A new species of Brachychthoniidae (Acari: Oribatida) from the Eastern Central Alps (Austria, Tyrol), with the proposal of a new genus
Schatz, Heinrich1
1✉ c/o Institute of Zoology, University of Innsbruck, Technikerstrasse 25, A-6020 Innsbruck, Austria.
2021 - Volume: 61 Issue: 2 pages: 365-379
https://doi.org/10.24349/acarologia/20214436ZooBank LSID: CF487C47-F525-4346-9CDC-95DFCC06AD8B
Original research
Keywords
Abstract
Introduction
Prior to the present study 174 species of the oribatid family Brachychthoniidae Thor, 1934 (including 12 spp. inquir., Schatz et al. 2011, updated) were described and grouped into 11 genera using morphological characters of the adult. Brachychthoniidae occur in all terrestrial zoogeographic regions. About two third of all species are recorded from the Holarctic region (Subías 2004, electronic update 2020), with 39 species known from Austria (Krisper et al. 2017). They are common inhabitants of forest litter and moss, but occur also in xeric grassland or in tundra with lichens, as well as in raw humus and soil, and in some localities, they were found with considerable abundances (Schweizer 1956, Moritz 1976a, b, Leonov 2020). Brachychthoniidae are among the smallest oribatid mites, the length of most adults varies between 120-250 µm. They are frequently overlooked and apparently underrepresented in several faunistic and ecological investigations.
The late Konrad Thaler, Innsbruck, forwarded me collections of oribatid mites from high mountains of the Eastern Alps, in particular from the summit of the Festkogel mountain. This material contains numerous remarkable findings which will be discussed elsewhere (Schatz in prep.). In the present paper a new species is presented and described. Its generic position is discussed, which leads to the conclusion that this species represents a new genus.
The Festkogel (3035 m a.s.l.), situated southwest of the locality Obergurgl, is the highest summit on the mountain ridge that descends from the Granatenkogel, between the Gaisberg valley and Ferwall valley. The summit area is on the upper edge of the alpine grassland belt (mainly formed by loose stands of Carex curvula) with the beginning small cushions of subnival vegetation (e.g. Androsace, Cerastium), lichens and mosses.
Material and methods
The specimens were mounted in lactic acid on temporary cavity slides for illustration, and subsequently preserved in ethanol. Drawings were made with a camera lucida attached to a transmission light microscope ''Nikon Eclipse E400''. Each parameter was measured from the particular optimal parallax free perspective. Measurements of total body length was done in lateral view, from tip of the rostrum to the posterior edge of the notogaster, body width as maximal width of notogaster from dorsal view. Measurements indicated are means, with range in parentheses; all measurements are given in micrometers (μm). The terminology of morphological features used in this paper follows that of Moritz (1976a) and Grandjean (summarized by Travé and Vachon 1975, Travé et al. 1996, Norton and Behan-Pelletier 2009). Since the new genus is currently monotypic, its description also serves as the diagnosis of the type species.
Following abbreviations are used: L—length; W—width; RO—rostrum; NA, NM, NP—anterior, median, posterior notogastral sclerites; AD, AN, PR—adanal, anal, peranal sclerites; SpC, SpD, SpE, SpF—suprapleural sclerites; (SpC)—indistinct part of suprapleural sclerite SpC; PA, PP—anterior, posterior pleural sclerites; ro, le, il, bo, exa—rostral, lamellar, interlamellar, bothridial, anterior exobothridial seta; exp—posterior exobothridial porus; c1-3 , d1-3 , e1 , e2 , f1 , f2 , h1- 3, p1-3 — notogastral setae; ia, im, ih, ip, ips—notogastral lyrifissures; mf—muscle fibres; 1a-c, 2a, 3a-c, 4a-d— epimeral setae; g1 , g7 —genital setae; ag—aggenital seta; ad1-3 — adanal setae; an1-2 —anal setae; pe—peranal seta; SC—subcapitulum; ru—rutellum; h, m, ma, a—subcapitular setae; or—adoral seta; MD—movable digit of chelicera; cha—cheliceral seta; ω, d, l, v, cm, acm, u, su—solenidion and setae on palp; σ, σ1, σ2, φ, ω, ω1 , ω2, ω3 —solenidia on genu, tibia, tarsus; dσ, dσ1 , dσ2 , dφ—coupled seta d on solenidion; ε—famulus on tarsus of leg I; ft, pl, tc, it, p, u, a, s, pv, d, l, v—setae on legs I–IV.
