Does diet affect morphological parameters of Neoseiulus tunus (De Leon) (Acari: Phytoseiidae)?
de Melo Ferreira, Mizael  1
; Micali Nuvoloni, Felipe
1
; Micali Nuvoloni, Felipe  2
; de Souza Mondin, Alexandre3
and Lofego, Antonio Carlos
2
; de Souza Mondin, Alexandre3
and Lofego, Antonio Carlos  4
4
1✉ Departamento de Zoologia e Botânica, UNESP, Rua Cristóvão Colombo, 2265, Jardim Nazareth, São José do Rio Preto, São Paulo, 15054-000, Brasil.
2Centro de Formação em Ciências Ambientais, Universidade Federal do Sul da Bahia, Rodovia Porto Seguro-Eunápolis, BR-367, km 10, Porto Seguro, Bahia, 45810-000, Brazil.
3Departamento de Zoologia e Botânica, UNESP, Rua Cristóvão Colombo, 2265, Jardim Nazareth, São José do Rio Preto, São Paulo, 15054-000, Brasil.
4Departamento de Zoologia e Botânica, UNESP, Rua Cristóvão Colombo, 2265, Jardim Nazareth, São José do Rio Preto, São Paulo, 15054-000, Brasil.
2021 - Volume: 61 Issue: 2 pages: 486-496
https://doi.org/10.24349/acarologia/20214443Orginal research
Keywords
Abstract
Introduction
Environment heterogeneity may affect plastic characteristics of animals in different ways (e.g. age and size at maturity) (Pigliucci 2001). Also, Mikolajewski et al. (2005) points out that the size of many mature organisms is clearly modified as a response to the food limitations and availability.
Morphological intraspecific variation has been reported in several groups of mites, being quite common for species and genus of Phytoseiidae. Furtado (1997) verified the occurrence of small variations of some structures, especially the length of setae when they are reared providing different diets. Lofego (1998) analyzed morphological parameters of around 50 phytoseiid species and found that some of them showed substantial variation among populations from different regions of Brazil, such as highlighted by the deviation up to 100% in the length of some setae from the dorsal shield of Euseius alatus (DeLeon). Tixier et al. (2008) studying 10 populations of Neoseiulus californicus (McGregor) from eight countries (Brazil, Chile, France, Greece, Italy (Tuscany), Italy (Sicily), Japan, Spain and Tunisia), pointed out that most variables had significant differences among the studied groups, and also proposed a statistical approach to study the intraspecific variations of characters used in species diagnosis (Tixier 2012). Several explanations are provided to explain such variation, the body size and biogeographic or biotope origin of the populations due to the environmental characteristics and their reproductive isolation.
The influence of the feeding habit on morphological traits of Phytoseiidae mites has been suggested in several publications (Croft et al. 1999; Tixier et al. 2003, 2004, 2006). However, more information is still needed to determine the dimension of this impact. Phytoseiidae (Acari: Mesostigmata) is a family of predatory mites, including some species used in biological control programs worldwide (McMurtry and Croft 1997; Gerson et al. 2003; Hoy 2011; McMurtry et al. 2013). This family comprises 2,866 described species included in 94 genera (Demite et al. 2021).
Different studies report that body size, weight and development characteristics of phytoseiids differ among species and between populations of a same species, depending on the quality and type food that is provided, such as pollen, preys or even artificial diets (McMurtry and Scriven 1966; Badii et al. 1990; Croft et al. 1999; Goleva et al. 2013, 2015; Nguyen et al. 2014; Lopes et al. 2018). Most of phytoseiid species are generalist predators, they can feed wide range of resources such as phytophagous mites (e.g. Tetranychidae, Eriophyioideae, Tenuipalpidae and Tarsonemidae), small insects (e.g. Scirtothrips dorsalis Hood, Thripidae and Bemisia tabaci Gennadius, Aleyrodidae), nematodes, fungi and even plant tissues such as pollen and exudates (Gerson et al. 2003; McMurtry et al. 2013; Amaral et al. 2018). The wide range of feeding items consumed by phytoseiids allows us to ask whether different diets used by the same species may result in morphological variation.
