271 pp.
- Haitlinger R. 1986. The genus Hauptmannia Oudemans 1910 (Acari, Prostigmata, Erythraeidae) in Poland. Polskie Pismo entomologiczne, 56: 181-191.
- Haitlinger R. 2002. A new larval Hauptmannia Oudemans 1910 and the first record of Abrolophus neobrevicollis Zhang, Goldarazena, 1996 (Acari: Prostigmata: Erythraeidae) from Madeira. Syst. Parasitol., 53: 115-119. https://doi.org/10.1023/A:1020414023652
- Jesionowska K. 2010. Cocceupodidae, a new family of eupodoid mites, with description of a new genus and two new species from Poland Part I. (Acari: Prostigmata: Eupodoidea). Genus (Wrocław), 21(4): 637-658. http://www.biol.uni.wroc.pl/cassidae/Jesionowska_Cocceupodidae.pdf

- Kaliszewski M. 1987. Siteroptes longisomus sp. n. from Poland, with remarks on the genus and key to species (Acari, Pygmephoroidea). Mitt. Zool. Mus. Hamburg, 9(130): 21-36. Available from: https://www.zobodat.at/pdf/Ent-Mitt-Zool-Mus-Hamburg_9_0021-0036.pdf

- Kaliszewski M. 1993. Key to Palearctic of the genus Tarsonemus. Wydawnictwo Naukowe UAM, Poznań. Seria: Zoologia, 14, pp. 204.
- Kaźmierski A. 1998. Tydeinae of the World: generic relationships, new and redescribed taxa and keys to all species. A revision of the subfamilies Pretydeinae and Tydeinae (Acari: Actinedida: Tydeidae). IV. Acta Zool. Cracov., 41(2): 283-441.
- Kaźmierski A., Marciniak M., Sikora B. 2018. Tydeinae mites (Acariformes: Prostigmata: Tydeidae) from bird nests with description of three new species. Syst. Appl. Acarol., 23: 803-823. https://doi.org/10.11158/saa.23.5.3
- Khaustov A.A. 2014. A new genus and species in the mite family Eupodidae (Acari, Eupodoidea) from Crimea. ZooKeys, 422: 11-22. https://doi.org/10.3897/zookeys.422.7802
- Krantz G.W., Walter D.E. 2009. A Manual of Acarology. Texas Tech University Press. Lubbock, Texas. pp. 807.
- Łaydanowicz J., Mąkol J. 2010. Correlation of heteromorphic life instars in terrestrial Parasitengona mites and its impact on taxonomy - the case of Leptus molochinus (C. L. Koch, 1837) and L. ignotus (Oudemans, 1903) (Acari: Actinotrichida: Prostigmata: Erythraeidae). J. Nat. Hist., 44(11): 669-697. https://doi.org/10.1080/00222930903383560
- Magowski W.Ł. 1997. Rewizja rodzaju Tarsonemus Canestrini i Fanzago, 1876 (Acari: Heterostigmata: Tarsonemidae) Europy. Rozprawa doktorska, Uniwersytet im. A. Mickiewicza (Revision of the genus Tarsonemus Canestrini & Fanzago, 1876 (Acari: Heterostigmata: Tarsonemidae) in Europe. Ph.D. dissertation, A. Mickiewicz University) Poznań, 281 s. + 204 ryc. (In Polish).
- Magowski W.Ł. 2004. Fauna Europaea Database. Tarsocheyloidea, Pyemotoidea, Trochometridioidea, Tarsonemoidea (Acari: Heterostigmata). [Internet]. [14 April 2021]. Available from: http://www.faunaeur.org

- Mąkol J. 2010. A redescription of Balaustium murorum (Hermann, 1804) (Acari: Prostigmata: Erythraeidae) with notes on related taxa. Ann. Zool., 60(3): 439-454. https://doi.org/10.3161/000345410X535424
- Mąkol J., Wohltmann A. 2012. An annotated checklist of terrestrial Parasitengona (Actinotrichida: Prostigmata) of the world, excluding Trombiculidae and Walchiidae. Ann. Zool., 62: 359-562. https://doi.org/10.3161/000345412X656671
- Michocka S. 1987. Polskie roztocze (Acari) z rodziny Bdellidae i Cunaxidae. PWN, Warszawa, Kraków, 14: 1-130. (In Polish).
- Niedbała W., Błaszak C., Błoszyk J., Kaliszewski M., Kaźmierski A. 1982. Soils Mites (Acari) of Warsaw and Mazovia. Memorabilia Zool., 36: 235-252.
- Rack G. 1981. Auftreten von Terpnacarus subterraneus Weis-Fogh, 1947 (Acarina, Actinedida, Terpnacaridae) in einem neuen Wohnhaus in Süddeutschland. Ent. Mitt. Zool. Mus. Hamburg, 7(111): 3-9. Available from: https://www.zobodat.at/pdf/Ent-Mitt-Zool-Mus-Hamburg_7_0003-0009.pdf