Taxonomy
Arcochthonius n. gen.
ZOOBANK: DE56E962-A279-49E4-AC76-FC4DC12FC679 ![]()
Type species: Arcochthonius roynortoni n. sp.
Diagnosis — Adults unique among known Brachychthoniidae (Thor, 1934) by following combination of character states: prodorsum and notogaster with sclerotized plates and ridges, rostral apex and lateral edge of prodorsum with teeth, anterior edges of medial and posterior notogastral sclerites with transverse thickenings, spanned by arched clamp-like ridges, notogastral seta d2 situated close to d1 , far from lateral notogaster margin, seta c3 inserting on distinct embossed plate on anterior notogastral sclerite NA, suprapleural sclerite SpE distinct, lateral to anterior transverse scissure, other suprapleural sclerites absent, suprapleural incision distinct, anterior and posterior pleural sclerites separated, epimeres IV with 4 pairs of setae, genital sclerites with 7 pairs of setae, aggenital sclerites present, adanal sclerites posteriorly fused, adanal seta ad2 hypertrophic, femur IV with 2 setae, genua III and IV each with 2 setae, tarsal setal formulas I–IV 18–14–13–11.
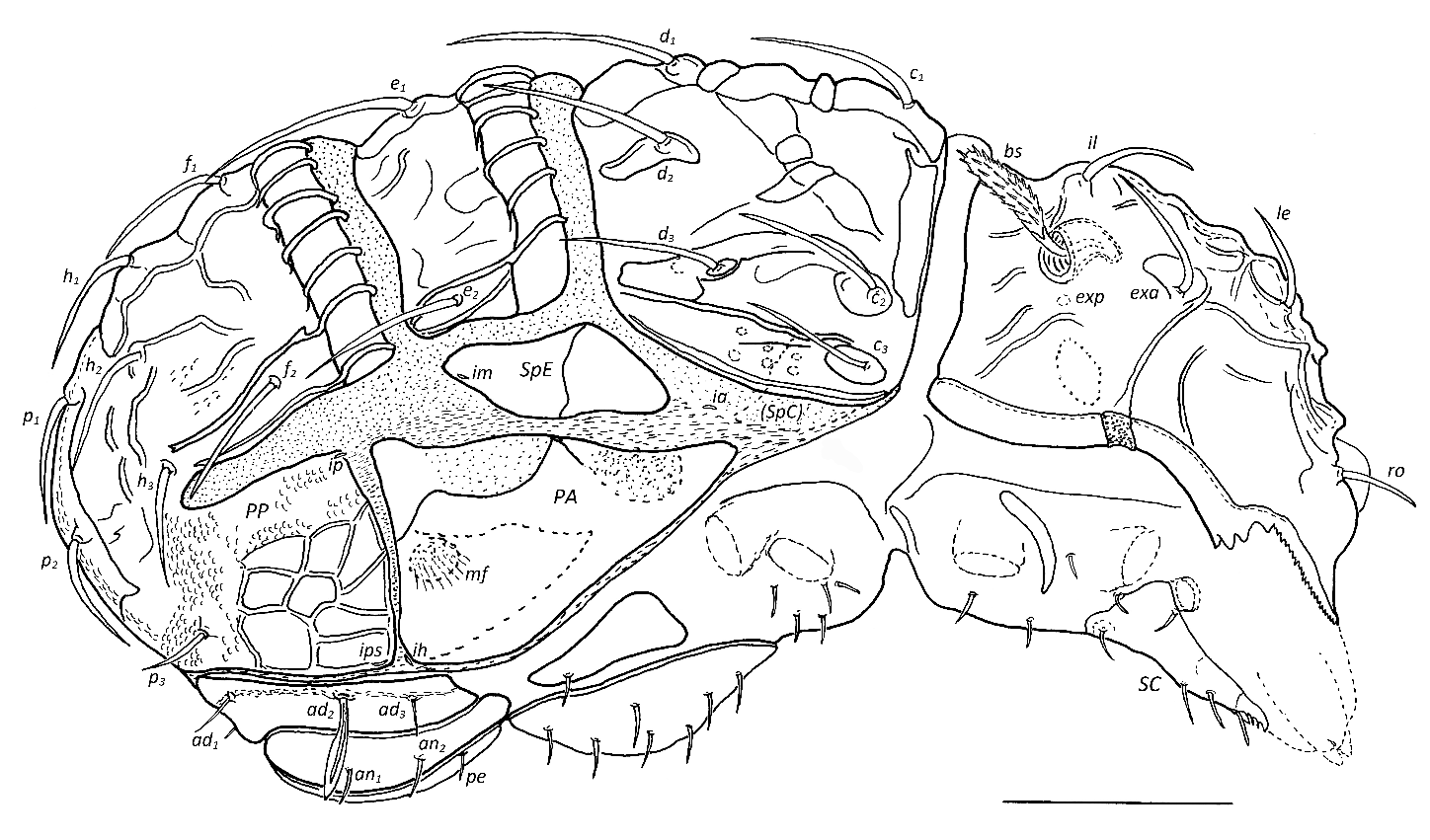
Description of adult — Enarthronote oribatid mite, placed in the family Brachychthoniidae. Body in lateral view arched, prodorsal and notogastral sclerites thickened and sculptured with plates and ridges. Prodorsum strongly elevated, anterior edge bent ventrad. Prodorsal surface with transverse and longitudinal ridges. Rostral apex with three teeth. Lateral edge of prodorsum serrated sawtooth-like, with 3–4 larger teeth posterior to serration. Five pairs of prodorsal setae (rostral, lamellar, interlamellar, anterior exobothridial, bothridial seta). Exobothridial hump protruding, posterior exobothridial seta represented by small pore. Bothridial seta small, with numerous small bristles. Notogaster elevated, divided in three separated sclerites by two broad transverse scissures. Anterior edges of notogastral sclerites NM and NP with distinct transverse thickenings, spanned by arched clamp-like ridges. All sclerites with larger and smaller plate-like ridges. Marginal suture on notogastral sclerite NA absent. 