New faunistical and taxonomic data on oribatid mites (Acari: Oribatida) of Ethiopia, with description of two new species of the superfamily Oripodoidea
Ermilov, Sergey G.1 ; Subias, Luis S.2 ; Shtanchaeva, Umukusum Ya.3 and Friedrich, Stefan4
1✉ Institute of Environmental and Agricultural Biology (X-BIO), Tyumen State University, Tyumen, Russia.
2Complutense University, Madrid, Spain.
3Complutense University, Madrid, Spain.
4SNSB-Bavarian State Collection of Zoology, Munich, Germany.
2021 - Volume: 61 Issue: 3 pages: 591-601
https://doi.org/10.24349/ynmN-vroHZooBank LSID: 4275811E-0F44-4DD8-8707-051B9A62A9DD
Original research
Keywords
Abstract
Introduction
This work is based on oribatid mite material (Acari, Oribatida) which was collected during a field trip in southwestern Ethiopia in 2019. The primary goal of our paper is to present a list of the identified taxa with notes on new findings (new records).
The secondary goal of our paper is to describe and illustrate two new species, one belonging to the genus Pilobates Balogh, 1960 (family Haplozetidae), the other to Zetorchella Berlese, 1916 (family Caloppiidae).
Pilobates was proposed by Balogh (1960) with Protoribates pilosellus Balogh, 1958 as type species. The genus comprises seven species which are distributed in the Afrotropical region (mostly Madagascar) and in the Mediterranean (Subías 2021). The revised generic diagnosis was presented by Ermilov (2020). An identification key to all species of Pilobates was provided earlier by Ermilov and Starý (2020). Zetorchella was proposed by Berlese (1916a) with Chaunoproctus cancellatus Pearce, 1906 as type species. The genus comprises 24 species which are distributed in the Afrotropical and Oriental regions, and Guyana (Subías 2021). The revised generic diagnosis and an identification key to species of Zetorchella of the Afrotropical region were presented by Ermilov and N'Dri (2019).
Until now, three Zetorchella species have been registered from Ethiopia (e.g. Ermilov et al. 2010; Ermilov 2016): Z. nortoni Ermilov, Sidorchuk and Rybalov, 2010; Z. pedestris Berlese, 1916; Z. vargai (Balogh, 1959). Representatives of Pilobates were not discovered earlier in the Ethiopian fauna.
Material and methods
Specimens — Substrate samples containing oribatid mites were collected by hand method from (Fig. 1) SW Ethiopia, 04°49'42''N, 36°43'22''E, Southern Nations, Nationalities and People's region, Debub Omo Zone, Woreda Hamer Bena, 50 km SE of Turmi, riverbed W Chew Bahir (Stephanie Wildlife Sanctuary), 700 m a.s.l., upper soil and leaf litter in forest, 02.IV.2019 (S. Friedrich and F. Wachtel). Mites were extracted from samples into 75% ethanol using Winkler's apparatus in laboratory conditions during 10 days.



As detailed below, types are deposited in three institutions: the Bavarian State Collection of Zoology, Munich, Germany (ZSM); the Tyumen State University Museum of Zoology, Tyumen, Russia (TSUMZ); and the Senckenberg Museum of Natural History, Görlitz, Germany (SMNH).
Observation and documentation — Specimens were mounted in lactic acid on temporary cavity slides for measurement and illustration. Body length was measured in lateral view, from the tip of the rostrum to the posterior edge of the notogaster. Notogastral width refers to the maximum width of the notogaster in dorsal view. Lengths of body setae were measured in lateral aspect. All body measurements are presented in micrometers. Formulas for leg setation are given in parentheses according to the sequence trochanter-femur-genu-tibia-tarsus (famulus included). Formulas for leg solenidia are given in square brackets according to the sequence genu-tibia-tarsus.
Drawings were made with a camera lucida using a Leica transmission light microscope ''Leica DM 2500''.
References for original descriptions of species in ''List of identified oribatid mite taxa'' are not presented in the References section.
Terminology — Morphological terminology used in this paper follows that of F. Grandjean: see Travé and Vachon (1975) for references, Norton (1977) for leg setal nomenclature, and Norton and Behan–Pelletier (2009), for overview.