- Silva G.L., Maicon H.M., da Silva O.S., Ferla N.J. 2016. Catalogue of the mite family Tydeidae (Acari: Prostigmata) with the world key to the species. Zootaxa, 4135(1): 1-68. https://doi.org/10.11646/zootaxa.4135.1.1
- Skubała P., Dethier M., Madej G., Solarz K., Mąkol J., Kaźmierski A. 2013. How many mite species dwell in subterranean habitats? A survey of Acari in Belgium. Zool. Anz., 252: 307-318. https://doi.org/10.1016/j.jcz.2012.09.001
- Smiley R.L. 1992. The predatory mite family Cunaxidae (Acari) of the world with a new classification. Indira Publ. House, West Bloomington, Michigan, 356 pp. https://www.cabdirect.org/cabdirect/abstract/19941107190

- Stålstedt J., Wohltmann A., Bergsten J., Mąkol J. 2016. Towards resolving the double classification in Erythraeus (Actinotrichida: Erythraeidae) matching larvae with adults using 28S sequence data and experimental rearing. Org. Divers. Evol., 16: 761-790. https://doi.org/10.1007/s13127-016-0283-5
- Strandtmann R.W. 1971. The Eupodoid Mites of Alaska. Pac. Insects, 13(1): 75-118. Available from: http://web.corral.tacc.utexas.edu/UAF/arctos/mediaUploads/ffdss/Strandtmann_1971_THE_EUPODOID_MITES_OF_ALASKA_13_1_75.pdf

- Szudarek-Trepto N., Kaźmierski A., Dabert M., Dabert J. 2020. Molecular phylogeny of Eupodidae reveals that the family Cocceupodidae (Actinotrichida; Eupodoidea) and its genus Filieupodes are valid taxa. Exp. Appl. Acarol., 80(4): 43-57. https://doi.org/10.1007/s10493-019-00455-4
- Thor S., Willmann C. 1941. Eupodidae, Penthalodidae, Penthaleidae, Rhagidiidae, Pachygnathidae, Cunaxidae. Das Tierreich, 71a: 1-186. https://doi.org/10.1515/9783111434568-004
- Uusitalo M. 2010. Terrestrial species of the genus Nanorchestes (Endeostigmata: Nanorchestidae) in Europe. In: M.W. Sabelis and J. Bruin (Eds). Trends in Acarology: Proceedings of the 12th International Congress: 161-166; Springer Science+Business Media B.V. https://doi.org/10.1007/978-90-481-9837-5_25
- Uusitalo M., Judson M. 2004. Fauna Europaea Database. Alicorhagiidae, Alycidae, Lordalychidae, Micropsammidae, Nanorchestidae, Nematalycidae, Oehserchestidae, Sphaerolichidae, Terpnacaridae (Acari: Endeostigmata). [Internet]. [14 April 2021]. Available from: http://www.faunaeur.org

- Walter D.E., Proctor H.C. 2010. Mites as Modern Models: Acarology in the 21st Century. Acarologia, 50(1): 131-141. https://doi.org/10.1051/acarologia/20101955
- Wohltmann A., Gabryś G., Mąkol J. 2007. Acari: Terrestrial Parasitengona inhabiting transient biotopes. In: R. Gerecke (Ed.). Süßwasserfauna von Mitteleur. 7/2-1, Chelicerata, Araneae, Acari I. Spektrum Elsevier, München: 158-240. https://doi.org/10.1007/978-3-662-55958-1_6
- Zacharda M. 1980: Soil mites of the family Rhagidiidae (Actinedida: Eupodoidea). Morphology, systematics, ecology. Acta Univ. Carol. Biol., (1978), 5-6: 489-785.



2021-06-05
Date accepted:
2021-09-01
Date published:
2021-09-23
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Laniecki, Ronald; Kaźmierski, Andrzej; Mąkol, Joanna; Laniecka, Izabella and Magowski, Wojciech
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