16 pairs of notogastral setae, c1 , c2 , c3 , d1 , d2 , d3 , e1 , e2 , f1 , and h1 inserting on tubercles on distinct plates, f2 , h2 , h3 , p1–p3 inserting on small tubercles on NP. Pygidium strongly arched caudally. Pygidial and pleural sclerites ventrally surrounded by disjugal suture. Pygidial sclerite with deep suprapleural incision, separating posterior pleural sclerite laterally about halfway from pygidium. Suprapleural sclerite SpE distinct, SpC partially fused with notogastral sclerite NA, visible part indistinctly protruding caudad. Sclerites SpD and SpF absent. Anterior and posterior pleural sclerites separated by distinct furrow. Lyrifissures ia – ips in pleural region. Subcapitulum anarthric, with 4 pairs of setae and 1 pair adoral setae. Rutellum atelobasic. Chelicera of chelate type, with one seta. Palp setal formula (trochanter to tarsus, solenidion in parentheses): 0–2–1–3–10(1), (u) fused and forked eupathidium on palptarsus, solenidion on palptarsus originating near base of seta cm. Epimeres I and II fused medially, separated from III and IV by sejugal furrow. Epimeres III and IV separated medially. Epimeral setal formula 3–1–3–4. Genital sclerites with 7 pairs of setae, aggenital sclerites undivided with 1 pair of setae, peranal sclerite narrow with 1 seta each, anal sclerites with 2 pairs of setae, adanal sclerites fused posteriorly, with 3 pairs of setae, seta ad2 hypertrophic. All legs monodactylous, claws smooth. Setal formulas of legs (trochanter to tarsus, famulus included, solenidia in parentheses): leg I 0–3–3(2)–5(1)– 18(3), leg II 0–4–3(1)–4(1)–14(1), leg III 2–3–2(1)–3(1)–13, leg IV 1–2–2(1)–4(1)–11. All setae setiform, smooth.