The present study aims to determine whether the diet causes variation in the morphological traits of the phytoseiid Neoseiulus tunus (De Leon), and which parameters are more affected by three distinct diets.
Material and methods
Origin of specimens and rearing techniques
Specimens of N. tunus in the experiment were obtained in samplings on Trichilia casaretti L. (Meliaceae) in a forest fragment located in the municipality of Icém – SP, Brazil (20°20'48'' S; 49°15'49'' W), in the year of 2014. Neoseiulus tunus is a widespread species, easily found in native and cultivated plants in the northeastern of São Paulo state. This species is barely studied for biological control purposes, although it was already observed consuming Tetranychus urticae in field and laboratory. So, due to the potential use for biological control and being widespread in the studied area, this species was elected for the study.
They were kept in the laboratory starting a stock colony at breeding units composed by a vinyl floor plate (Paviflex®) placed on a foam, inside a tray containing distilled water. The edges of the plates were framed by cotton in contact with the foam, consequently both (the foam and the cotton) remained continuously soaked by the water kept in the tray. These breeding units were placed in an incubator chamber (B.O.D.) at 25 ± 1 °C, relative humidity of 60 ± 10% and photoperiod of 12 hours. The mites from the stock colony were feeding with diverse food items, including cattail pollen (Typha angustifolia L.) (Thyphaceae), added each 2 days, supplied with recently emerged individuals and eggs of Tyrophagus putrescentiae (Schrank) (Acaridae) who served as an alternative food source for phytoseiids. Throughout the development of the colonies, Aculops lycopersici (Massee) (Eriophyidae) and Tetranychus urticae (Kock) (Tetranychidae) were offered as complementary feeding resource in their diet. The populations of the stock colony were transferred to new breeding units at least once a month or whenever necessary, thus avoiding presence of accumulated debris, fungi and excretes that could damage the feeding and reproduction of these mites.
The cattail pollen was continuously collected and according to its availability in the municipalities of São José do Rio Preto - SP and Bady Bassitt - SP, brought to the acarology laboratory and stored / frozen in vials.
The colonies of phytophagous mites A. lycopersici were maintained on Petunia sp. and Lycopersicon esculentum Mill, both plants of the family Solanaceae, while Tetranychus urticae was maintained on Canavalia ensiformes (L.) Decandolle (Fabaceae). The host plants were sowed in greenhouses and then kept in the laboratory condition while the experiment was carried on.
Experimental design
All experiments were conducted in experimental units identical to the rearing units of the stock colony in the laboratory.
Three different treatments (diets) were set to analyze morphological variation of N. tunus: cattail pollen (Typha angustifolia L.) (Thyphaceae), T. urticae and A. lycopersici. For each treatment we stablished seven new colonies (equal to seven breeding units), which were started with 10 adults and possibly fertile females taken from the original stock colony kept in the laboratory. Forty-nine (49) females were randomly withdrawn from the stock colony and mounted in microscopy slides to evaluate the morphology of the initial population, for posterior comparison with treatments. The population of N. tunus of each treatment received only one type of food resource (cattail pollen, T. urticae and A. lycopersici) for the time period of 60 days and were kept in an incubator chamber (B.O.D.) at 25 ± 1 °C, with relative humidity of 60 ± 10% and photophase of 12 hours. In the end of this period, seven adult females were removed from each breeding unit (totaling 49 specimens) and mounted on microscopy slides for the measurement of all the structures and parameters. Every two days the food item was replaced in each colony to ensure the full development of the mites. The water in the tray was replaced whenever necessary to maintain the moisture of the foam and cotton.
Morphological analysis
The chaetotaxy nomenclature followed the system proposed by Rowell et al. (1978) and Chant & Hansell (1971) for the dorsal and ventral shields, respectively. All measurements are given in micrometers (μm).
