Phytoseiid mites of the French Guiana (Acari: Mesostigmata)
Kreiter, Serge  1
and Douin, Martial2
1
and Douin, Martial2
1✉ Institut Agro (Montpellier SupAgro), UMR CBGP INRA/ IRD/ CIRAD/ IA (SupAgro), 755 Avenue du Campus Agropolis (Baillarguet), CS 30016, 34988 Montferrier-sur-Lez cedex, France.
2Institut Agro (Montpellier SupAgro), UMR CBGP INRA/ IRD/ CIRAD/ IA (SupAgro), 755 Avenue du Campus Agropolis (Baillarguet), CS 30016, 34988 Montferrier-sur-Lez cedex, France.
2021 - Volume: 61 Issue: 2 pages: 468-478
https://doi.org/10.24349/acarologia/20214441Original research
Keywords
Abstract
Introduction
Mites of the family Phytoseiidae are known mostly for their predatory habit on phytophagous mites and small insects, like thrips and whiteflies, on cultivated plants and plants of the natural vegetation.
Several species are biological control agents commercialized for the control of pest organisms in both open and protected crops all around the world (McMurtry and Croft 1997; McMurtry et al. 2013; Knapp et al. 2018).
This family is widespread, present on all continents except Antarctica, and it consists of about 2,500 valid species in 94 genera and three subfamilies (Demite et al. 2021).
Biodiversity surveys in poorly investigated areas is still an urgent need and might result in the discovery of additional species potentially useful for biological control as well as in the acquisition of information on the biodiversity of these areas (Kreiter et al. 2018a, b, c, 2020a, b, c, d, 2021a, b, c; Ferragut and Baumann 2019, 2020, 2021; Döker et al. 2020; Kreiter and Abo-Shnaf 2020a, b; Fang et al. 2020; Demite et al. 2021; Toldi et al. 2021).
Located close to Amazonas, Guianas constitutes one of the world's highest biodiversity places. The concept of biodiversity hotspot was defined by Myers (1988) in order to identify the most immediately important areas for biodiversity conservation. These hotspots hold high endemism levels and have lost at least 70% of their original natural vegetation (Myers et al. 2000). Knowledge about the phytoseiid diversity in these areas may contribute to future establishment of conservation programs.
Located in South America, in the North Atlantic Ocean Coast, at more than 7000 km from Paris, French Guiana is one of the Guianas and the only one territorial collectivity of France in this area. It borders Brazil to the east and south (State of Amapá) and Suriname to the west.
Only one phytoseiid species has been recorded until now from this large geographical area, Amblydromalus limonicus (Garman and McGregor), recorded by Murphy (1984).
The objective of this paper is to present the phytoseiid species recorded during a small survey conducted at the end of 2001 in some localities of French Guianas.
Material and methods
The survey took place in October – November, 2001, collecting the phytoseiid mites found on leaves of cultivated and wild plants examined in the field and transferring them to vials containing 70% ethanol. All collections were made by Cécile Rault (Sud-Agro Formation, Bordeaux, France).
The mites were then mounted in Hoyer's medium (Walter and Krantz 2009) for later examination and identification using a phase and interferential contrast microscope (DMLB, Leica Microsystèmes SAS, Nanterre, France). Characters of specimens were measured using a graded eyepiece.
Chant and McMurtry's (1994, 2007) concepts of the taxonomy of the family Phytoseiidae for identification and the world catalogue database of Demite et al. (2014, 2021) for distribution and information on descriptions and re-descriptions were used. The setal nomenclature system adopted was that of Lindquist & Evans (1965) and Lindquist (1994) as adapted to the phytoseiids by Rowell et al. (1978) and Chant & Yoshida-Shaul (1989) for the dorsal surface and by Chant & Yoshida-Shaul (1991) for the ventral surface. Pore (= solenostome) and poroid (= lyrifissure) notations are that of Athias-Henriot (1975). Macrosetal notation (Sge = genual macroseta; Sti = tibial macroseta; St = tarsal macroseta) are that of Muma and Denmark (1970). Numbers of teeth on the fixed and movable cheliceral digits do not include the respective apical tooth. Setae not referred to in the result section should be considered as absent. All measurements are given in micrometres and presented with the mean in bold followed by the range in parenthesis. Type of spermatheca or insemination apparatus are that of Denmark and Evans (2011). Only some species with only few measurements mentioned in the literature are provided in this paper. Classification of plants follows the APG IV classification of 2016 (ex. Byng et al. 2018).
Specimens of each species are deposited in the mite collections of Montpellier SupAgro conserved in UMR CBGP INRA/IRD/CIRAD/SupAgro/University of Montpellier.
The following abbreviations are used in table 1 of this paper for morphological characters: dsl = dorsal shield length just anteriad of j1 to just posteriad of J5 in the middle line; dsw = dorsal shield width at the level of s4; Z4 ser., Z5 ser. = Z4, Z5 serrated (if Z4 and Z5 without ser. = not serrated); gensl = genital shield length; gensw st5 = genital shield width at level of steae st5; gensw post. cor. = genital shield width at level of posterior corners; lisl = primary or largest inguinal sigilla (= ''metapodal plate'') length; lisw = primary or largest inguinal sigilla (= ''metapodal'') width; sisl = secondary or smallest inguinal sigilla (= ''metapodal'' length; vsl = ventrianal shield length; gv3 – gv3 = distance between centers of solenostomes gv3 on the ventrianal shield; vsw ZV2 & vsw anus = ventrianal shield width at ZV2 level and at paranal setae level; scl: calyx total length; scw = calyx widest width; Fdl = fixed digit length; Mdl = movable digit length; Nb teeth Fd = number of teeth on the fixed digit; Nb teeth Md = number of teeth on the movable digit; Shaft = length of the shaft of spermatodactyl; toe = length of the toe; BCA = Biological control agent; aasl = altitude above sea level; imm.: immature.
