Kinetics of Dermatophagoides pteronyssinus and Dermatophagoides farinae growth and an analysis of the allergen expression in semi-synthetic culture medium
Rodríguez, David1 ; Palacios, Ricardo2 ; Martínez, Jorge3 ; Guisantes, Jorge A.4 and Postigo, Idoia5
1DIATER Laboratories S. A. Av. Gregorio Peces Barba, 2. Parque Tecnológico Leganes, 28919-Leganés. Madrid. Spain.
2DIATER Laboratories S. A. Av. Gregorio Peces Barba, 2. Parque Tecnológico Leganes, 28919-Leganés. Madrid. Spain.
3Department of Immunology, Microbiology and Parasitology. Faculty of Pharmacy. Paseo de la Universidad, 7. 01006 - Vitoria. Spain.
4Department of Immunology, Microbiology and Parasitology. Faculty of Pharmacy. Paseo de la Universidad, 7. 01006 - Vitoria. Spain.
5✉ Department of Immunology, Microbiology and Parasitology. Faculty of Pharmacy. Paseo de la Universidad, 7. 01006 - Vitoria. Spain.
2021 - Volume: 61 Issue: 2 pages: 403-411
https://doi.org/10.24349/acarologia/20214439Original research
Keywords
Abstract
Introduction
House dust mite allergens are a major risk parameter for allergies worldwide (Eraso et al. 1998). Dermatophagoides pteronyssinus (Trouessart, 1897) and Dermatophagoides farinae (Hughes, 1961) represent more than 90% of the mites found in house dust samples worldwide and are the main cause of respiratory allergies (Weghofer et al. 2008). Voorhorst et al. (1969) observed that these mites could be grown in the laboratory using artificial culture media. Many studies have been conducted to determine the most favourable culture medium to produce extracts for allergy testing (Eraso et al. 1997; Eraso et al. 1998; Cardona et al. 2004).
Manufactured allergenic extracts from house dust mite (HDM) cultures, available on the market to be employed for diagnosis and immunotherapy, are heterogeneous in terms of allergen concentrations and composition (Valenta et al. 2018; González-Pérez et al. 2019).
Clearly, the variability in the allergen composition, allergen concentration and culture medium composition are among the most important factors consider when determining regulatory requirements.
Several studies have demonstrated the importance of the composition of the culture media in obtaining consistent and high-quality mite extracts in terms of the content of relevant allergens. Vidal-Quist et al. (2017) demonstrated that the use of culture media containing human epidermal derivatives decreases the content of major allergens. However, Batard et al. (2006) demonstrated that mite extracts produced from cultures based on wheat germ, yeast and amino acids under pharmaceutical conditions exhibited all the relevant mite allergens. Avula-Poola et al. (2012) also demonstrated that the mite diet influences the dynamics of the production of relevant allergens. These researchers obtained good rates of growth with cultures based on rodent chow/yeast and egg/yeast, but the inclusion of animal or egg proteins in the diet limits the pharmaceutical use of these extracts.
Moreover, to improve the standardization of allergenic extracts for diagnosis and immunotherapy, it is important to determine the concentration and frequency of occurrence of the components, including both the major and the minor allergens, during the growth period of the mite population (Eraso et al. 1998) to prepare the mite culture extract during the most appropriate time point during the growth period. Cardona et al. (2004) and Eraso et al. (1997) demonstrated that laboratory cultures of Dermatophagoides and Blomia using rodent chow/yeast medium exhibited three well-differentiated growth phases and that the quality of the allergenic extract production depended on the kinetics of allergen expression.
In this study, house dust mites D. pteronyssinus and D. farinae laboratory cultures were used to study the mite population dynamics in a semi-synthetic culture medium made with non-animal origin components to ensure consistency with the pharmaceutical rules. The kinetics of allergen expression during the mite growth cycle were analysed to optimize the quality of the allergenic extracts.