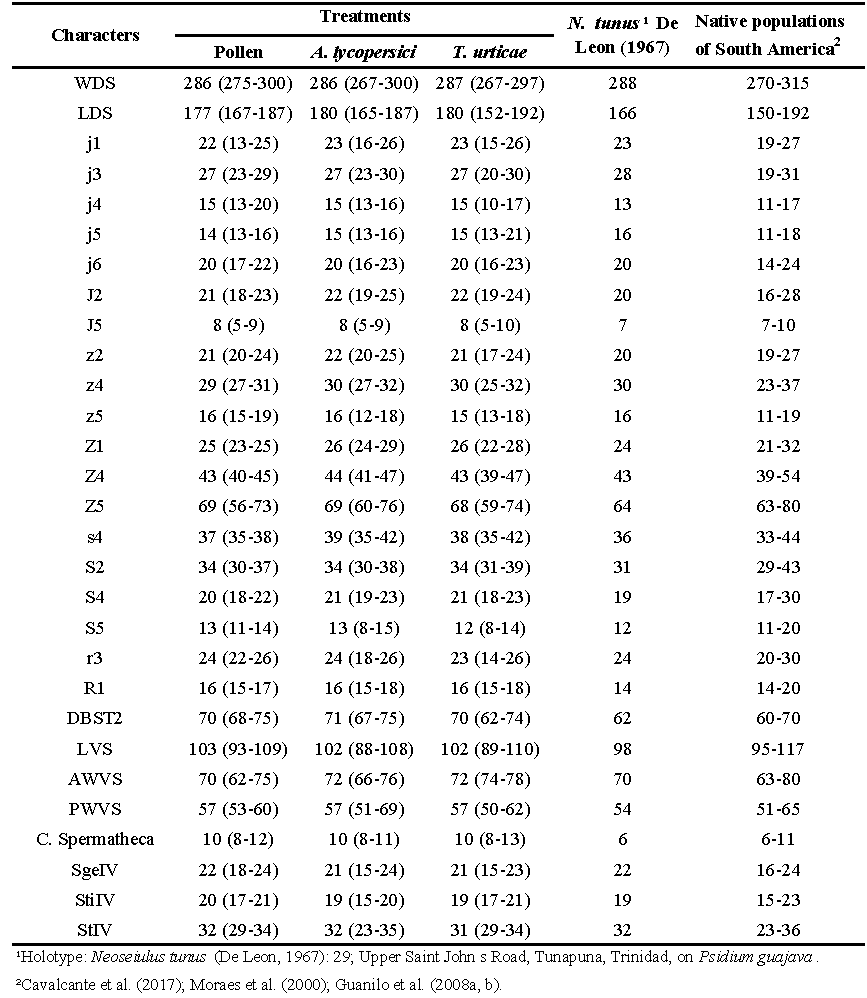
Several structures were analyzed as length of the dorsal shield (LDS), from the base of the j1 setae to the posterior extremity; width of the dorsal shield (WDS) at the level of the setae s4; distance between the bases of the setae ST2 (sternal) (DBST2) and between the bases of the setae ST5 (genital) (DBGS); anterior width of the ventrianal shield (AWVS) at the level of the setae ZV2; posterior width of the ventrianal shield (PWVS), at the level of the para-annals setae; length of the ventrianal shield (LVS) (Figure. 1); length of the dorsal setae j1, j3, j4, j5, j6, J2, J5, z2, z4, Z5, Z1, Z4, Z5, s4, S2, S4, S5, r3 and R1; length of the macrosetae Sge IV (genu IV), Sti IV (tibia IV) and St IV (tarsus IV); length of the cervix of the spermatheca. The measurements were made using an optical microscope with phase contrast (Zeiss Scope A.1), provided with a millimeter ocular lens.