Abbreviations — Prodorsum: lam = lamella; tlam = translamella; slam = sublamella; Al = sublamellar porose area; ro, le, in, bs, ex = rostral, lamellar, interlamellar, bothridial, and exobothridial seta, respectively; tu = tutorium; Ad = dorsosejugal porose area; D = dorsophragma; P = pleurophragma. Notogaster: Aa, A1, A2, A3, A4 = porose areas; Sa, S1, S2, S3 = sacculi; c, d, da, dm, dp, la, lm, lp, h, p = setae; ia, im, ip, ih, ips = lyrifissures; gla = opisthonotal gland opening. Gnathosoma: a, m, h = subcapitular setae; or = adoral seta; d, l, sup, inf, cm, ul, su, vt, lt = palp setae; ω = palp solenidion; cha, chb = cheliceral setae; Tg = Trägårdh's organ. Epimeral and lateral podosomal regions: 1a–c, 2a, 3a–c, 4a–c = epimeral setae; PdI, PdII = pedotectum I, II, respectively; dis = discidium; cpc = circumpedal carina. Anogenital region: g, ag, an, ad = genital, aggenital, anal, and adanal setae, respectively; iad = adanal lyrifissure; Amar = marginal porose area; po = preanal organ. Legs: Tr, Fe, Ge, Ti, Ta = leg trochanter, femur, genu, tibia, tarsus, respectively; pa = porose area; ω, φ, σ = leg solenidia; ɛ = leg famulus; d, l, v, bv, ev, ft, tc, it, p, u, a, s, pv, pl = leg setae.
List of identified oribatid mite taxa
Licnodamaeidae
Pedrocortesella parva Pletzen, 1963: 5 ex. Distribution: South Africa. New record of the species in Ethiopia.
Granuloppiidae
Hammerella sp.: 1 ex. This presumably is a new species; however, we do not describe it here due to the presence of only one specimen. New record of the family and genus in Ethiopia.
Scutoverticidae
Ethiovertex sculperens (Kok, 1968): 1 ex. Distribution: Ethiopian region. New record of the species in Ethiopia.
Scutovertex fossatus Wallwork, 1967: 2 ex. Distribution: Chad. New record of the species in Ethiopia.
Passalozetidae
Bipassalozetes deserticus Bayartogtokh and Aoki, 1997: 3 ex. Distribution: Mongolia, Yemen. New record of the family, genus and species in Ethiopia.
Bipassalozetes sp.: 1 ex. This presumably is a new species; however, we do not describe it here due to the presence of only one specimen.
Tegoribatidae
Hypozetes cf. andreii Ermilov, Hugo-Coetzee, Khaustov and Kontschán, 2019: 12 ex. Distribution: South Africa. New record of the family, genus and species in Ethiopia.
Remarks. The Ethiopian specimens completely correspond morphologically to specimens from South Africa (Ermilov et al. 2019) except for the slightly wavy end of the interlamellar seta (versus straight). We believe this is intraspecific variability.
Caloppiidae
Zetorchella robertbecki n. sp.: 28 ex.
Oribatulidae
Oribatula incompleta Mahunka, 1987: 4 ex. Distribution: Nigeria. New record of the species in Ethiopia.
Scheloribatidae
Scheloribates pallidulus (Koch 1841): 2 ex. Distribution: Cosmopolitan.
Haplozetidae
Pilobates wachteli n. sp.: 2 ex. New record of the genus in Ethiopia.
\subsubsection*
Hence, we found 11 species from 10 genera and nine families. Of these: four species are new to science; six species, four genera and two families are recorded in the fauna of Ethiopia for the first time.
Taxonomy
Pilobates wachteli n. sp.