Material and methods
Mite Cultures
Dermatophagoides pteronyssinus and D. farinae (Laboratory of Parasitology, Department of Immunology, Microbiology and Parasitology, University of the Basque Country, Vitoria, Spain) were cultured on a semi-synthetic culture medium containing a 1:1 (wt:wt) autoclaved mixture of rolled oats (Laboratorios YNSADIET, Leganés, Madrid)/dried yeast powder (Vigor®, Santiveri, Barcelona) and a mixture of three amino acids: 4.5 mg of glycine (Sigma G-2879 /gr of medium, 7.0 mg of arginine (Sigma A-5131)/gr of medium and 8.3 mg of lysine (Sigma L-5751)/gr of medium.
Thirty-six cultures per species (12 in triplicate) were prepared simultaneously by inoculating with 3,600 mites in 10 g of medium and subsequent incubation at 24-26 °C and 75-80% relative humidity, as in Cardona et al. (2005).
Each species was stored in a separated incubator, and cultures were examined regularly and separately to assess mite growth. The identification of mites was carried out using the keys developed by Collof and Spieksma (1992).
At two-week intervals, a small culture sample (50 mg) was used to determine the mite concentration (living and dead mites) by stereoscopic microscope counting. After that step, cultures were frozen for the production of the allergenic extracts (Cardona et al. 2004).
Extracts Preparation
Extraction from each whole culture containing mites was performed by shaking for 4 hours at 4 °C in phosphate buffered saline (PBS), pH 7.2 (1 g of raw material/10 ml of buffer). The extract suspension was clarified by centrifugation at 5,000 rpm for 30 min, and the supernatant was filtered through Whatman N °1 and sterilizing filters (Millipore, Bedford. USA). The extracts were dialyzed against distilled water at 4 °C for 24 hours using membranes with a 5,000 Da cut-off point. Dialyzed material was centrifuged at 10,000 rpm for 30 min at 4 °C, and the supernatant was finally freeze-dried (Cardona et al. 2005).
An extract from the culture medium without mites was also obtained according to the same protocol. The standardization of extracts was carried out according to the recommendations of the European Academy of Allergy and Clinical Immunology (Dreborg et al. 1993).
The bicinchoninic acid (Lowry et al. 1951) method was employed to determine the protein percentages in the extracts obtained from D. pteronyssinus and D. farinae cultures at different time periods.
SDS-PAGE and SDS-PAGE-Immunoblotting
Protein separation was performed by polyacrylamide gel electrophoresis in the presence of sodium dodecyl-sulfate (SDS-PAGE) using polyacrylamide concentrations of 12% and 4% for separating and stacking gels according to Laemmli (1970), as modified by Eraso et al. (1997). A protein concentration of 8 mg/ml was loaded to each lane.
Separated protein bands were analysed by Coomassie Blue staining or transferred to polyvinyl difluoride membranes (Immuno-Blot PVDF Membrane, Bio-Rad, 162-0174) using a horizontal semi-dry transfer system (Towbin et al. 1979).
The PVDF membranes were incubated overnight (4 °C with agitation) with pooled human serum (5 sera pooled 1:1 with a RAST class ≥ 2) from patients sensitized to house dust
mites (National Register of Biobank Serum Collections, code C.0002774; Instituto de Salud Carlos III, Ministry of Economy and Competitiveness/Lascaray Research Center, University of the Basque Country, Vitoria, Spain). Bound IgE antibodies were detected using HRP-conjugated goat anti-human IgE and an ECL-Western blotting kit (Amersham ECL Plus Western Blotting Detection System, GE Healthcare UK Ltd, Buckinghamshire, UK) (Kyhse-Andersen et al. 1984, Shen et al. 1988).
The results were recorded in a Molecular Imager ChemiDoc XRS System (Bio-Rad).
Quantification of major allergens
Enzyme-Linked ImmunoSorbent Assay (ELISA) tests were applied to measure the concentrations of the major allergens Der p 1, Der p 2, Der f 1 and Der f 2 in the D. pteronyssinus and D. farinae culture extracts obtained at various time points following the manufacturer's instructions, as described by Eraso et al. (1996). The ELISA kits employed in this study were Der p 1 (5H8/4C1), Der f 1 (6A8/4C1) and Mite Group 2 (1D8/7A1) (INDOOR Biotechnologies, Ltd., Manchester UK).