Etymology — The genus name has two meanings: The prefix ''Arco-'' derives from the arched (latin arcus) clamp-like ridges on the anterior edges of notogaster sclerites. It is also a homage to the municipality of Arco/Trentino (Northern Italy), my second home, where I prepared part of this work. It is compounded with ''-chthonius'', a common generic root for Pseudoscorpions and enarthronote genera (from ancient Greek χθόνιος, meaning ''underworld'' or ''subterranean'').
Arcochthonius roynortoni n. sp.
ZOOBANK: 987EFFA4-700F-4213-8B45-63A61F876EC0 ![]()
Figures 1–5













Diagnosis
As for the genus.
Description
Adult female
Measurements — Total body size (n=11): L 267 (range: 260–280), W 154 (150–165), length of notogaster 202 (196–211, ~75% of total body length), lateral height (genital sclerite – base of seta d1 in lateral view) 131 (125–140).
Integument — Prodorsum sculptured with longitudinal and transversal ridges, notogaster with numerous smaller and larger plates-like sculpturing, ridges and lines. Ventral part of posterior notogastral sclerite (PP) with small tubercles and mosaic-like plates. Body colour pale yellowish to light brown, some specimens whitish.
Prodorsum (Figs 1, 3, 4D, E) — Rostrum extended anteriad and ventrad, apex with 3–5 larger teeth, laterally serrated. Lateral edge of prodorsum with thickened ridge, with some teeth at level of lamellar setae. Between rostral setae two protrusions bulging anteriad, with small, not protruding scale medially (Fig. 4D). Anterior prodorsal region with 4 transverse ridges with small knobs, additional transverse and longitudinal ridges between interlamellar setae, posterior 3–4 pairs of muscle sigillae visible. Prodorsal setae smooth, stout, attenuating, inserting on small tubercles, of medium length. Rostral seta (ro) directed anteriomediad (18–22), lamellar seta (le) directed posteromediad (25–30), interlamellar seta (il) directed anteromediad (30), thus le and il forming an ''X'', exobothridial seta (exa) on well-developed lateral hump anterolateral of bothridium, strongly bent posteriad (25). Posterior exobothridial pore (exp) lateral to bothridium. Bothridium cup-shaped, directed posterolaterad, bothridial seta (bs, 40–50) spoon-shaped, with short stalk and long and narrow head, unilaterally with numerous small bristles.
Notogaster (Figs 1–3, 4F) — Shape subquadrangular in dorsal view, lateral edges almost parallel. Anterior edges of medial and posterior notogaster sclerites with distinct and broad (12–15) transverse thickenings, spanned by arched clamp-like ridges, formed by raised sculpturing. Number of ridges variable (NM 11/12, NP 14/15). On both sclerites one single ridge medially, proximal two ridges posteriorly connected, others partially with posterior appendices, also formed by raised sculpturing. Two medial bands of embossed sculpturing traversing notogaster longitudinally, at level of setal row c1–d1 on anterior notogastral sclerite (NA), at level of e1 on medial (NM), and at level of row f1–h1 on posterior sclerite (NP). Additional plate-like sculpturing at base of most setae, posterolateral of c1 and along posterior edges of sclerites NA and NM. Regular medial field groups on all notogastral sclerites. Some maculae on sclerite NA, mainly around insertion plate of seta d2 . Notogastral setae smooth, stout, attenuating, setae of rows c, d, e, and seta f1 and h1 on distinct sculptured plates, seta p1 and p2 on common plate, f2 , h2 , h3 , p3 not on sculptured plates. All setae inserting on tubercles and of medium length (c1 40, c2 35–40, c3 27–32, d1 42–45, d2 , 45–50, d3 40, e1 45–50, e2 30–35, f1 , f2 , h1–h3 , p1 , p2 25–30, p3 15–18). Seta d2 close to d1 , seta p2 posterior to p1 . Setae p2 and p3 visible in lateral and ventral view.