The following abbreviations are used in this paper for institutions: CBGP = Centre de Biologie pour la Gestion des Populations; CIRAD = Centre International de Recherche Agronomique pour le Développement; IA = Institut Agro; INRAE = Institut National de Recherche pour l'Agriculture, l'Alimentation et l'Environnement; INRAPE = Institut National de Recherche pour l'Agriculture, la Pêche et L'Environnement; IRD = Institut de Recherche pour le Développement; MSA = Montpellier SupAgro, France; UMR = Unité Mixte de Recherche; UR = Unité de Recherche.
Results and discussion
A total of five species have been recorded.
Subfamily Amblyseiinae Muma
Amblyseiinae Muma 1961: 273.
Tribe Typhlodromipsini Chant & McMurtry
Typhlodromipsini Chant & McMurtry 2005b: 318.
Genus Typhlodromips De Leon
Typhlodromipsini Chant & McMurtry 2005b: 318; 2006: 137; 2007: 55.
Typhlodromips scleroticus De Leon
Typhlodromips scleroticus De Leon, 1966: 96; Moraes et al. 1986: 147, 2004: 223; Chant & McMurtry 2005b: 327, 2007: 63.
This species was described by De Leon (1966) from the Nature Reserve of Bartica at Mazaruni Potaro, in British Guyana, on Pouteria sp. (Sapotaceae). It belongs to the ariri species group of the genus Typhlodromips as seta J1 is absent and spermatheca has calyx cup- or bell-shaped or saccular. Its biology is totally unknown and this is the first record of that species for French Guiana.
World distribution: Guyana (British Guyana).
Specimens examined: A single ♀ collected during this study. Saint-Laurent-du-Maroni, Chemin de Mana (20 m aasl, lat. 5°29'18'' N, long. 53°58'02'' W), 1 ♀ on Mangifera indica (Anacardiaceae), 27/X/2001.
Remarks: the description of this species is very concise, including small illustrations. A complementary and more detailed description is presented, with measurements of our single specimen (in bold) compared to those of the original description (between parenthesis).
Adult female (Figs. 1a – e)
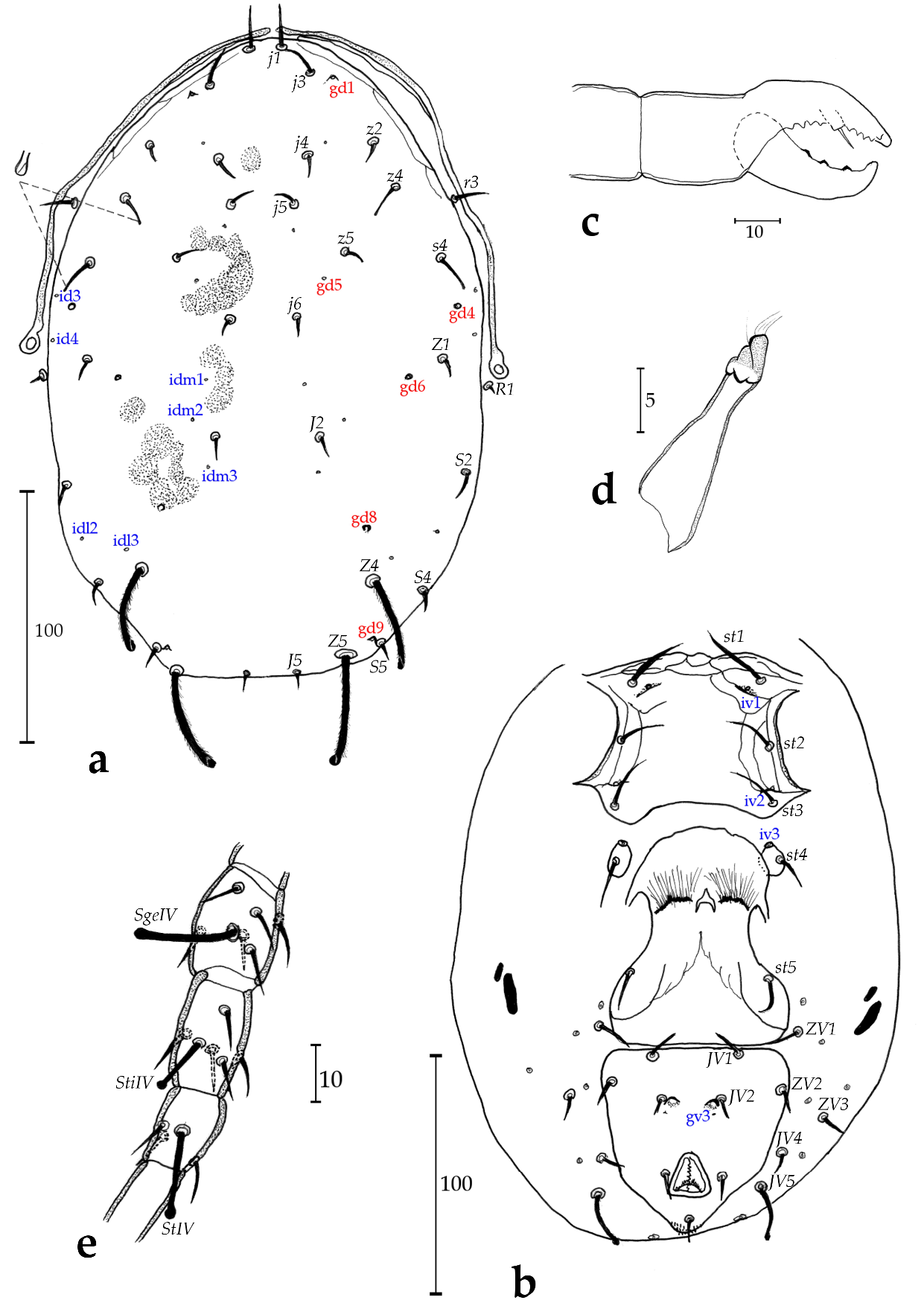


Dorsum (Fig. 1a) — Dorsal shield fused, smooth except for scant reticulation near the anterolateral margins of the dorsal shield, 270 (271) long and 173 (181) wide, with five solenostomes (gd1, gd5, gd6, gd8, and gd9, gd1 and gd9 being crateriform, others being oblong), only 10 pairs of poroids visible, 17 pairs of dorsal setae and two pairs of sub-lateral setae off the dorsal shield: j1 15 (15), j3 15 (15), j4 9 (11), j5 10 (7), j6 8 (7), J2 9 (7), J5 7 (7), z2 8 (7), z4 13 (14), z5 8 (7), Z1 9 (10), Z4 35 (32), Z5 40 (36), s4 15 (15), S2 10 (9), S4 6 (5), S5 5 (5), r3 10, R1 9. All setae smooth except Z4 and Z5 which are serrated and all setae sharp-tipped except z4, s4, Z4, Z5 and JV5 which are knobbed, Z4 and Z5 being also serrated.