Results
Kinetics of mite growth



Figure 1 shows the kinetics of D. pteronyssinus and D. farinae growth in the culture medium employed, expressed as the number of living or dead mites per gram of culture at different weeks of growth. Both species of Dermatophagoides showed an excellent rate of growth in the culture medium, reaching maximum values in week 20 for D. pteronyssinus and in week 14 for D. farinae.
The results obtained showed three different growth phases: the latency phase (weeks 0-10 in both Dermatophagoides cultures); the exponential growth phase (weeks 10-20 in D. pteronyssinus cultures and weeks 10-14 in D. farinae cultures); and a final phase of death (weeks 20-28 and 14-18 in D. pteronyssinus and D. farinae cultures, respectively).
Dermatophagoides pteronyssinus reached its maximum growth at weeks 18-22, with maximum differences between living and death mites at weeks 16-18. Only after week 24 did the ratio of living mites/death mites become < 1.
Dermatophagoides farinae reached its maximum growth at week 14 with maximum differences between living and death mites at week 14. This species had a living mite/death mite ratio < 1 after week 16.
The maximum protein content analysed from the extracts at different times of culture incubation was observed after 24 weeks for D. pteronyssinus (44%) and after 16 weeks for D. farinae (38%). The yield of cultures according to the ratio of mg of extract/g of culture media was 72 and 74 mg extract/g culture media for D. peronyssinus and D. farinae, respectively, when they reached their maximum growth.