ZOOBANK: F8DAB902-DC32-4263-A4AD-67174C022C3C ![]()
(Figures 2, 3)
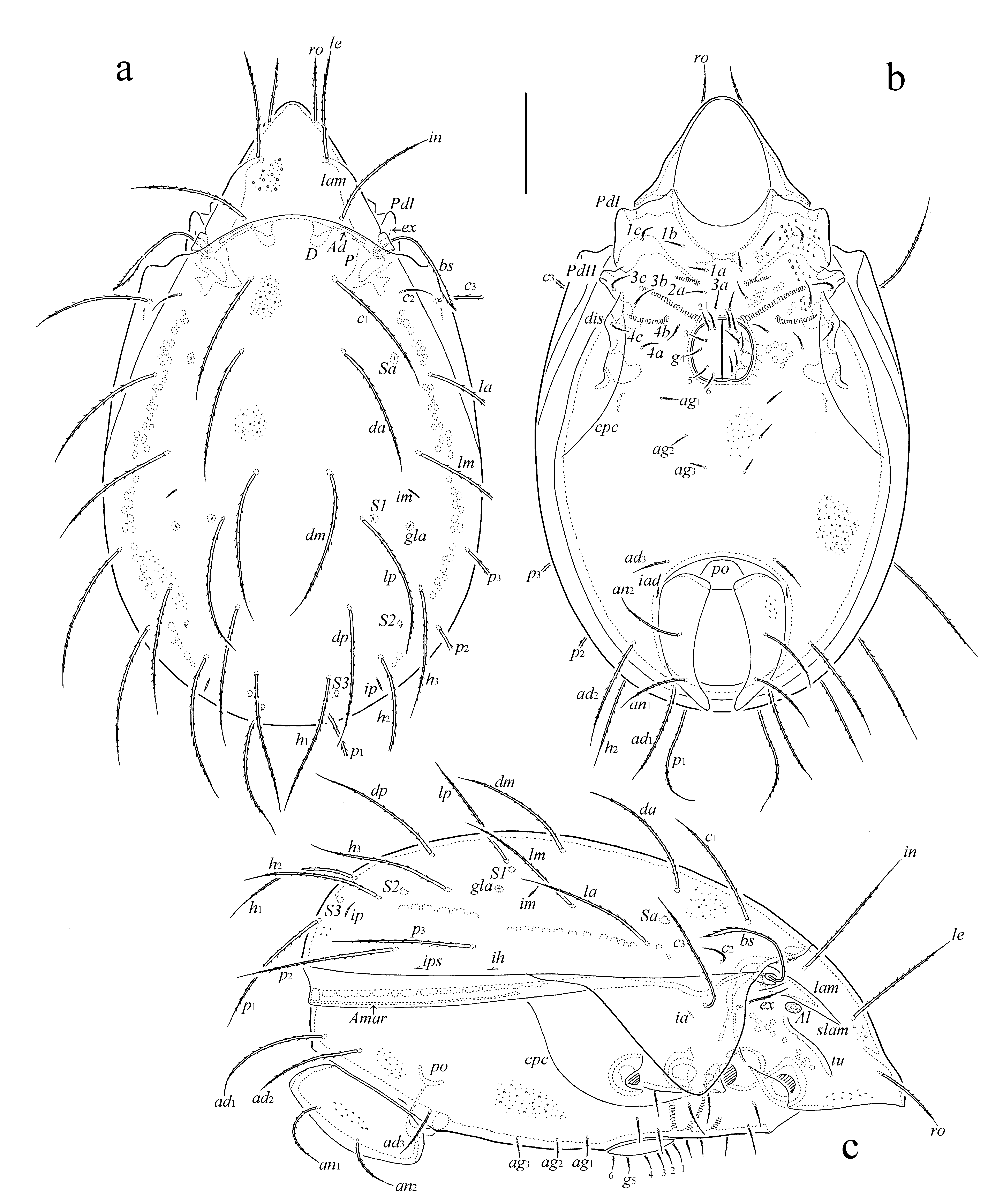





Diagnosis — Adult. Body size: 630–713 × 365–431. Body surface densely microgranulate and sparsely foveolate. Rostral, lamellar and interlamellar setae setiform, barbed; ro shortest, in longest. Bothridial seta long, lanceolate, barbed. Tutorium of medium length. With 15 pairs of setiform, barbed notogastral setae, c2 comparatively short, others long. Epimeral and anogenital setae setiform, barbed; anal and adanal (ad1, ad2) setae long, ad3 of medium length. With six pairs of genital and three pairs of aggenital setae. All leg tarsi with three claws. Trochanter IV without tooth distodorsally.
Description of adult — Measurements – Body length: 630 (holotype: male), 713 (one paratype: female); body width: 365 (holotype), 431 (paratype).
Integument (Figs 2a-c, 3a) – Body color light brown. Body surface densely microgranulate and (except median part of epimeral region) sparsely foveolate (diameter of foveola up to 4).
Prodorsum (Figs 2a, 2c) – Rostrum rounded. Lamella about 1/2 length of prodorsum. Prolamella absent. Sublamella slightly shorter than lamella. Sublamellar porose area oval (20–24 × 12–16), located ventral to sublamella. Tutorium similar to lamella in length, ridge-like, distinctly not reaching rostral margin. Rostral (73–77), lamellar (131–143), interlamellar (151–164), and exobothridial (53–57) setae setiform, barbed; ex thinnest. Bothridial seta (123–135) lanceolate, barbed, with long stalk and short head. Dorsosejugal porose area band-like, poorly visible.