Peritreme and peritrematal plate (Fig. 1a) — Reaching the level between insertion of setae j1; peritrematic plate fused with dorsal shield at level of j1.
Venter (Fig. 1b) — All ventral shields mostly smooth. Sternal shield with few striae, three pairs of setae and two pairs of lyrifissures; metasternal seta (st4) on large metasternal plates that also bear a large poroid; posterior margin of the sternal shield straight to slightly concave. Distances between st1-st1 54, st2-st2 60, st3-st3 65, st1-st3 50, st4-st4 68. Genital shield length 90, width at the level of st5 63, width at the level of the posterior corners 75, distance st5-st5 56. Two pairs of metapodal plates, the larger 13 long and 2 wide, the smaller 8 long and 2 wide. Ventrianal shield with three pairs of preanal setae (JV1, JV2 and ZV2), two small oblong pre-anal solenostomes 20 apart. Cribrum spicules on three lines. Membrane surrounding ventrianal shield with four pairs of setae (ZV1, ZV3, JV4 and JV5), and seven pairs of round to oblong poroids around genital/ventrianal shields; ventrianal shield 80 (80) long, 80 (76) wide at level of anterior corners (ZV2), and 60 wide at level of anus. JV5 knobbed but smooth 23 (24) long.
Chelicera (Fig. 1c) — Chelicerae regular size for phytoseiids. Fixed digit 35 (27) long with 9 (8-9) teeth and a pilus dentilis; movable digit 38 long with 3 (3) teeth.
Spermatheca (Fig. 1d) — Spermatheca saccular, with a moderately elongate calyx 13 (4.5) long and 5 (4.2) wide at maximum, slightly wider next to the vesicle; atrium distinct, although not enlarged.
Legs (Fig. 1e) — Macrosetae on all legs, all knobbed; one on genu of leg I, II and III, one on tibia III and three on leg IV, with one on each of genu, tibia and basitarsus: SgeI 15 (15), SgeII 13 (13), SgeIII 14 (14), Sti III 12, SgeIV 29 (29), StiIV 15 (14), StIV 25 (24). Genu II and III with seven setae each, chaetotactic formula of genu II: 2-2/0, 2/0-1; genu III: 1-2/1, 2/0-1.
Remarks: this species is distinguished from other Typhlodromips by the following characteristics: z4, s4, Z4, Z5 and JV5 and macrosetae of all legs knobbed, Z5 slightly longer than Z4, gv3 adjacent to insertion of JV2.
It is most similar to Typhlodromips igapo Nuvoloni, Lofego, Rezende and Feres (Nuvoloni et al. 2015) and T. sabaculus Denmark and Muma (Denmark and Muma 1973).
Typhlodromips igapo has longer Z5 (62) and leg macrosetae longer (SgeIV and StIV: 32).
Typhlodromips sabaculus has longer Z4 (48) and Z5 (79), macrosetae longer (SgeIV and StIV 38 and 46 respectively).
In the latter two species, z4, s4, Z4 and Z5 are not knobbed and gv3 relatively far from the insertion of JV2.
Tribe Amblyseiini Muma
Amblyseiini Muma 1961: 268.
Sub-tribe Amblyseiina Muma
Amblyseiina Muma 1961: 269.
Genus Amblyseius Berlese
Amblyseius Berlese 1914: 143.
Amblyseius anacardii De Leon
Amblyseius anacardii De Leon 1967: 23; Moraes et al. 1986: 7; 1991: 117, 2000: 238, 2004: 14; Kreiter et al. 2006: 3, Chant & McMurtry 2004: 203, 2007: 75.
Amblyseius (Amblyseius) anacardii, Denmark & Muma 1989: 17.
This species belongs to the obtusus species group of the genus Amblyseius and to the aerialis species subgroup. Amblyseius anacardii is frequent in Antilles (Moraes et al. 2000, Kreiter et al. 2006) and in Central America. Its biology remains unknown. This is the first record in French Guiana.
World distribution: Colombia, Ecuador, Guadeloupe, Les Saintes, Marie-Galante, Martinique, Saint-Martin, Trinidad.
Specimens examined: four specimens (2 ♀♀, 1 ♂, 1 imm.) collected during this study. Saint-Laurent-du-Maroni, Chemin de Mana (20 m aasl, lat. 5°29'18'' N, long. 53°58'02'' W), 1 ♀ and 1 imm. on Citrus limon (L.) Burman (Rutaceae), 1 ♂ on Cocos nucifera L. (Arecaceae) and 1 ♀ on Hibiscus rosa-sinensis L. (Malvaceae), 27/X/2001.
Remarks: morphological and morphometric characters and all measurements fit well the measurement given by De Leon (1967), Denmark and Muma (1989) and Moraes et al. (2000).
Genus Amblyseius largoensis (Muma)
Amblyseiopsis largoensis Muma 1955: 266.
Typhlodromus (Amblyseius) largoensis, Chant 1959: 96.