The extract produced by the culture medium without mites exhibited 67 mg/g dry weight and 25% protein content.
Extracts characterization
Analysis of IgE binding components by immunoblotting
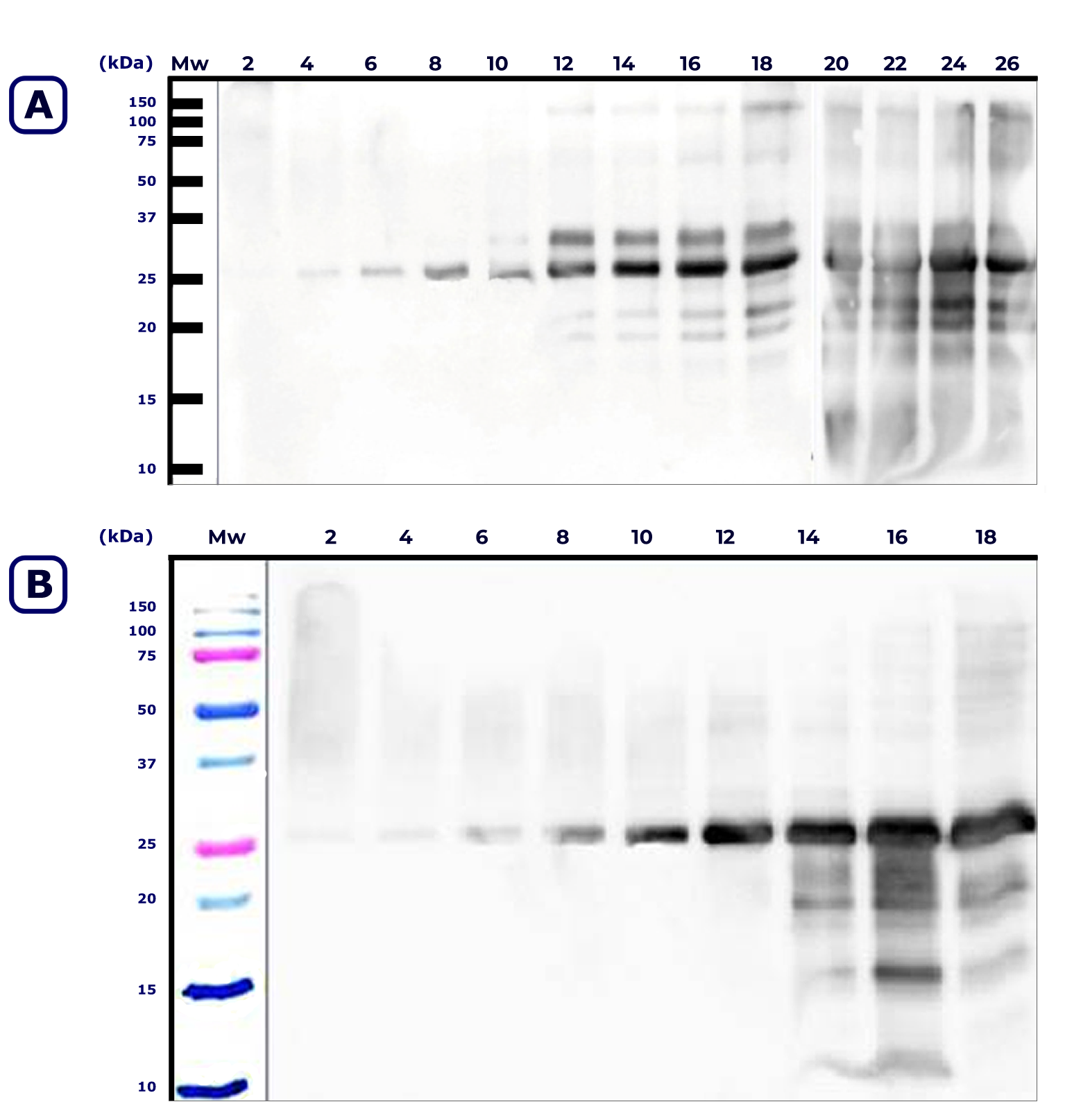


Figure 2 shows SDS-PAGE IgE immunoblotting results of D. pteronyssinus and D. farinae culture extracts at different times. The results revealed the presence of IgE binding components after the fourth week in both species. The first IgE binding component most likely expressed was the major allergen Der 1 (25-27 kDa).
The number of IgE binding components increased according to the incubation time, reaching a maximum at week 20 for D. pteronyssinus and at week 16 for D. farinae.
Quantification of major allergens
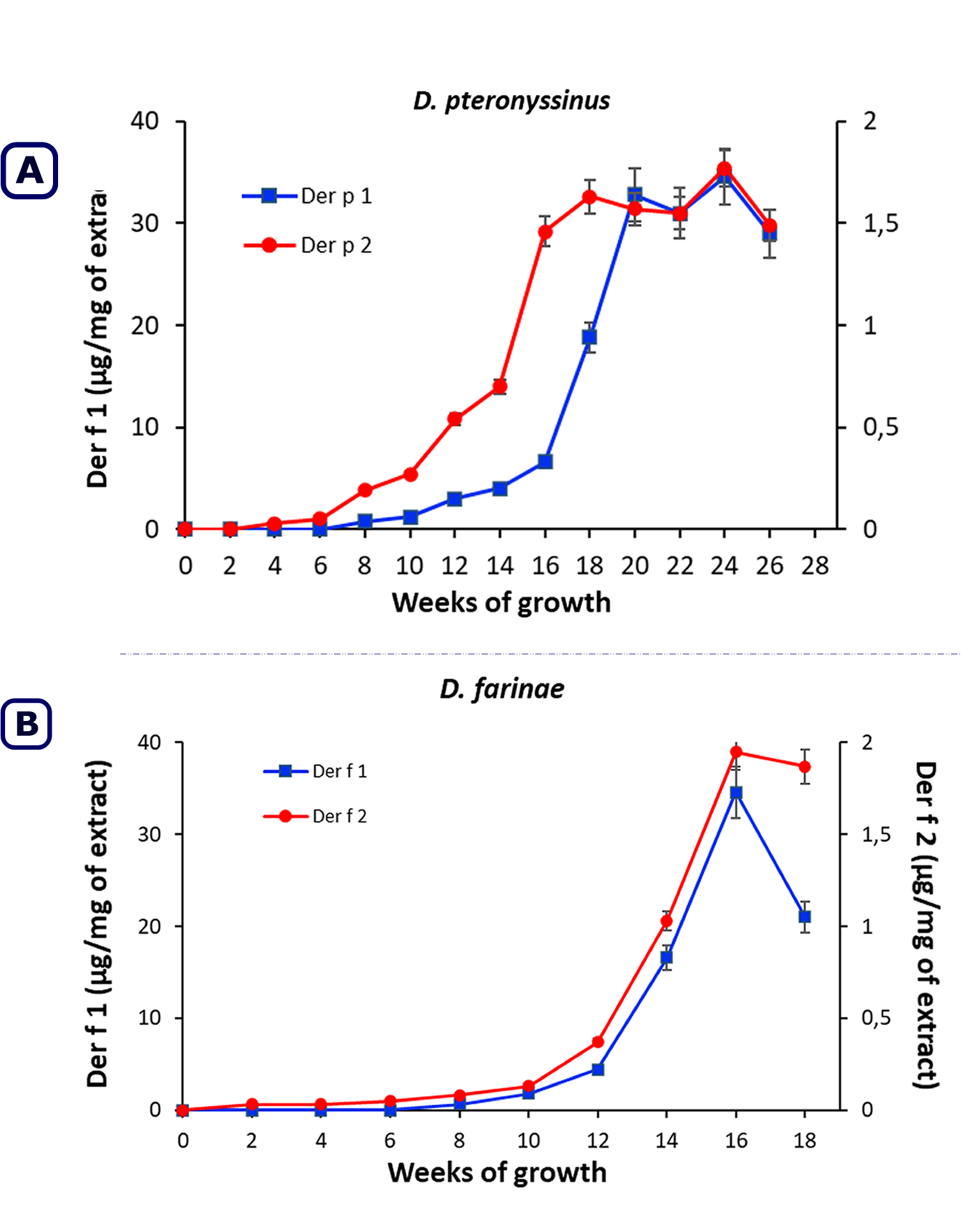


Figure 3 shows the concentration of the major allergens Der p 1, Der p 2, Der f 1 and Der f 2 in D. pteronyssinus and D. farinae culture extracts at different times of growth (weeks). The results of the ELISA tests showed high expression of the major allergens (Der p 1, Der p 2, Der f 1 and Der f 2) at the last phases of mite growth, matching the major protein content of the extracts. The maximum allergen concentration was observed at week 24 for Der p 1 (35.8 µg/mg) and Der p 2 (1.8 µg/mg) in D. pteronyssinus extracts and at week 16 for Der f 1 (34.8 µg/mg) and for Der f 2 (2.0 µg/mg) in D. farinae extracts.