Notogaster (Figs 2a, 2c) – Anterior notogastral margin slightly convex medially. Pteromorph subtriangular, rounded laterally, with well-developed hinge. Fifteen pairs of notogastral setae (c2: 36–41; others: 135–147) setiform, barbed; c2 thinnest. Four pairs of sacculi drop-like. Opisthonotal gland opening and all lyrifissures distinct.
Gnathosoma (Figs 3a-d) – Subcapitulum size: 147–151 × 98–106. Subcapitular setae setiform, a and h (24–28) barbed, m (20–24) slightly barbed, thinnest. Adoral seta (16–20) setiform, barbed. Palp (61–65) with setation 0–2–1–3–9(+ω). Postpalpal seta (8) spiniform, roughened. Chelicera (159–164) with two setiform, barbed setae (cha: 57–61; chb: 36–41).
Epimeral and lateral podosomal regions (Figs 2b, 2c) – Epimeral setal formula: 3–1–3–3. Setae (1a, 1c, 2a, 3a: 20–24; 3c: 49–53; others: 36–41) setiform, barbed. Circumpedal carina long, directed to pedotectum II. Discidium subtriangular.
Anogenital region (Figs 2b, 2c) – Six pairs of genital (20–24), three pairs of aggenital (20–24), two pairs of anal (73–82), and three pairs of adanal (ad1, ad2: 114–121; ad3: 45–53) setae setiform, barbed. Adanal lyrifissure located close and parallel to anal plate. Marginal porose area complete, band-like.
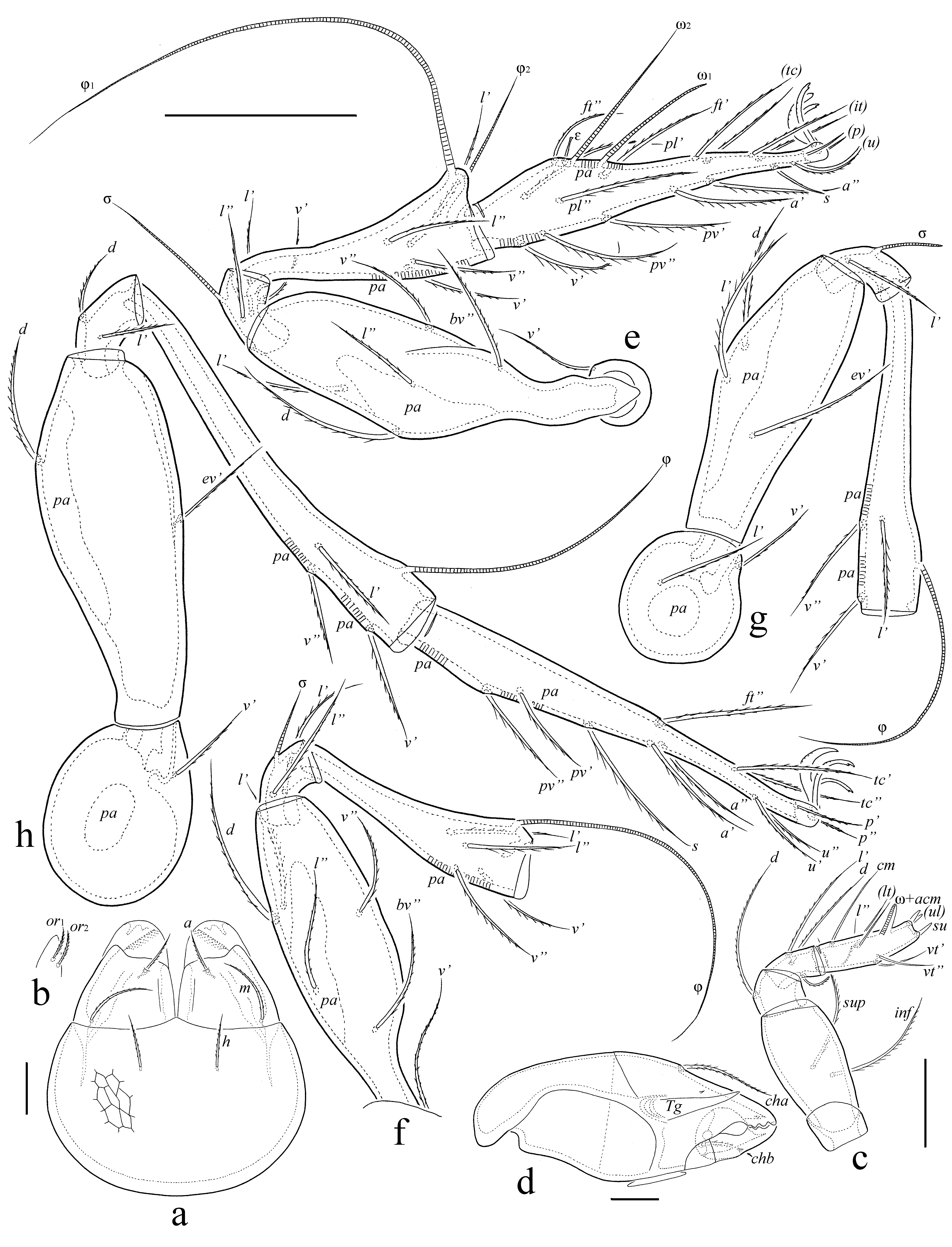
Legs (Figs 3e-h) – Tridactylous. Median claw distinctly thicker than lateral claws, all slightly barbed on dorsal side. Tibia I with small dorsodistal apophysis bearing solenidia φ1 and φ2. Tibiae I and II with triangular tubercle proximoventrally. All femora rounded distoventrally. Trochanter IV rounded distodorsally (without tooth). Dorsoparaxial