The voucher material was deposited in the collection Acari (DZSJRP) – http://www.splink.cria.org.br ![]() , of the Department of Zoology and Botany, Universidade Estadual Paulista (UNESP), São José do Rio Preto, São Paulo, Brazil.
, of the Department of Zoology and Botany, Universidade Estadual Paulista (UNESP), São José do Rio Preto, São Paulo, Brazil.



Statistical analysis
In order to compare the effect of each treatment in the set of 30 morphological traits we performed a permutational analysis of variance (PERMANOVA, 9999 permutations) with a pairwise comparison. Specific analysis of variance (one-way ANOVA) was also performed for each parameter followed by a post hoc Tukey's test (p < 0.05) considering the variation among treatments. The raw data from the measures were transformed considering Euclidian distance, and were defined 1000 permutations for each test. Therefore, two multivariate statistical analyses were also performed on the 30 quantitative traits. We run a principal component analysis (PCA) to reveal any difference in the morphological variation among the specimens from different treatments and to indicate the contribution of each morphological parameter to each axis of analysis. Thereafter we performed a non-metric multi-dimensional scaling (NMDS) to identify which morphological traits that contributed the most to the morphological differentiation (based on R2 values) among the populations of N. tunus allowing to distinguish them for each treatment. The analyses were performed using vegan package (Oksanen et al. 2019) in R (R core team 2020).
Results
The permutational analysis of variance (PERMANOVA) including all the morphological traits and in relation to the treatments, indicated a considerable high level of significance for the diet test with a (F3,183 = 9.13, p =0.001). Considering the pairwise comparison a posteriori, the morphologic traits of each treatment were distinct from the stock population, and varied among each other (A. lycopersici x Stock population F3,183 = 15.31, p=0.002; Pollen x Stock population F3,183 = 11.79, p=0.02; T. urticae x Stock population F3,183 = 13.061, p= 0.012; T. urticae x pollen F3,183 = 6.29, p= 0.012; A. lycopersici x pollen F3,183 = 6.17, p= 0.013), unless the treatments feed on A. lycopersici and T. urticae that did not differ from each other (F3,183 = 1.37, p=0.273). Those results corroborated the multivariate analysis, since they indicated that most specimens may be grouped according to each treatment, however there were a higher overlap mainly between the treatments of phytophagous species, A. lycopersici and T. urticae (Figure 2). On the other hand, the specimens from stock population and from pollen treatment represented the most distinct populations.
The first two axes of PCA analysis explained about 70% of total morphological variation (PC1: 63.36%; PC2: 6.15%), being the first mainly associated to the dorsal setae (such as z4, Z5, j2, z2, LDS, j6, j1, j3, j6 and Z4), while the second was compound by ventral shields length and spermatheca (DBST2, DBGS, WDS and Spermatheca) (Figure 2). The partial overlap of specimens from distinct treatment indicate that these populations may not be totally separated and distinguished by their morphological traits, despite there are some evident effects of the feeding habit over some characteristics.



The NMDS analysis also presented similar results from PCA, with a supplementary information about the main morphological traits that allowed to distinguish the specimens among treatment (Table1). These results indicate that traits related to ventral shields and dorsal setae are the main drivers of morphological differences among those population (Table 1).
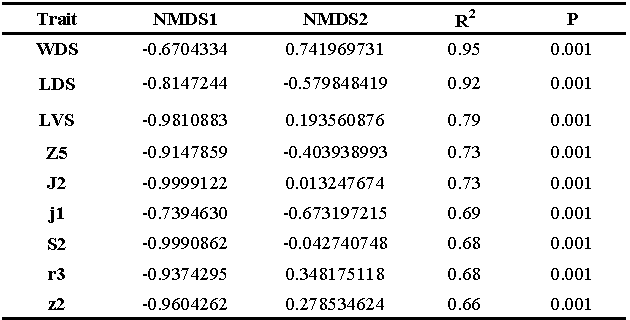


Among the thirty variables, eighteen of them showed significant differences between the diets. (WDS, DBGS, PWVS, Sge IV, Sti IV, St IV, j3, j2, z2, z4, z5, Z1, Z4, s4, S4, S5, r3 and R1).