The ratio mg of extract/g culture medium of the mite cultures at their maximum growth did not show statistically significant differences when compared with the extract obtained from the culture medium. When the percentage of protein was compared (controls vs. mite cultures), statistically significant differences were only observed in D. pteronyssinus at week 24 at the beginning of the drop phase (p=0.01).
Discussion
Since the study by Voorhorst et al. (1969), who observed that HDM could grow in the laboratory using artificial culture media, mite cultures have been the main key for producing raw material to elaborate allergenic extracts for use in clinical applications.
Currently, mite allergenic extracts are the basis for producing raw material to obtain key reagents for diagnosing and treating IgE-mediated allergies. Until two decades ago, the opinion of experts and the production of allergenic extracts under Good Manufacturing Practices (GMP) regulations was sufficient to use products for ''in vivo'' diagnosis and immunotherapy in type I allergies. Currently, the situation has changed significantly, and countries increasingly only allow the use of this type of product under strict regulations, which require safety and efficacy to be demonstrated, strict Good Manufacturing Practice regulations to be followed and consistent studies of quality to be performed.
Regarding the material referred to as the mites, it is essential to determine the relevant allergens and their concentrations, as well as establishing the culture conditions and the type of material that is going to be used as a raw material to obtain the intermediate product (Carnés et al. 2017; Valenta et al. 2018).