porose area on femora I–IV and on trochanters III, IV well visible; proximoventral porose area on tarsi I–IV and distoventral porose area on tibiae I–IV not observed. Formulas of leg setation and solenidia: I (1–5–3–4–18) [1–2–2], II (1–5–3–4–15) [1–1–2], III (2–3–1–3–15) [1–1–0], IV (1–2–2–3–12) [0–1–0]; homology of setae and solenidia indicated in Table 1. Famulus on tarsus I short, erect, slightly swollen distally, inserted between solenidion ω2 and seta ft''. Seta s on tarsus I eupathidial, located before setae a' and a''. Seta l'' on genu I located on small apophysis. Solenidion ω1 on tarsi I, ω1 and ω2 on tarsus II and σ on genu III bacilliform, other solenidia setiform.
Type deposition — The holotype is deposited in the collection of the ZSM; one paratype is deposited in the collection of the TSUMZ.
Etymology — The specific name is dedicated to our friend and colleague, the coleopterologist and all-around biologist Franz Wachtel (Grünwald, Germany) for his expertise and assistance by collecting the Ethiopian mites.



Remarks — In having tridactylous legs, Pilobates wachteli n. sp. is morphologically most similar to Pilobates incisura (Berlese, 1916) from Somalia (see Berlese 1916b; Mahunka 1994), however, the new species differs from the latter by the presence of 15 pairs of notogastral setae including c3 (versus 14 pairs, c3 absent), very long notogastral (except comparatively short c2), anal and adanal (ad1, ad2) setae (versus all these setae comparatively short), lanceolate bothridial seta (versus setiform), and foveolate body integument (versus not foveolate).
Zetorchella robertbecki n. sp.