Amblyseius (Amblyseialus) largoensis, Muma 1961: 287.
Typhlodromus largoensis, Hirschmann 1962: 2.
Amblyseius (Amblyseius) largoensis, Ehara,1966: 22.
Amblyseius largoensis, Swirski & Golan 1967: 225, Moraes et al. 1986: 17, 2004: 33, Chant & McMurtry 2004: 208, 2007: 78.
Amblyseius magnolia, Muma 1961: 289 (Synonymy according to Denmark & Evans 2011).
Amblyseius sakalava, Blommers 1976: 96 (Synonymy according to Ueckermann & Loots 1988).
Amblyseius amtalaensis, Gupta 1977: 53 (Synonymy according to Gupta 1986).
Amblyseius largoensis belongs to the largoensis species group and to the largoensis species subgroup. It is widespread in tropical and subtropical regions of the world and was the most abundant species collected by Moraes et al. (2000) in French Caribbean Islands and an important species in La Réunion Island (Moraes et al. 2012).
Using morphometric analyses of 36 characters, molecular analyses and crossing tests, Navia et al. (2014) studied specimens collected in Brazil, La Réunion Island and Trinidad and Tobago to determine whether A. largoensis populations from different geographic origins belong to the same taxonomic entity. Though differences in the lengths of some setae were observed, molecular analyses and crossing experiments indicated that populations from Indian Ocean and America were conspecific. This is its first record in French Guiana.
World distribution: this species is widely distributed in tropical and subtropical regions of Africa, America, Asia and the Pacific Islands.
Specimens examined: sixteen specimens (13 ♀♀, 1 ♂ and 2 imm.) collected during this study. Saint-Laurent-du-Maroni, Chemin de Mana (20 m aasl, lat. 5°29'18'' N, long. 52°35'00'' W), 1 ♀ and 1 imm. on Musa parasidiaca L. (Musaceae), 27/X/2001; Île du Salut (3 m aasl, lat. 5°17'01'' N, long. 53°58'02'' W), 2 ♀♀ on Cocos nucifera L. (Arecaceae), 4 ♀♀ on an unknown hostplant, 5 ♀♀ on M. paradisiaca, 1/XI/2001, 1 ♀, 1 ♂ and 1 imm. on Capsicum frutescens L. (Solanaceae), 2/XI/2001.
Remarks: morphological and morphometric characters and all measurements fit well the measurement given by Navia et al. (2014) for specimens from Brazil, La Réunion and Trinidad & Tobago.
Sub-tribe Proprioseiopsina Chant & McMurtry
Proprioseiopsina Chant & McMurtry 2004: 219.
Genus Proprioseiopsis Muma
Proprioseiopsis Muma 1961: 277.
Proprioseiopsis neotropicus (Ehara)
Amblyseius neotropicus Ehara 1966: 133, Moraes & Mesa 1988: 79; Moraes et al. 1991: 127.
Proprioseiopsis neotropicus, Moraes et al., 1986: 119; 2004: 183; Gondim & Moraes, 2001: 81; Zacarias & Moraes, 2001: 582, Ferla & Moraes, 2002b: 1019, Lofego et al., 2004: 9; Chant & McMurtry 2005a: 15, 2007: 89; Feres et al., 2005: 4; Guanilo et al., 2008a: 9; 2008b: 12.
Proprioseiopsis neotropicus belongs to the belizensis species group. However, the specimens collected differ from other species in this group by having a macroseta on genu I. As the spermatheca of that species is saccular, it belongs to the belizensis species subgroup (Chant and McMurtry 2005a). Its biology is totally unknown. This is the first record in French Guiana.



World distribution: Argentina, Brazil, Colombia, Ecuador, Peru.
Specimens examined: two specimens (2 ♀♀) collected during this study. Île du Salut (3 m aasl, lat. 5°17'01'' N, long. 53°58'02'' W), 2 ♀♀ on an unknown species of Melastomataceae, 2/XI/2001.
Remarks: this species was described in detail by Ehara (1966) and there is no need for it to be redrawn in this paper. Morphological and morphometric characters and all measurements of our two specimens agree well with measurement given in the literature for specimens from Argentina, Brazil, Colombia and Peru (Ehara 1966, Moraes and Mesa 1988, Lofego et al. 2004, Guanilo et al. 2008a, b), especially for specimens from Brazil (Lofego et al. 2004).
Proprioseiopsis ovatus (Garman)
Amblyseiopsis ovatus Garman 1958: 78.
Typhlodromus (Amblyseius) ovatus, Chant 1959: 90.
Amblyseiulus ovatus, Muma 1961: 278.
Typhlodromus ovatus, Hirschmann 1962: 2.
Proprioseiopsis (Proprioseiopsis) ovatus, Karg 1989: 208.
Proprioseiopsis ovatus, Moraes et al. 1986: 121, 2004: 184; Chant & McMurtry 2005a: 15, 2007: 89.
Proprioseiopsis cannaensis (Muma 1962: 4) (synonymy according to Denmark & Evans 2011).
Proprioseiopsis peltatus (van der Merwe: 1968: 119) (synonymy according to Tseng 1983).
Proprioseiopsis hudsonianus (Chant & Hansell 1971: 723) (synonymy according to Denmark & Evans 2011).
Proprioseiopsis parapeltatus (Wu & Chou, 1981: 274) (synonymy according to Tseng 1983).
Proprioseiopsis antonelli Congdon 2002: 15 (synonymy according to Denmark & Evans 2011).