Pleural region (Figs 2, 3) — Anterior and posterior pleural sclerites (PA, PP) separated, in ventral region closer and almost touching, but never overlapping. Sclerite PA triangular, large (75–80 x 50), anterior apex reaching close to region of lyrifissure ia. Insertion of fine muscle fibres (?, mf) visible on anterior pleural sclerite. Anteroventral part of sclerite PP with polygonal tesselate plate-shaped sculpturing. Suprapleural sclerite SpE rhombic (50 x 20), lateral to notogastral sclerites NA and NM, with thin suture at level of scissure between NA and NM. Lyrifissures as small cupules, in usual position (cf. Moritz 1976a).
Gnathosoma (Figs 2, 4A–C) — Size of subcapitulum 45–55 x 40–45. Four pairs of short attenuating setae (h, m, a 7–10, ma 10–12), 1 pair of spiniform adoral setae (or, 8–10). Rutellum (ru) small (15 x 4–5), with distal teeth directed anteromediad. Palp (45–50) setal formula as for the genus, setae (u) fused, bifid. Lyrifissure on palptarsus positioned proximodorsally. Chelicera (55–60) with one setiform and smooth seta on dorsum of fixed digit (cha, 13), directed anteriad. Movable digit (MD) small. Cheliceral teeth very weakly developed.
Epimeral region (Fig 2) — Epimeral setal formula as for genus, setae setiform (~8–10), setae 1a, 2a on round plates.
Anogenital region (Figs 2, 3) — Genital sclerites large (L 60–65, W ~25), 7 pairs of genital setae in two longitudinal rows, 4 medially, 3 laterally (all ~10–12), aggenital sclerites distinct, large, 1 pair of aggenital setae (~8), peranal sclerites narrow, tapering posteriad, 1 pair of peranal setae (~8), anal sclerites small, long, tapering posteriad, 2 pairs of anal setae (~8–10) on small ridge, adanal sclerites fused, forming a common U-shaped sclerite (W 10–12), width posteriorly slightly diminished by broadened ridge around anal sclerite, adanal setae on small raised ridge, ad1 , ad3 setiform (12), ad2 hypertrophic, lance-shaped, with small velum (~20–25).