Although the available data suggest that molecular TIA strategies have many advantages over TIA based on allergen extracts (Zhernov et al. 2019) at present, the natural allergen extracts are still the basis for obtaining the biological products to be used in the treatment of type I allergies (immunotherapy). From the classic culture medium containing dermic derivatives (Vidal-Quist et al. 2017) to the use of semi-synthetic media (Batard et al. 2006), several studies have attempted to determine the most favourable culture medium to produce extracts for allergy testing (Avula-Poola et al. 2012; Batard et al. 2006; Eraso et al.1997; Eraso et al. 1998; Cardona et al. 2004; Vidal-Quist et al. 2017). Thus, manufactured allergenic extracts from HDM cultures, available in the market for diagnosis and immunotherapy, are heterogeneous in terms of allergen concentrations and composition. Therefore, the use of more strict in-house references and the standardization of culture processes can be justified as means of obtaining better material for human applications (Casset et al. 2012; González-Pérez et al. 2019).
To improve the standardization of allergenic extracts for diagnosis and immunotherapy, it is important to determine the frequency and concentration of the components, both the major and the minor allergens, at different times of mite growth (Eraso et al. 1998) for processing the mite culture during the most appropriate growth time. Previously, Eraso et al. (1997); Martinez et al. (2000), and Cardona et al. (2004), demonstrated that laboratory cultures of Dermatophagoides and Blomia using rodent chow/yeast medium exhibited three well-differentiated growth phases and that the quality of the allergenic extract production depended on the kinetics of allergen expression.
The results obtained in this work showed similar growth behaviour of both mite species found by other authors (Cardona et al. 2004; Eraso et al. 1997; Eraso et al. 1998; Martínez et al. 2000) using conventional culture media and the same methodology. The main differences between both species were observed in the time taken to reach the maximum number of living mites per gram of culture medium; this period was 20 weeks for D. pteronyssinus and 14 weeks for D. farinae.
Similarly, the results of this study demonstrated that variation in the percentage of protein during mite growth does not constitute a valid parameter for follow-up study of the allergen expression. The comparison of the growth curves obtained in this work with those obtained by Eraso et al. (1997) using conventional diet showed that the main difference is observed in the amount of mite counts during some steps of the curve. Maximum values of mites were 2 times higher for D. pteronyssinus and 5 times higher for D. farinae with semi-synthetic medium than with conventional diet. Immunoblotting results showed an active change in the protein profiles. The most relevant changes in allergenic diversity began at the end of the exponential growth phase. Later, during the drop phase, the detection of IgE binding was observed to increase in both components and band intensity.
The comparison of the immunoblotting results obtained in this work with those obtained by Eraso et al. (1997) using conventional diet showed more IgE binding components with wider molecular weight range and better results regarding allergenic variability with semi-synthetic media than with conventional media. Higher allergen concentrations for both species, occurred during the final growth phase (death): week 18 for Der p2, week 20 for Der p2 and week 16 for Der f 1 and Der f 2.
The kinetics of the Der p 1 major allergen in D. pteronyssinus cultures reached its maximum concentration in week 20, which was when the number of living mites and the total number of mites were maximal. After this week, the Der p1 concentration remained equal to or greater than mite concentrations (week 24), even though the number of dead mites was increasing. The major allergen Der p 2 showed a similar pattern, but its highest concentration was achieved at week 18, which was before the mite growth reached its maximum. After that step, mite cultures showed equal or greater Der p 2 concentrations (week 22, week 24). The well-defined semi-synthetic medium used in this study enables the most important allergenic HDM species to grow in a reproducible way. The extracts obtained contain the most relevant allergens for desensitization purposes and in vivo diagnosis and, they are consistent with the current regulations of health authorities.
Some authors have demonstrated the negative impact of some steps in the mite culture process for the production of allergenic extracts. Avula-Poola et al. (2012) indicated that mite washing resulted in the loss of up to 88% of the allergens in the extracts produced. Santos da Silva et al. (2019) demonstrated the clear impact of inbreeding on allergen content and concluded that the quantification and/or identification of allergens from in-bred lines should be routinely considered for mite cultivation in order to select breeds with higher amounts of major allergens. Finally, we have observed that in a semi-synthetic culture medium the maximum allergen concentrations do not always agree with the optimal growth phase of living mites. The results demonstrate that the mite-culturing diet directly affects population growth and these results are in accordance with those of Avula-Poola et al. (2012), Batard et al. (2006) and Vidal Quist et al. (2017). The dynamics of individualized allergen expression and concentrations during the growth curve, the culture media composition and the follow-up investigation of the growth conditions are highly influential parameters in ensuring the quality and consistency of mite extracts used for pharmaceuticals.