As reported for the previous species, P. ovatus belongs to the belizensis species group and to the belizensis species subgroup (Chant and McMurtry 2005a). This species is known from Guadeloupe, Marie-Galante and Martinique (Kreiter and Moraes 1997; Moraes et al. 2000; Mailloux et al. 2010; Kreiter et al. 2013, 2018c). It was found in large numbers only in a previous study in Guadeloupe, also in the Caribbean region (Mailloux et al. 2010) and in a recent study in La Réunion (Kreiter et al. 2020d). In other habitats, it seems to be rare. Similar to P. mexicanus, P. ovatus seems to be abundant on weeds and other low vegetation (Mailloux et al. 2010). Denmark and Evans (2011) indicated that this species is associated with Oligonychus pratensis (Banks) and Brevipalpus spp. It was also found in association with Tetranychus evansi Baker and Pritchard (Furtado et al. 2014), but it was mentioned as poor predator of that species by these authors. Despite this information, the biology of P. ovatus remains unknown. This is the first record in French Guiana.
World distribution: Argentina, Brazil, Colombia, Costa Rica, Cuba, Ecuador, Egypt, Ghana, Hawaii, Honduras, Japan, Malaysia, Martinique Island, Mozambique, Peru, Philippines, Puerto Rico, La Réunion Island, Saudi Arabia, Sierra Leone, South Africa, Spain, Sri Lanka, Taiwan, Thailand, Turkey, USA, Venezuela.
Specimens examined: A single ♀ collected during this study. Saint-Laurent-du-Maroni, Chemin de Mana (20 m aasl, lat. 5°29'18'' N, long. 52°35'00'' W), 1 ♀ on Etlingera eliator (Jack) Smith (Zingiberaceae), 27/X/2001.
Remarks: morphological and morphometric characteristics and all measurements of our single specimen fit well those given Kreiter et al. (2020d) and other measurements of the literature mentioned by these authors (van der Merwe 1968, Blommers 1976, Moraes and McMurtry 1983, Moraes et al. 2004a, Moraes et al. 2007a, Oliveira et al. 2012, Kreiter et al. 2018c, Kreiter et al. 2020d) for specimens from La Réunion and other parts of the world.
Conclusion
A total of five phytoseiid species, all Amblyseiinae, was collected in this study. However, only six species have so far been reported from French Guiana, namely A. limonicus, A. anacardii, A. largoensis, P. neotropicus, P. ovatus and T. scleroticus.
At least two of these species have been mentioned as biological control agents (BCA): A. largoensis and A. limonicus. In addition ecological interest concerning the determination of the distribution of the phytoseiid mites worldwide, the results of this study also has a pragmatic importance. Given the restrictions imposed by some countries in relation to the transportation of beneficial organisms between different geographic areas for practical use, the determination of the distribution of a species with potential value as biological control agent may allow the selection of local species for that purpose.
French Guiana is the largest French Department, with a territory of comparable size to some European countries. Given its location next to the equator, it is expected that many more phytoseiid species can be found in that region.
This large area belongs to Amazonas biome. So, further surveys and investigations are of great potential interest.
Acknowledgements
To Mrs Cécile Rault (Sud-Agro Formation, Bordeaux, France), the collector of the specimens considered in this study, who made them available to the authors of this publication for identification.
References
- Athias-Henriot C. 1975. Nouvelles notes sur les Amblyseiini. II. Le relevé organotaxique de la face dorsale adulte (Gamasides protoadéniques, Phytoseiidae). Acarologia, 17(1): 20-29.
- Berlese A. 1914. Acari nuovi. Manipulus IX. Redia, 10: 113-150.
- Blommers L. 1976. Some Phytoseiidae (Acarina: Mesostigmata) from Madagascar, with descriptions of eight new species and notes on their biology. Bijd. Dierk., 46(1): 80-106. doi:10.1163/26660644-04601005
- Byng J.W., Smets E.F., van Vugt R., Bidault E., Davidson C., Kenicer G., Chase M.W., Christenhusz M.J.M. 2018. The phylogeny of angiosperms poster: a visual summary of APG IV family relationships and floral diversity. The Global Flora, 4-7.
- Chant D.A. 1959. Phytoseiid mites (Acarina: Phytoseiidae). Part I. Bionomics of seven species in southeastern England. Part II. A taxonomic review of the family Phytoseiidae, with descriptions of thirty-eight new species. Can. Entomol., 61(12): 1-166. doi:10.4039/entm9112fv
- Chant D.A., Hansell R.I.C. 1971. The genus Amblyseius (Acarina: Phytoseiidae) in Canada and Alaska. Can. J. Zool., 49(5): 703-758. doi:10.1139/z71-110
- Chant D.A., McMurtry J.A. 2004. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae): Part III. The tribe Amblyseiini Wainstein, subtribe Amblyseiina n. subtribe. Intern. J. Acarol., 30(3): 171-228. doi:10.1080/01647950408684388
- Chant D.A., McMurtry J.A. 2005a. A review of the subfamily Amblyseiina Muma (Acari: Phytoseiidae): Part V. Tribe Amblyseiini, subtribe Proprioseiopsina Chant and McMurtry. Intern. J. Acarol., 31(1): 3-22. doi:10.1080/01647950508684412
- Chant D.A., McMurtry J.A. 2005b. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae) Part VII. Typhlodromipsini n. tribe. Intern. J. Acarol., 31(4): 315-340. doi:10.1080/01647950508683673
- Chant D.A., McMurtry J.A. 2006. A review of the subfamily Amblyseiinae Muma (Acari: Phytoseiidae): Part IX. An overview. Intern. J. Acarol., 32(2): 125-152. doi:10.1080/01647950608684453
- Chant D.A., McMurtry J.A. 2007. Illustrated keys and diagnoses for the genera and subgenera of the Phytoseiidae of the world (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, 219 pp.
- Chant D.A., Yoshida-Shaul E. 1989. A world review of the tiliarum species group in the genus Typhlodromus Scheuten (Acari: Phytoseiidae). Can. J. Zool., 67(4): 1006-1046. doi:10.1139/z89-144
- Chant D.A., Yoshida-Shaul E. 1991. Adult ventral setal patterns in the family Phytoseiidae (Acari: Gamasina). Intern. J. Acarol., 17(3): 187-199. doi:10.1080/01647959108683906
- Congdon B.D. 2002. The family Phytoseiidae (Acari) in western Washington State with descriptions of three new species. Intern. J. Acarol., 28(1): 3-27. doi:10.1080/01647950208684275
- De Leon D. 1966. Phytoseiidae of British Guyana with keys to species (Acarina: Mesostigmata). Studies on the Fauna of Suriname and other Guyanas, 8: 81-102.