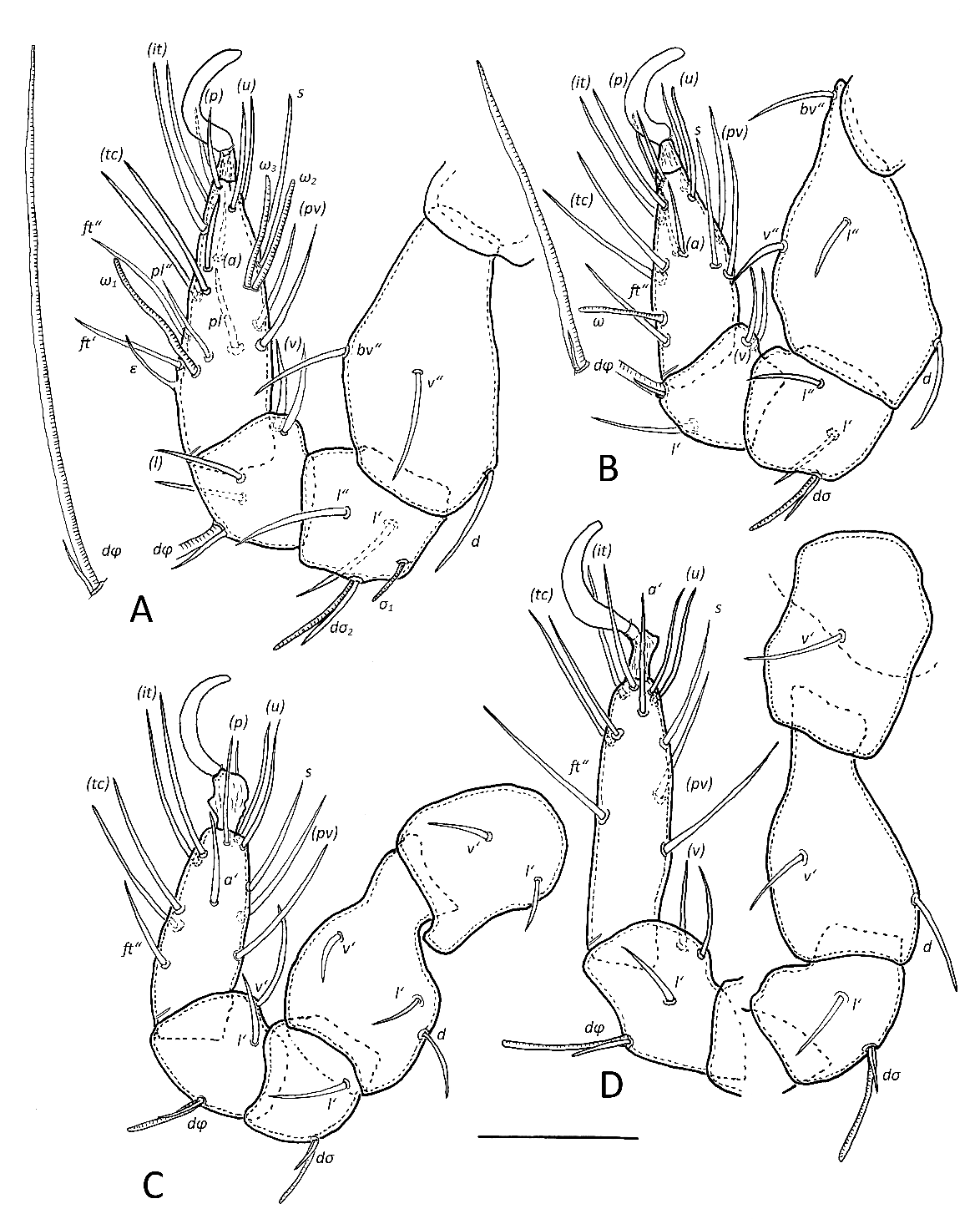
Legs (Figs 5 A–D) — Length of legs (incl. claws) moderate (leg I–III 39–42%, leg IV 52% of body length). Leg IV considerably longer than other legs (trochanter IV 30–32). Leg I 110–125, leg II 100–115, leg III 105–120, leg IV 135–150. Setal formula of legs as for the genus. Solenidion φ on tibia I long (68–77), on tibiae II shorter (37–45), other solenidia (σ, ω) short (~15–25), σ1 on genu I very short (7–9). Famulus ε on tarsus I of medium length (~10). Coupled setae and solenidia on genua and tibiae I–IV. Proximodorsal lyrifissures present on all tarsi. Homology of leg setae and solenidia as indicated in Table 1.



Male — unknown (all Brachychthoniidae species are supposed to be parthenogenetic, Norton and Palmer 1991, Cianciolo and Norton 2006).
Immatures — unknown.
Eggs — One specimen with one egg (127 x 77), surface smooth.
Variation — Two specimens with 7/8 genital setae (see remark #6)
Material examined
Austria, Central Alps, Ötztal Alps, Obergurgl (municipality Sölden), summit of Festkogel mountain (13. June 1999, 46°51.17'N, 11°3.07'E, 3030 m a.s.l.), soil samples, TH 359 BP2b, moss cushion (1 adult specimen), TH 359 BPc, grass tuft (10 adult specimens), all leg. Konrad Thaler.
Type deposition
The holotype (adult female) and two paratypes (females) from the type locality, preserved in ethanol, are deposited in the collection of the Senckenberg Museum, Görlitz, Germany (SMNG). Additional material is deposited in the collection of the author, which will finally also be placed in the Senckenberg Museum, Görlitz.
Etymology
This species is named in honour of my friend and colleague Dr. Roy A. Norton, who has extensively contributed to our knowledge of oribatid mites. He supported and motivated my work in many ways.