ZOOBANK: 30E6D357-50A5-4D87-8780-82FCE151785A ![]()
(Figures 4, 5)
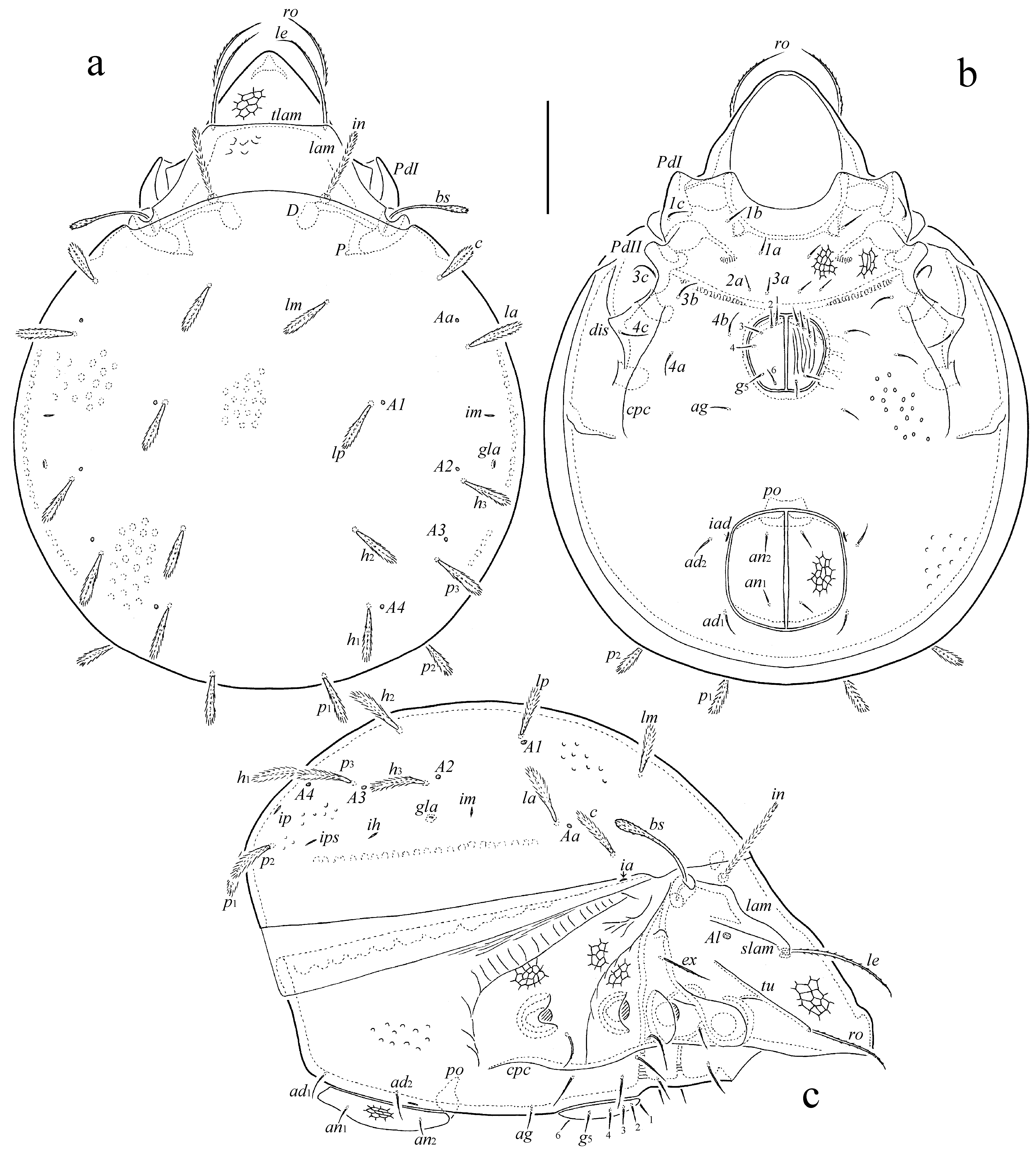




Diagnosis — Adult. Body size: 481–614 × 348–464. Genital plate striate; anal plate foveate-reticulate. Rostral and lamellar setae long, setiform, barbed; interlamellar seta long, thickened, heavily barbed; le longest. Bothridial seta long, clavate, barbed. With 10 pairs of comparatively short, phylliform, heavily barbed notogastral setae. Epimeral and anogenital setae setiform, barbed. With two pairs of adanal setae. Leg tarsus I with 19 setae (l'' absent).
Description of adult — Measurements – Body length: 498 (holotype: male), 481–614 (27 paratypes: 14 males and 13 females); body width: 381 (holotype), 348–464 (27 paratypes). No difference between males and females in body size.
Integument (Figs 4a-c, 5a) – Body color brown. Body surface densely microgranulate. Subcapitular mentum, epimeres I, II, anal plate, anterior part of prodorsum, and podosomal region between bothridium and acetabula III, IV foveate-reticulate. Interlamellar region slightly and partially foveate. Notogaster, anogenital region and epimeres III, IV sparsely foveolate (poorly visible). Genital plate longitudinally striate.
Prodorsum (Figs 4a, 4c) – Rostrum rounded. Lamella about 1/2 length of prodorsum. Translamella lineate. Sublamella about 1/2 length of lamella. Sublamellar porose area oval (6 × 4), located ventral to sublamella. Tutorium long, similar to lamella in length, ridge-like, distinctly not reaching rostral margin. Rostral (77–86), lamellar (98–106) and exobothridial (41–45) setae setiform, barbed; ex thinnest. Interlamellar seta (73–82) thickened, heavily barbed. Bothridial seta (69–77) clavate, barbed, with long stalk and short head. Dorsosejugal porose area not observed.