In conclusion, the use of semi-synthetic culture media without components of animal origin and the follow-up study of the mite population growth should be considered key points in optimizing the reproducibility of the production conditions and reducing batch-to-batch variability, enabling products to be more consistent with the regulations of health authorities.
Acknowledgments
This work was supported by the Basque Country Government (Consolidated Research Groups of the Basque University Research System: grant IT-1043-16).
References
- Avula-Poola S., Morgan M.S., Arlian L.G. 2012. Diet influences growth rates and allergen and endotoxin contents of cultured Dermatophagoides farinae and Dermatophagoides pteronyssinus house dust mites. Int. Arch. Allergy Immunol.159: 226-34 doi:10.1159/000336026
- Batard T., Hrabina A., Bi X.Z., Chabre H., Lemoine P., Couret M.N., Faccenda D., Villet B., Harzic P., André F., Goh S.Y., André C., Chew F.T., Moingeon P. 2006. Production and proteomic characterization of pharmaceutical-grade Dermatophagoides pteronyssinus and Dermatophagoides farinae extracts for allergy vaccines. Int. Arch. Allergy Immunol. 140: 295-305. doi:10.1159/000093707
- Cardona G., Eraso E., Serna L.A., Guisantes J.A., Martínez J. 2004. Analysis of the allergen expression of Blomia tropicalis (van Bronswijk, Cock and Oshima) and Blomia kulagini (Zakhvatkin) (Astigmata: Glycyphagidae) cultures. J. Med. Entomol. 41:1068-1072. doi:10.1603/0022-2585-41.6.1068
- Cardona G., Guisantes J., Postigo I., Eraso E., Serna L.A., Martínez J. 2005. Allergenic cross-reactivity between Blomia tropicalis and Blomia kulagini (Acari: Echymiopodidae) extracts from optimizad mite cultures. J. Invest. Allergol. Clin. Immunol.15: 259-265.
- Carnés J., Iraola V., Cho S.H., Esch R.E. 2017. Mite allergen extracts and clinical practice. Ann. Allergy Asthma Immunol. 118: 249-256. doi:10.1016/j.anai.2016.08.018
- Casset A., Mari A., Purohit A., Resch Y., Weghofer M., Ferrara R., Thomas W.R., Alessandri C., Chen K.W., de Blay F., Valenta R., Vrtala S. 2012. Varying allergen composition and content affects the in vivo allergenic activity of commercial Dermatophagoides pteronyssinus extracts. Int. Arch. Allergy Immunol. 159: 253-62. doi:10.1159/000337654
- Colloff M.J., Spieksma F.T.M. 1992. Pictorial keys for the identification of domestic mites. Clin. Exp. Allergy. 22: 823-830. doi:10.1111/j.1365-2222.1992.tb02826.x
- Dreborg S., Frew A. 1993. Allergen standardization and skin test. Allergy. 14: 63-70. doi:10.1111/j.1398-9995.1993.tb04759.x
- Eraso E. 1996. Estudio de la expresión de los componentes alergénicos en cultivos de ácaros del género Dermatophagoides Bogdanov, 1864. Consideraciones de tipo diagnóstico. [Phd Thesis] Vitoria: University of The Basque Country.
- Eraso E., Guisantes J.A., Martínez J., Sáenz de Santamaría M., Martínez A., Palacios R., Cisterna R. 1997. Kinetics of allergen expression in cultures of house dust mites, Dermatophagoides pteronyssinus and Dermatophagoides farinae (Acari: Pyroglyphidae). J. Med. Entomol. 34: 684-689. doi:10.1093/jmedent/34.6.684
- Eraso E., Martínez J., García-Ortega P., Martínez A., Palacios R., Cisterna R, Guisantes J.A. 1998. Influence of mite growth culture phases on the biological standardization of allergenic extracts. J. Investig. Allergol. Clin. Immunol. 8: 201-206.