- De Leon D. 1967. Some mites of the Caribbean Area. Part I. Acarina on plants in Trinidad, West Indies. Allen Press Inc., Lawrence, Kansas, USA: 1-66.
- Demite P.R., McMurtry J.A., Moraes G.J. de. 2014. Phytoseiidae Database: a website for taxonomic and distributional information on phytoseiid mites (Acari). Zootaxa, 3795 (5): 571-577. doi:10.11646/zootaxa.3795.5.6
- Demite P.R., Moraes G.J. de, McMurtry J.A., Denmark H.A., Castilho R.C. 2021. Phytoseiidae Database. Available from: www.lea.esalq.usp.br/phytoseiidae
 (last access 16/03/2021).
(last access 16/03/2021). - Denmark H.A., Evans G.A. 2011. Phytoseiidae of North America and Hawaii (Acari: Mesostigmata). Indira Publishing House, West Bloomfield, USA, 451 pp.
- Denmark H.A., Muma M.H. 1973. Phytoseiid mites of Brazil. Rev. Brasil. Biol., 33(2): 235-276.
- Denmark H.A., Muma M.H. 1989. A revision of the genus Amblyseius Berlese, 1914 (Acari: Phytoseiidae). Occas. Pap. Fla State Coll. Arthropods, USA, 4, 149 pp.
- Döker I., Kazak C., Karut K. 2020. The genus Amblyseius Berlese (Acari: Phytoseiidae) in Turkey with discussion on the identity of Amblyseius meridionalis. Syst. Appl. Acarol., 25(8): 1395-1420. doi:10.11158/saa.25.8.4
- Ehara S. 1966. A tentative catalogue of predatory mites of Phytoseiidae known from Asia, with descriptions of five new species from Japan. Mushi, 39: 9-30.
- Fang X., Nguyen V.-L., Ouyang G.-C., Wu W.-N. 2020. Survey of phytoseiid mites (Acari: Mesostigmata, Phytoseiidae) in citrus orchards and a key for Amblyseiinae in Vietnam. Acarologia 60(2): 254-267 doi:10.24349/acarologia/20204366
- Feres R.J.F., Lofego A.C., Oliveira A.R. 2005. Ácaros plantícolas (Acari) da Estação Ecológica do Noroeste Paulista, estado de São Paulo, Brasil. Biota Neotropica, 5(1): 43–56. doi:10.1590/S1676-06032005000100005
- Ferla N.J., Moraes G.J. de. 2002. Ácaros predadores (Acari) em plantas nativas e cultivadas do Estado do Rio Grande do Sul, Brasil. Rev. Brasil. Zool., 19(4): 1011-1031. doi:10.1590/S0101-81752002000400006
- Ferragut F., Baumann J. 2019. New phytoseiid mites (Mesosstigmata: Phytoseiidae) of Mauritius, with the description of two new species. Syst. Appl. Acarol., 24(5), 825-856. doi:10.11158/saa.24.5.8
- Ferragut F., Baumann J. 2020. Hidden biodiversity in the Atlantic Islands. Amblyseiinae (Acari: Phytoseiidae) from Madeira archipelago. Syst. Appl. Acarol., 25(6): 1113-1138. doi:10.11158/saa.25.6.14
- Ferragut F., Baumann J. 2021. New species and new records of phytoseiid mites (Acari: Phytoseiidae) from Cape Verde archipelago. Syst. Appl. Acarol., 26(2), 395-426. doi:10.11158/saa.26.2.6
- Furtado I.P., Moraes G.J. de, Kreiter S., Flechtmann C.H.W., Tixier M.-S., Knapp M. 2014. Plant inhabiting phytoseiid predators of Midwestern Brazil, with emphasis on those associated with the tomato red spider mite, Tetranychus evansi (Acari: Phytoseiidae, Tetranychidae). Acarologia, 54(4): 425-431. doi:10.1051/acarologia/20142138
- Garman P. 1958. New species belonging to the genera Amblyseius and Amblyseiopsis with keys to Amblyseius, Amblyseiopsis, and Phytoseiulus. Ann. Entomol. Soc. Amer., 51: 69-79. doi:10.1093/aesa/51.1.69
- Gondim Jr. M.G.C., Moraes G.J. de 2001. Phytoseiid mites (Acari: Phytoseiidae) associated with palm trees (Arecaceae) in Brazil. Syst. Appl. Acarol., 6: 65-94. doi:10.11158/saa.6.1.11
- Guanilo A.D., Moraes G.J. de, Knapp M. 2008a. Phytoseiid mites (Acari: Phytoseiidae) of the subfamily Amblyseiinae Muma from Peru, with description of four new species. Zootaxa, 1880: 1-47. doi:10.11646/zootaxa.1880.1.1
- Guanilo A.D., Moraes G.J. de, Toledo S., Knapp M. 2008b. Phytoseiid mites (Acari: Phytoseiidae) from Argentina, with description of a new species. Zootaxa, 1884: 1-35. doi:10.11646/zootaxa.1884.1.1
- Gupta S.K. 1977. Some undescribed and little-known species of Amblyseius (Acarina: Phytoseiidae) from western and northern India. Ind. J. Acarol., 1: 28-37.
- Gupta S.K. 1986. Fauna of India (Acari: Mesostigmata) Family Phytoseiidae. Zoological Survey of India, Calcutta, India, 350 pp.
- Hirschmann W. 1962. Gangystematik der Parasitiformes. Acarologie Schriftenreihe fur Vergleichende Milbenkunde, Hirschmann-Verlag, Furth/Bay, 5(5-6), 80 pp.+ 32 plates.