Remarks
- Body size — Arcochthonius roynortoni n. sp. belongs to the largest species of the Brachychthoniidae (total length of 260–280 µm). Few species of this family reach a length greater than 250 µm: Eobrachychthonius latior (Berlese, 1910): 270–335 µm, E. oudemansi Hammen, 1952: 250–335 µm, Neobrachychthonius magnus Moritz, 1976: 230–265 µm.
- Rostral tectum — The small scale on the rostral tectum might contain an internal ocellus (''oeil infère'', defined by its cuticular morphology) which has lost its pigment and original function (Grandjean 1963). This structure was found in several early derivative oribatid taxa, some Brachychthoniidae and other Enarthronota, Palaeosomata, and also in Endeostigmata (Grandjean 1958, Alberti & Moreno-Twose 2012). A variability in the jagged outline of the frontal rostral apex as in Arcochthonius roynortoni n. sp. (cf. fig. 4E) was also observed in Synchthonius crenulatus by Engelmann (1969 fig. 1, sub S. boschmai).
- Cuticular structures on notogaster — The arched clamp-like ridges on the anterior edges of the medial and posterior notogastral sclerites of Arcochthonius roynortoni n. sp. are unique among Brachychthoniidae species. Nevertheless, similarities of the general notogastral sculpturing of this species with the sculptured cuticular pattern of the Synchthonius species are discernible. The illustration of Synchthonius elegans (Fig. 67 in Chinone & Aoki 1972) shows certain resemblances with A. roynortoni n. sp., especially in the embossed sclerotisation in the medial region of the notogaster. According to Balogh & Mahunka (1983) the ''heavy, irregular sculpture'' and ''definite ornamentation'' of Synchthonius with median trilobate transverse structures on the notogastral sclerites is ''fundamentally different from that of all other Brachychthoniid genera''. Arcochthonius roynortoni n. sp. exhibits curved lines in the median area of the notogastral sclerites, partially in similar position as the ''irregular'' sculptures on Synchthonius species, but neither trilobate nor with ''small chitinous thickenings'' (Hammen 1952).
- Suprapleural sclerite SpC and position of notogastral seta c3 — Species of most Brachychthoniidae genera have a distinct anterior suprapleural sclerite SpC. This sclerite is partially integrated into the anterior notogastral sclerite NA in Synchthonius Hammen, 1952, Verachthonius Moritz, 1976, Arcochthonius n. gen., in Mixochthonius Niedbała, 1972, integrated to a large extent, and in Liochthonius Hammen, 1959, completely integrated. The notogastral seta c3 always arises from sclerite SpC, if this sclerite is present. In Synchthonius, Verachthonius and Mixochthonius species seta c3 arises on the unsclerotized integument of NA in the position of the integrated sclerite SpC, but in Arcochthonius roynortoni n. sp. seta c3 arises on a distinct sculptured plate on NA.
- Connection of pleural sclerites — The anterior and posterior pleural sclerites are separated to varying degrees in most Brachychthoniidae genera. Hammen (1952: p. 20) stated in his generic diagnosis of Synchthonius that ''the ventral shield in its anterior part (i.e. PA) is separated from the pygidial shield (PP)'', which can be seen in his Fig. 1c (p. 16) of Synchthonius boschmai (= S. crenulatus). However, Grandjean (1963, p.125) discussed this character controversially (translated from the original French): ''There is a strong separation between the triangular and the posterior part, which I currently call spt (=sillon pleurotransversal) or pleurotransverse groove, which runs from one edge to the other of the shield. Synchthonius elegans does not have a complete ascleritic suture because, despite several attempts, I was unable to divide the shield along this line. The cuticle resists or it tears if one pulls harder by not following the line spt. In my figures we only see spt at its paraxial end or lower (Figures 4A, 4B, 5A)''. Also in other available figures of Synchthonius as well in own material (Schatz 2004, 2008) the pleural sclerites are fused or partially shaped as an overlapping suture (Ghilarov & Krivolutsky 1975, fig. 63b, Moritz 1976b, figs 5b, c, 6b, and authors using the figures of Moritz, Chinone 1978, fig. 10, Forsslund 1956, fig. 2, Chinone & Aoki 1972, figs 67, 68). In Archochthonius roynortoni n. sp. the furrow is small, but never formed as overlapping suture as in Synchthonius.