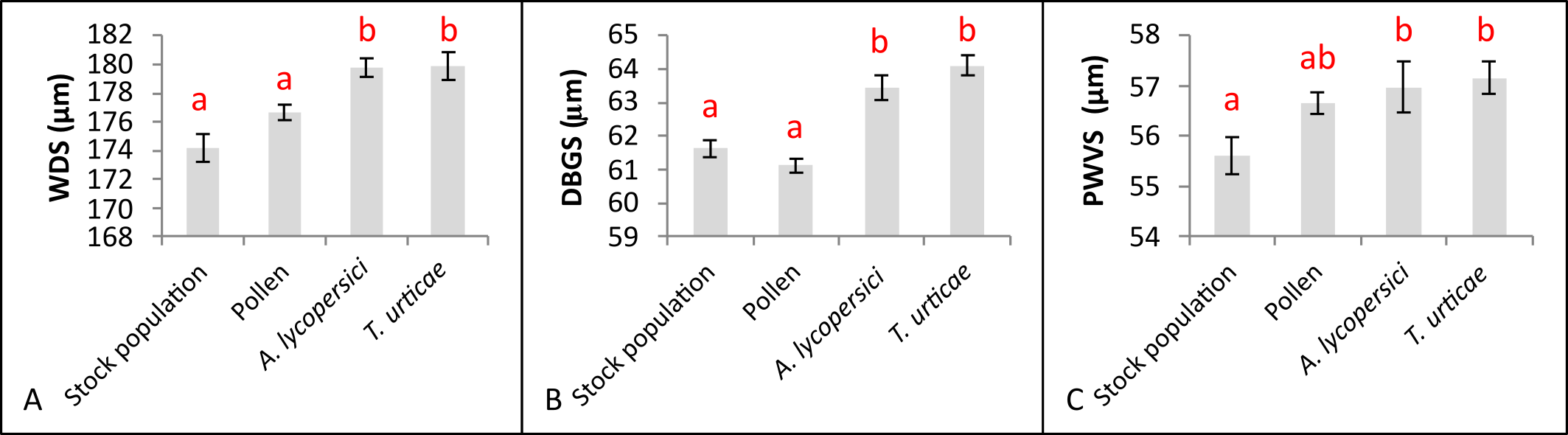
For the morphological traits corresponding to shields (WDS, DBGS and PWVS), the measurements of these structures were significant higher when the mites were fed with phytophagous mites than with pollen (Figure 3), despite they did not differ between each other.


Considering the measures of macrosetae from the legs, those of the genu IV and tibia IV presented longer dimensions in the treatments with pollen and A. lycopersici when compared with the stock population (Figures 4. a & b). The macroseta of the tarsus IV was significantly lower only when the prey provided was T. urticae (Figure 4.c).



The effect of diet type was observed in most of the dorsal setae. The longest setae size was remarked when A. lycopersici was offered as food item, since this pattern was observed for 9 of 12 dorsal setae studied (Figure 5). Considering the other two treatments, the variation in the setae size did not present a clear pattern (Figure 5).



Despite the differences observed in the setae length among the treatments, it is possible to observe that the range of measurements obtained are within the spectrum of length obtained by specimens of N. tunus collected in the field from different regions by Cavalcante et al. (2017); Moraes et al. (2000); Guanilo et al. (2008a, b) (Table 2).


Discussion
Variations in the morphological traits are usually observed in the populations of phytoseiid mites. According to our results, part of these variations may be related to the feeding resource available. Some food items might have the tendency to induce the increasement of the structure lengths, such as observed for A. lycopersici; or influencing either reduction or increasement other structures, as observed for T. urticae.