- Karg W. 1989. Neue Raubmilbenarten der Gattuig Proprioseiopsis Muma, 1961 (Acarina, Parasitiformes) mit Bestimmungsschlusseln. Zool. Jahrb. Syst., 116(2): 199-216.
- Knapp M., Van Houten Y., Van Baal E., Groot T. 2018. Use of predatory mites in commercial biocontrol: current status and future prospects. Acarologia, 58 (Suppl):72-82. doi:10.24349/acarologia/20184275
- Kreiter S., Abo-Shnaf R.I.A. 2020a. Phytoseiid mites of Rodrigues Island. Acarologia, 60(2): 449-468. doi:10.24349/acarologia/20204376
- Kreiter S., Abo-Shnaf R.I.A. 2020b. New records of phytoseiid mites from Mauritius Island (Acari: Mesostigmata). Acarologia 60(3): 520-545. doi:10.24349/acarologia/20204382
- Kreiter S., Abo-Shnaf R.I.A., Payet R.-M. 2020a. Phytoseiid mites of Mayotte Island (Acari: Mesostigmata). Acarologia, 60(3): 622-642. doi:10.24349/acarologia/20204391
- Kreiter S., Amiri K., Douin M., Bohinc T., Trdan S., Tixier M.-S. 2020b. Phytoseiid mites of Slovenia (Acari: Mesostigmata): new records and first description of the male of Amblyseius microorientalis. Acarologia, 60(2): 203-242. doi:10.24349/acarologia/20204364
- Kreiter S., Bopp M.-C., Douin M., Nguyen D.T., Wyckhuys K. 2020c. Phytoseiidae of Vietnam with description of a new species. Acarologia, 60(1): 75-110. doi:10.24349/acarologia/20204362
- Kreiter S., Douin M., Tixier M.-S. 2021a. New records of phytoseiid mites (Acari: Mesostigmata) from Madeira Island. Acarologia 61(2): 217-240. doi:10.24349/acarologia/20214428
- Kreiter S., Fontaine O., Payet R.-M. 2018a. New records of Phytoseiidae (Acari: Mesostigmata) from Mauritius. Acarologia, 58(4): 773-785. doi:10.24349/acarologia/20184273
- Kreiter S., Mailloux J., Tixier M.-S., Le Bellec F., Douin M., Guichou S., Etienne J. 2013. New phytoseiid mites of the French West Indies, with description of a new species, and new records (Acari: Mesostigmata). Acarologia, 53(3): 285-303. doi:10.1051/acarologia/20132095
- Kreiter S., Moraes G.J.de 1997. Phytoseiidae mites (Acari: Phytoseiidae) from Guadeloupe and Martinique. Fla Entomol., 80(3): 376-382. doi:10.2307/3495770
- Kreiter S., Payet R.-M., Douin M., Fontaine O., Fillâtre J., Le Bellec F. 2020d. Phytoseiidae of La Réunion Island (Acari: Mesostigmata): three new species and two males described, new synonymies, and new records. Acarologia, 60(1): 111-195. doi:10.24349/acarologia/20204361
- Kreiter S., Payet R.-M., Abdou Azali H. 2021b. Phytoseiid mites of Anjouan Island (Acari: Mesostigmata). Acarologia, 61(1): 62-83. doi:10.24349/acarologia/20214418
- Kreiter S., Payet R.-M., Abdou Azali H. 2021c. Phytoseiid mites of Mohéli Island (Acari: Mesostigmata). Acarologia, 61(1): 94-114. doi:10.24349/acarologia/20214419
- Kreiter S., Payet R.-M., Fillâtre J., Abdou Azali H. 2018b. First records of Phytoseiidae from one island of the Comoros Archipelago. Acarologia, 58(3): 529-545. doi:10.24349/acarologia/20184256
- Kreiter S., Payet R.-M., Mouigni H., Douin M., Tixier M.-S., Abdou Azali H. 2021d. New records of phytoseiid mites (Acari: Mesostigmata) from Grande Comore Island (Comoros Archipelago). Acarologia, 61(2): 241-273. doi:10.24349/acarologia/20214429
- Kreiter S., Tixier M.-S., Etienne J. 2006. New records of phytoseiid mites from the French Antilles, with description of Neoseiulus cecileae sp. nov. Zootaxa, 1294: 1-27. doi:10.11646/zootaxa.1294.1.1
- Kreiter S., Zriki Z., Ryckewaert P., Pancarte C., Douin M., Tixier M.-S. 2018c. New phytoseiid mites of Martinique, with redescription of four species and new records. Acarologia, 58 (2): 366-407. doi:10.24349/acarologia/20184248
- Lindquist E.E. 1994. Some observations on the chaetotaxy of the caudal body region of gamasine mites (Acari: Mesostigmata), with a modified notation for some ventrolateral body setae. Acarologia, 35: 323-326.
- Lindquist E.E., Evans G.W. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Mem. Entomol. Soc. Canada, 47: 1-64. doi:10.4039/entm9747fv
- Lofego A.C., Moraes G.J. de, Castro L.A.S. 2004. Phytoseiid mites (Acari: Phytoseiidae) on Myrtaceae in State of São Paulo, Brazil. Zootaxa, 516: 1-18. doi:10.11646/zootaxa.516.1.1
- Mailloux J., Le Bellec F., Kreiter S., Tixier M.-S., Dubois P. 2010. Influence of ground cover management on diversity and density of phytoseiid mites (Acari: Phytoseiidae) in Guadeloupean citrus orchards. Exp. Appl. Acarol., 52: 275-290. doi:10.1007/s10493-010-9367-7
- McMurtry J.A., Croft B.A. 1997. Life-styles of phytoseiid mites and their roles in biological control. Ann. Rev. Entomol., 42: 291-321. doi:10.1146/annurev.ento.42.1.291
- McMurtry J.A., Moraes G.J. de, Sourassou N.F. 2013. Revision of the life styles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18: 297-320. doi:10.11158/saa.18.4.1
- Moraes G.J. de, Castro T.M.G. de, Kreiter S., Quilici S., Gondim Jr. M.G.C., Sá L.A. 2012. Search for natural enemies of Raoiella indica Hirst in Réunion Island (Indian Ocean). Acarologia, 52(2): 129-134. doi:10.1051/acarologia/20122043
- Moraes G.J. de, Kreiter S., Lofego A.C. 2000. Plant mites (Acari) of the French Antilles. 3. Phytoseiidae (Gamasida). Acarologia, 40(3): 237-264.