Notogaster (Figs 4a, 4c) – Anterior notogastral margin slightly convex medially. Humeral process small, tubercle-like. Ten pairs of notogastral setae (41–45) phylliform (dilated mediodistally), heavily barbed. Five pairs of porose areas rounded (4). Opisthonotal gland opening and all lyrifissures distinct.
Gnathosoma (Figs 5a-d) – Subcapitulum size: 114–123 × 98–102. Subcapitular setae (a and h: 24–28; m: 32–36) setiform, barbed. Adoral seta (12) setiform, barbed. Palp (77–82) with setation 0–2–1–3–9(+ω). Postpalpal seta (8) spiniform, roughened. Chelicera (123–131) with two setiform, barbed setae (cha: 41–45; chb: 24–28).
Epimeral and lateral podosomal regions (Figs 4b, 4c) – Epimeral setal formula: 3–1–3–3. Setae (1a, 1c, 2a, 3a: 16–20; 3c, 4c: 26–41; others: 24–32) setiform, barbed. Circumpedal carina long, strong. Discidium subtriangular.
Anogenital region (Figs 4b, 4c) – Six pairs of genital (16–20), one pair of aggenital (16–20), two pairs of anal (16–20), and two pairs of adanal (24–32) setae setiform, barbed. Adanal lyrifissure located close and parallel to anal plate. Marginal porose area absent.
Legs (Figs 5e-h) – Tridactylous. Median claw distinctly thicker than lateral claws, all slightly barbed on dorsal side; each lateral claw with small tooth ventrodistally. Tibia I with small dorsodistal apophysis bearing solenidia φ1 and φ2. Dorsal porose area on tarsi I, II, dorsoparaxial porose area on femora I–IV and on trochanters III, IV, proximoventral porose area on tarsi I–IV, and distoventral porose area on tibiae I–IV well observed; proximoventral and distoventral porose areas often divided into two parts. Formulas of leg setation and solenidia: I (1–5–3–4–19) [1–2–2], II (1–5–2–4–15) [1–1–2], III (2–3–1–3–15) [1–1–0], IV (1–2–2–3–12) [0–1–0]; homology of setae and solenidia indicated in Table 1. Famulus on tarsus I short, erect, slightly swollen distally, inserted between solenidion ω2 and seta ft''. Seta s on tarsus I eupathidial, located before setae a' and a''. Solenidion ω1 on tarsi I, ω1 and ω2 on tarsus II and σ on genu III bacilliform, other solenidia setiform.
Type deposition — The holotype is deposited in the collection of the ZSM; three paratypes are deposited in the collection of the SMNH; 24 paratypes are deposited in the collection of the TSUMZ.
Etymology — The specific name is dedicated to our friend and colleague, the coleopterologist Robert Beck (Munich, Germany) for his more than generous provision of all necessary facilities during this field trip.
Remarks – In having phylliform notogastral and thickened interlamellar setae, Zetorchella robertbecki n. sp. is morphologically most similar to Zetorchella rugosa (Mahunka, 1992) from Senegal (see Mahunka 1992), however, the new species differs from the latter by the presence of long bothridial seta (versus short), setiform lamellar seta (versus thickened), the same length of setae p1, p2 to other notogastral setae (versus distinctly shorter), striate genital plate (versus not striate), and foveate-reticulate anal plate (versus not foveate-reticulate).
From the Afrotropical species, Z. robertbecki n. sp. is morphologically similar also to Z. cotedivoirensis Ermilov, 2019 (see Ermilov and N'Dri 2019) and Z. pedestris Berlese, 1916 (see Berlese 1916a; Balogh 1960 – as Caloppia papillata Balogh, 1958; Mahunka 1991) in having phylliform dorsal notogastral setae, however, the new species differs from the former by the presence of 10 pairs of notogastral setae (p3 present) including well-developed phylliform p2 (versus nine pairs, p3 absent, p2 short, setiform), thickened interlamellar seta (versus phylliform), striate genital plate (versus not striate), and foveate-reticulate anal plate (versus not foveate-reticulate), and the absence of specific cerotegumental tuberculate pattern on the notogaster (versus present) and strong sinuous ridges forming partially unclear polygonal pattern on the ventral plate (versus present); from the latter by the presence of long bothridial seta (versus short), thickened interlamellar seta (versus phylliform), striate genital plate (versus not striate), and foveate-reticulate anal plate (versus not foveate-reticulate).