Some of the most significant variations were observed in the length of the dorsal setae and the in the macrosetae of leg IV. A similar pattern was found by Noronha (2002) in an experiment carried out on Euseius citrifolius (Denmark & Muma). Abou-Setta et al. (1987) studding Euseius mesembrinus (Dean) from Florida and Texas (USA) pointed out some morphological range in the setae according to geographic and diet effect, also noticed that such variation occurred in organisms collected in the field although it was not observed in laboratory conditions.
Hoying et al. (1977) reported morphological variations among eight populations of Galendromus longipilus (Nesbitt) and 12 populations of Galendromus occidentalis (Nesbitt) collected in different regions of the United States of America. In this study they tracked the main variation associated to the length of the peritreme and length of the dorsal shield. In the present study we did not evaluate peritremal shield, but the length of the dorsal shield (LDS) had no variation considering distinct diets. However, the width of the dorsal shield (WDS) was a character that changed in dimension when the resource offered was phytophagous mites (A. lycopersici and T. urticae). Few studies reporting morphological variations in the dorsal shields, Tixier et al. (2008) highlighted that specimens from Brazil had higher values for the length of the metapodal plate 1 (not measured in the present study) and width of the ventrianal shield. In our experiment, some variation was observed only for measurements of the posterior region of the ventrianal shield, and only when phytophagous mites were provided as food resource.
Goleva et al. 2015 shed a light about the tendency of variation in the in morphological traits in diverse Phytoseiidae populations, considering that limitation and quality of the food supply are strictly linked to the development, longevity, and reproduction of predatory mites. The study highlights the influence of different types of pollen (Betula pendula Roth, Typha latifolia L., Pinus sylvestris L., among other sources) and moth eggs (Cydia pomonella L. (Lepidoptera, Tortricidae) mainly on the body weight of the mites, linking body weight with reduction or increase in the body sizes due to the diet which the phytoseiids were subjected to.
Our data supports that diet type is a key element driving the shifts of morphometric traits in the populations of N. tunus. Understanding these variations is important since the morphological determination of a species is based in the entire range of its traits and structures. The particular determination of this range can only be done with laboratory experiments at controlled conditions. This way, we can ensure that a certain range in the morphological variation may correspond in fact to the intraspecific variation and not due to contamination by another species in a given population. In this context, there is also a necessity of more efforts to investigate the effects of diets on other species of the family Phytoseiidae, in order to better understand the causes of intra-specific variations and consequently avoid errors of identification.
Acknowledgements
To the Coordenadoria de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for granting scholarships to M. M Ferreira and CNPq by a research productivity fellowship to A.C. Lofego (No. 310617/2018-9). To Ms. Felipe S. R. Do Amaral and Letícia Molina Hernandes for their contribution in the maintenance of breeding arenas.
References
- Abou-Setta M.M., Childers C.C. 1987. Biology of Euseius mesembrinus (Acari: Phytoseiidae): life tables on ice plant pollen at different temperatures with notes on behavior and food range. Exp. Appl. Acarol., 3: 123-130. doi:10.1007/BF01270474
- Amaral F.S.R., Lofego A.C., Cavalcante A.C.C. 2018. Oviposition rates of Amblyseius aerialis (Muma) and Amblyseius chiapensis De Leon (Acari: Phytoseiidae) under seven foods different patterns for the same genus. Syst. Appl. Acarol., 23(5): 795-798. doi:10.11158/saa.23.5.1
- Badii M.H., McMurtry J.A., Johnson H.G. 1990. Comparative life-history studies on the predaceous mites Typhlodromus annectens and T. porresi (Acari: Mesostigmata: Phytoseiidae). Exp. Appl. Acarol., 10: 129-136. doi:10.1007/BF01194088
- Cavalcante A.C.C., Demite P.R., Amaral F.S.R., Lofego A.C., Moraes G.J. 2017. Complementary description of Neoseiulus tunus (De Leon) (Acari: Mesostigmata: Phytoseiidae) and observation on its reproductive strategy. Acarologia, 57(3): 591-599. doi:10.24349/acarologia/20174178
- Chant D.A., Hansell R.I.C. 1971. The genus Amblyseius (Acarina: Phytoseiidae) in Canada and Alaska. Can. J. Zool., 49(5): 703-758. doi:10.1139/z71-110
- Croft B.A., Luh H.K., Schausberger P. 1999. Larval size relative to larval feeding, cannibalism of larvae, egg or adult female size and larval adult setal patterns among 13 phytoseiid mite species. Exp. Appl. Acarol., 23: 599-610.