- Moraes G.J. de, McMurtry J.A., Denmark H.A. 1986. A catalog of the mite family Phytoseiidae. References to taxonomy, synonymy, distribution and habitat. EMBRAPA, Brasilia, Brazil, 353 pp.
- Moraes G.J. de, McMurtry J.A., Denmark H.A., Campos C.B. 2004. A revised catalog of the mite family Phytoseiidae. Zootaxa, 434: 1-494. doi:10.11646/zootaxa.434.1.1
- Moraes G.J. de, Mesa N.C. 1988. Mites of the family Phytoseiidae (Acari) in Colombia, with descriptions of three new species. Intern. J. Acarol., 14(2): 71-88. doi:10.1080/01647958808683790
- Moraes G.J. de, Mesa N.C., Braun A. 1991. Some phytoseiid mites of Latin America (Acari: Phytoseiidae). Intern. J. Acarol., 17(2): 117-139. doi:10.1080/01647959108683892
- Muma M.H. 1955. Phytoseiidae (Acarina) associated with citrus in Florida. Ann. Entomol. Soc. Amer., 48: 262-272. doi:10.1093/aesa/48.4.262
- Muma M.H. 1961. Subfamilies, genera, and species of Phytoseiidae (Acarina: Mesostigmata). Fla St. Mus. Bul., 5(7): 267-302.
- Muma M.H. 1962. New Phytoseiidae (Acarina: Mesostigmata) from Florida. Fla Entomol., 45: 1-10. doi:10.2307/3492897
- Muma M.H., Denmark H.A. 1970. Phytoseiidae of Florida. Arthropods of Florida and neighbouring land areas, 6. Florida Department of Agriculture and Consumer Services, Division of Plant Industry, Gainesville, USA, 150 pp.
- Murphy S.T. 1984. Biological control of the cassava green mite (Mononychellus spp.) in east Africa. Integrated Pest Management of Cassava Green Mite. Proceedings Regional Training Workshop in East Africa, 30 April - 4 May 1984. CIBC, Kenya: 55-61.
- Myers N. 1988. Threatened biotas: hostspots in tropical forests. Environmentalist, 8: 187-208. doi:10.1007/BF02240252
- Myers N., Mittermeier R.A., Mittermeier C.G., Da Fonseca G.A., Kent J. 2000. Biodiversity hotspots for conservation priorities. Nature, 403: 853-858. doi:10.1038/35002501
- Navia D., Domingos C.A., Mendonça R.S., Ferragut F., Rodrigues M.A.N., de Morais E.G.F., Tixier M.-S., Gondim Jr. M.G.C. 2014. Reproductive compatibility and genetic and morphometric variability among populations of the predatory mite, Amblyseius largoensis (Acari: Phytoseiidae), from Indian Ocean Islands and the Americas. Biol. Cont., 72: 17-29. doi:10.1016/j.biocontrol.2014.01.011
- Nuvoloni F.M., Lofego A.C., Rezende J.M., Feres R.J.F. 2015. Phytoseiidae mites associated with Hevea spp. From the Amazon region: a hidden diversity under the canopy of native trees. Syst. Biodiv., 13(2): 182-206. doi:10.1080/14772000.2014.985344
- Swirski E., Golan Y. 1967. On some phytoseiid mites (Acarina) from Luzon Island (Phillipines). Isr. J. Agric. Res., 17: 225-227.
- Toldi M., Freitas E.M. de, Silva V.L. da, Caumo M., Ferla J.J., Orlandi C.R., Costa T. Da, Johann L., Ferla N. J. 2021. Communities of predatory mites (Phytoseiidae and Stigmaeidae) in different environments of the Brazilian Pampa. Acarologia 61(1): 20¬30.
- Tseng Y.H. 1983. Further study on phytoseiid mites from Taiwan (Acarina: Mesostigmata). Chin. J. Entomol., 3: 33-74.
- Ueckermann E.A., Loots G.C. 1988. The African species of the subgenera Anthoseius De Leon and Amblyseius Berlese (Acari: Phytoseiidae). Entomol. Mem., Dep. Agric. Water Supply, Rep. South Africa 73, 168 pp.
- van der Merwe G.G. 1968. A taxonomic study of the family Phytoseiidae (Acari) in South Africa with contributions to the biology of two species. Entomol. Mem. South Africa Dep. Agric. Techn. Serv., 18: 1-198.
- Walter D.E., Krantz G.W. 2009. Collecting, rearing and preparing specimens. In: Krantz G.W., Walter D.E. (eds) A manual of acarology, 3rd ed. Texas Tech University Press, Lubbock. 807 pp.
- Wu W.N., Chou F.W. 1981. A new species of Amblyseius (Acarina: Phytoseiidae) from Guangdong Province. Zool. Res., 2: 273-274 [in Chinese].
- Zacarias M.S., Moraes G.J. de. 2001. Phytoseiid mites (Acari) associated with rubber trees and other euphorbiaceous plants in southeastern Brazil. Neotrop. Entomol., 30(4): 579-586. doi:10.1590/S1519-566X2001000400011



2021-04-07
Date accepted:
2021-05-01
Date published:
2021-05-18
Edited by:
Tixier, Marie-Stéphane

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Kreiter, Serge and Douin, Martial
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