Acknowledgements
We cordially thank Robert Beck (Munich, Germany) and Franz Wachtel (Grünwald, Germany) for their invaluable help during this expedition; and Dr. Julia Baumann (Graz, Austria) and two anonymous reviewers for valuable comments. Furthermore S. Friedrich wants to thank the Hamer people from Turmi for their overwhelming hospitality during his stay.
References
- Balogh J. 1958. Oribatides nouvelles de l'Afrique tropicale. Rev. Zool. Bot. Afr., 58(1-2): 1-34.
- Balogh J. 1960. Descriptions complémentaires d'Oribates (Acari) d'Angola et du Congo Belge (Ière série). Comp. Diam. Angola, Lisboa, 51: 89-105.
- Berlese A. 1916a. Centuria prima di Acari nuovi. Redia, 12: 19-67.
- Berlese A. 1916b. Centuria terza di Acari nuovi. Redia, 12: 289-338.
- Ermilov S.G. 2016. Additions to the oribatid mite fauna (Acari, Oribatida) of Ethiopia: results of the Joint Russian-Ethiopian Biological Expedition (2014) to the vicinity of Lake Tana. Acarologia, 56(3): 367-378. https://doi.org/10.1051/acarologia/20162253
- Ermilov S.G. 2020. Two new species of Pilobates (Acari, Oribatida, Haplozetidae) from Madagascar. Syst. Appl. Acarol., 25(7): 1319-1328. https://doi.org/10.11158/saa.25.7.11
- Ermilov S.G., N'Dri, J.K. 2019. Contribution to the knowledge of the oribatid mite genus Zetorchella (Acari: Oribatida: Caloppiidae). Trop. Zool., 32(1): P. 1-9. https://doi.org/10.1080/03946975.2018.1538199
- Ermilov S.G., Starý J. 2020. A new species of the genus Pilobates (Acari, Oribatida, Haplozetidae) from Madagascar. Acarina, 28(2): 123-128. https://doi.org/10.21684/0132-8077-2020-28-2-123-128
- Ermilov S.G., Sidorchuk E.A., Rybalov L.B. 2010. Zetorchella nortoni, a new species of oribatid mite from Ethiopia (Acari: Oribatida: Caloppiidae). Acarina, 18(1): 61-65.
- Mahunka S. 1991. Notes, additions and redescriptions of the oribatid species of Berlese (Acari). Acta Zool. Hung., 37(1-2): 27-58.
- Mahunka S. 1992. New and interesting mites from the Geneva Museum LXIII. A survey of the oribatid fauna of Senegal (Acari: Oribatida). Rev. suisse Zool., 99(3): 673-712. https://doi.org/10.5962/bhl.part.79846
- Mahunka S. 1994. Further notes, additions and redescriptions of the oribatid species preserved in the Berlese collection (Acari, Oribatida) I. Acta Zool. Acad. Sci. Hung., 40(1): 29-49.
- Norton R.A. 1977. A review of F. Grandjean's system of leg chaetotaxy in the Oribatei (Acari) and its application to the family Damaeidae. In: Dindal D.L. (Ed.). Biology of oribatid mites. Syracuse: SUNY College of Environmental Science and Forestry. pp. 33-61.
- Norton R.A., Behan-Pelletier V.M. 2009. Oribatida. Chapter 15. In: Krantz G.W., Walter D.E. (Eds.). A Manual of Acarology. Lubbock: Texas Tech University Press. pp. 430-564.
- Pearce N.D.F. 1906. VII - On some Oribatidae from the Sikkim Himalaya. Journ. Roy. Microscop. Soc., 5: 269-273. https://doi.org/10.1111/j.1365-2818.1906.tb01643.x
- Subías L.S. 2021. Listado sistemático, sinonímico y biogeográfico de los Ácaros Oribátidos (Acariformes: Oribatida) del mundo (excepto fósiles), 16ª actualización. 532 pp. Available from: http://bba.bioucm.es/cont/docs/RO_1.pdf
 (accessed March 2021).
(accessed March 2021). - Travé J., Vachon M. 1975. François Grandjean. 1882-1975 (Notice biographique et bibliographique). Acarologia, 17(1): 1-19.



2021-05-13
Date accepted:
2021-07-05
Date published:
2021-07-21
Edited by:
Baumann, Julia

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Ermilov, Sergey G.; Subias, Luis S.; Shtanchaeva, Umukusum Ya. and Friedrich, Stefan
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