- De Leon D. 1967. Some mites of the Caribbean Area. Part I. Acarina on plants in Trinidad, West Indies. Allen Press Inc., Lawrence. pp. 66.
- Demite P.R., Moraes G.J., McMurtry J.A., Denmark H.A., Castilho R.C. 2021. Phytoseiidae Database. Available from: http://www.lea.esalq.usp.br/phytoseiidae. Accessed 17/03/2021.
- Furtado I.P. 1997. Biosystematics and Biology of Euseius (Acari: Phytoseiidae) species associated with cassava [Phd Thesis]. Piracicaba: Escola Superior de Agricultura "Luiz de Queiroz", University of São Paulo. pp. 105
- Gerson U., Smiley R.L., Ochoa R. 2003. Mites (Acari) for pest control. Blackell Science, Oxford, UK. doi:10.1002/9780470750995
- Goleva I., Cadena E.C.R., Ranabhat N.B., Beckereit C., Zebitz C.P.W. 2015. Dietary effects on body weight of predatory mites (Acari, Phytoseiidae). Exp. Appl. Acarol., 66: 541-553. doi:10.1007/s10493-015-9920-5
- Goleva I., Zebitz C.P.W. 2013. Suitability of different pollen as alternative food for the predatory mite Amblyseius swirskii Athias-Henriot (Acari, Phytoseiidae). Exp. Appl. Acarol., 61: 259-283. doi:10.1007/s10493-013-9700-z
- Guanilo A.D., Moraes G.J., Knapp M. 2008a. Phytoseiid mites (Acari: Phytoseiidae) of the subfamily Amblyseiinae Muma from Peru, with description of four new species. Zootaxa, 1880: 1-47. doi:10.11646/zootaxa.1880.1.1
- Guanilo A.D., Moraes G.J., Toledo S., Knapp M. 2008b. Phytoseiid mites (Acari: Phytoseiidae) from Argentina, with description of a new species. Zootaxa 1884: 1-35. doi:10.11646/zootaxa.1884.1.1
- Hoy M. 2011. Agricultural acarology: introduction to integrated mite management. CRC Press, Boca Raton.
- Hoying S.A., Croft B.A. 1977. Comparisons between populations of Typhlodromus longipilus Nesbitt and T. occidentalis Nesbitt: taxonomy, distribuition, and hybridization. Ann. Entomol. Soc. Am., 70(1): 150-159. doi:10.1093/aesa/70.1.150
- Lofego A.C. 1998. Morphological characterization and geographic distribution of the species of Amblyseiinae (Acari: Phytoseiidae) in Brazil [Phd Thesis]. Institute of Biosciences, University of São Paulo. pp. 167.