- González-Pérez R., Poza-Guedes P., Barrios Del Pino Y., Matheu V., Sánchez-Machín I. 2019. Evaluation of major mite allergens from European standardized commercial extracts for in vivo diagnosis: addressing the need for precision medicine. Cli. Transl. Allergy. 9:14. doi:10.1186/s13601-019-0254-y
- Kyhse-Andersen J. 1984. Electroblotting of multiple gels: a simple apparatus without buffer tank for rapid transfer of proteins from polyacrylamide to nitrocellulose. J. Biochem. Biophys. Methods. 10: 203-209. doi:10.1016/0165-022X(84)90040-X
- Laemmli U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 277: 680-685. doi:10.1038/227680a0
- Lowry O.H., Rosembrough N.J., Farr A.L., Randall R.J. 1951. Protein measurement with the Folin-Phenol reagent. Journal of Biological Chemistry. 193: 265-275. doi:10.1016/S0021-9258(19)52451-6
- Martínez J., Eraso E., Palacios R., Guisantes J.A. 2000. Cross-reactions between Dermatophagoides pteronyssinus and Dermatophagoides farinae (Acari: Pyroglyphidae) related to the different growth phases of cultures. J. Med. Entomol. 37: 35-39. doi:10.1603/0022-2585-37.1.35
- Santos da Silva E., Marques Ponte J.C., Barbosa da Silva M., Silva Pinheiro C., Carvalho Pacheco L.G., Ferreira F., Briza P., Alcantara-Neves N.M.. 2019. Proteomic Analysis Reveals Allergen Variability among Breeds of the Dust Mite Blomia tropicalis. Int Arch Allergy Immunol. 27:1-14. doi:10.1159/000501964
- Shen H.D., Wang S.R., Tang R.B., Chang F.Z.N., Su S.N., Han S.N. 1988. Identification of allergens and antigens of Bermuda grass pollen by immunoblot analysis. Clin. Exp. Allergy.18: 401-410. doi:10.1111/j.1365-2222.1988.tb02888.x
- Towbin H., Stahelin I., Gordon J. 1979. Electrophoretic transfer of proteins from polyacrilamide gels to nitrocellulose sheets: procedure and some applications. P. Natl. Acad. Sci. USA.76: 4350-4354. doi:10.1073/pnas.76.9.4350
- Valenta R., Karaulov A., Niederberger V., Zernov Y., Elusiutina O., Campana R., Focke-Tejkl M., Curin M., Namazova-Baranova L., Wang J.Y., Pawankar R., Khaitov M. 2018. Allergen extracts for in vivo diagnosis and treatment of allergy: Is there a future? J. Allergy Clin. Immunol. Prac. 6(6):1845-1855. doi:10.1016/j.jaip.2018.08.032
- Vidal-Quist J.C., Ortego F., Rombauts S., Castañera P., Hernández-Crespo P. 2017. Dietary shifts have consequences for the repertoire of allergens produced by the European house dust mite. Med. Vet. Entomol. 31(3): 272-280. doi:10.1111/mve.12234
- Voorhorst R., Spieksma F.Th.M, Varekamp H. 1969. House dust atopy and the house dust mite Dermatophagoides pteronyssinus (Trouessart, 1897). Leiden: Stafleu's Scientific Publishing Company
- Weghofer M., Thomas W.R., Kronqvist M., Mari A., Purohit A., Pauli G., Horak F., Grönlund H., van Hage M., Valenta R., Vrtala S. 2008. Variability of IgE reactivity profiles among European mite allergic patients. Eur. J. Clin. Invest. 38: 959-965. doi:10.1111/j.1365-2362.2008.02048.x
- Zhernov Y, Curin M, Khaitov M, Karaulov A, Valenta R. 2019. Recombinant allergens for immunotherapy: state of the art. Curr Opin Allergy Clin Immunol. 19(4): 402-414. doi:10.1097/ACI.0000000000000536



2020-07-10
Date accepted:
2021-04-30
Date published:
2021-05-06
Edited by:
Roy, Lise

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Rodríguez, David; Palacios, Ricardo; Martínez, Jorge; Guisantes, Jorge A. and Postigo, Idoia
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