- Lopes P.C., Kanno R.H., Sourassou, N.F., Moraes G.J. 2018. Effect of temperature and diet on the morphology of Euseius concordis (Acari: Phytoseiidae). Syst. Appl. Acarol., 23(7): 1322-1332. doi:10.11158/saa.23.7.9
- McMurtry J.A., Croft B.A. 1997. Life-styles of Phytoseiid mites and their roles in biological control. Ann. Rev, Entomol., 42: 291-321. doi:10.1146/annurev.ento.42.1.291
- McMurtry J.A., Moraes G.J., Sourassou N.F. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. doi:10.11158/saa.18.4.1
- McMurtry J.A., Scriven G.T. 1966. Effects of artificial foods on reproduction and development of four species of phytoseiid mites. Ann. Entomol. Soc. Am., 59: 267-269. doi:10.1093/aesa/59.2.267
- Mikolajewski D.J., Brodin T., Johansson F., Joop G. 2005. Phenotypic plasticity in gender specific life history: effects of food availability and predation. Oikos. 110(1): 91-100. doi:10.1111/j.0030-1299.2005.13766.x
- Moraes G.J. de, Kreiter S., Lofego A.C. 2000. Plant mites (Acari) of the French Antilles. 3. Phytoseiidae (Gamasida). Acarologia, 40: 237-264.
- Nguyen D.T., Vangansbeke D., DeClercq P. 2014. Solid artificial diets for the phytoseiid predator Amblyseius swirskii. Biocontrol, 59: 719-727. doi:10.1007/s10526-014-9607-6
- Noronha A.C.S. 2002. Morphological and molecular characterization of predatory mites of the genus Euseius (Acari, Phytoseiidae) [Phd Thesis]. Piracicaba: Escola Superior de Agricultura "Luiz de Queiroz", University of São Paulo. 110 p.
- Oksanen J., Blanchet F.G., Friendly M., Kindt R., Legendre P., McGlinn D., Wagner H. 2019. vegan: Community ecology package. R package version 2.5-6. Available from: https://CRAN.R-project.org/package=vegan
 .
. - Pigliucci M. 2001. Phenotypic plasticity. In: Fox C.W., Roff D.A., Fairbairn D.J., (eds.) Evolutionary Ecology: Concepts and Case Studies. Oxford University Press, Oxford. pp. 58-69. doi:10.1093/oso/9780195131543.003.0009
- R Core Team. 2020. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available from: https://www.R-project.org/

- Rowell H.J., Chant D.A., Hansell R.I.C. 1978. The determination of setal homologies and setal patterns on the dorsal shield in the family Phytoseiidae (Acarina: Mesostigmata). Can. Entomol., 110: 859-876. doi:10.4039/Ent110859-8
- Tixier, M-S. 2012. Statistical approaches to assess intraspecific variations of morphological continuous characters: the case study of the family Phytoseiidae (Acari: Mesostigmata). Cladistics, 28: 489-502. doi:10.1111/j.1096-0031.2012.00394.x
- Tixier M-S., Guichou S., Kreiter S. 2008. Morphological variation in the biological control agent Neoseiulus californicus (McGregor) (Acari: Phytoseiidae): consequences for diagnostic reliability and synonymies. Invertebr. Syst., 22: 453-469. doi:10.1071/IS07052
- Tixier M-S., Kreiter S., Barbar Z., Ragusa S., Cheval B. 2006. The status of two cryptic species: Typhlodromus exhilaratus (Ragusa) and Typhlodromus phialatus (Athias-Henriot) (Acari: Phytoseiidae): consequences for taxonomy. Zool. Scr., 35: 115-122. doi:10.1111/j.1463-6409.2006.00222.x
- Tixier M-S., Kreiter S., Cheval B., Auger P. 2003. Morphometric variation between populations of Kampimodromus aberrans (Oudemans) (Acari: Phytoseiidae). Implications for the taxonomy of the genus. Invertebr. Syst., 17: 349-358. doi:10.1071/IS02004
- Tixier M-S., Kreiter S., Croft B.A., Cheval B. 2004. Morphological and molecular differences in the genus Kampimodromus (Nesbitt). Implications for taxonomy. Phytophaga, 14: 361-375.



2020-11-17
Date accepted:
2021-05-04
Date published:
2021-05-25
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 de Melo Ferreira, Mizael; Micali Nuvoloni, Felipe; de Souza Mondin, Alexandre and Lofego, Antonio Carlos
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