- Number of genital setae — Most Brachychthoniidae species have 7 pairs of genital setae; Liochthonius (Afroliochthonius) reductus Mahunka, 1995 is an exception with 6 pairs of genital setae. According to most authors (Grandjean 1963, Moritz 1976b, Balogh & Mahunka 1983, Weigmann 2006) 6 pairs of genital setae is an apomorphy of the genus Synchthonius. However, according to Chinone & Aoki (1972) the genital setae of S. elegans ''seem to have a tendency to vary in number'', and they indicate an asymmetrical number of genital setae on their investigated specimen with 8 setae on the right and 6 setae on the left side. Also, the figure of the original description of S. elegans (Forsslund 1956) exhibits 5 genital setae on the right and 6 setae on the left genital sclerite. Two specimens of Arcochthonius roynortoni n. sp. also have a variable and asymmetrical number of genital setae with 8 setae on the right and 7 setae on the left genital sclerite, the other investigated specimens have 7 setae on each genital sclerite, but never less than 7.
- Leg chaetotaxy — The chaetotaxy of leg segments vary in different genera of Brachychthoniidae (Moritz 1976a). These variations concern the following segments: Leg I: tarsus (famulus included) 16–18 setae (Arcochthonius 18), leg II: tarsus 12–15 setae (Arcochthonius 14), leg III: genu 2–3 setae (Arcochthonius 2), tarsus 11–13 setae (Arcochthonius 13), leg IV: genu 2–3 setae (Arcochthonius 2). The legs of Synchthonius elegans have the same setal formulas as Arcochthonius roynortoni n. sp. Moritz (op. cit.) indicates the close position of setae d with solenidia on genua and tibiae I–IV. The presence of these cover setae is constant within the family, but they were overlooked on tibiae I and II by Grandjean (1963, supplementary description of Synchthonius elegans), Reeves & Marshall (1971, redescription of Brachychthonius lydiae), and could not be observed on tibia I by Akrami (2016, description of Eobrachychthonius iranicus).
Systematic relationship
Arcochthonius roynortoni n. sp. shares several morphological character states with known Synchthonius species: body surface strongly sclerotized and arched in lateral view, rostral apex anteriorly with large teeth, lateral edge of rostrum serrated and with large teeth, position of notogastral seta d2 close to d1 , prosomatic suprapleurite absent, suprapleural sclerite SpE present, sclerite SpC partially fused with NA, other suprapleural sclerites absent, suprapleural incision deep, aggenital sclerite present, adanal seta ad2 hypertrophic, setal formula on legs.
Arcochthonius differs from the Synchthonius species in the following: pleural sclerites separated in A. roynortoni n. sp. (vs. fused in Synchthonius, see remark #5), seven pairs of genital setae in A. roynortoni n. sp. (vs. six pairs in Synchthonius, see remark #6), large exobothridial hump in A. roynortoni n. sp. (vs. small in Synchthonius), notogastral seta c3 on distinct embossed sculpturing in A. roynortoni n. sp. (vs. directly on integument of notogastral sclerite NA in Synchthonius, see remark #4),
The genus Synchthonius Hammen, 1952 includes two described species, S. crenulatus (Jacot, 1938) and S. elegans Forsslund, 1956. Moritz (1976b: p. 243) proposed Synchthonius boschmai Hammen, 1952 as a synonym of S. crenulatus (Jacot, 1938). Subías (2017, unpublished electronic update of his 2004 checklist) placed Liochthonius lentus Chinone, 1974 to Synchthonius without any comment, a suggestion I reject. This species has some faint irregular spots, but lacks the character states of known Synchthonius species. The figure of the ventral side of this species (Chinone 1974, fig. 71) depicts different numbers of setae (6/7) on the genital sclerites (see also remark #6).
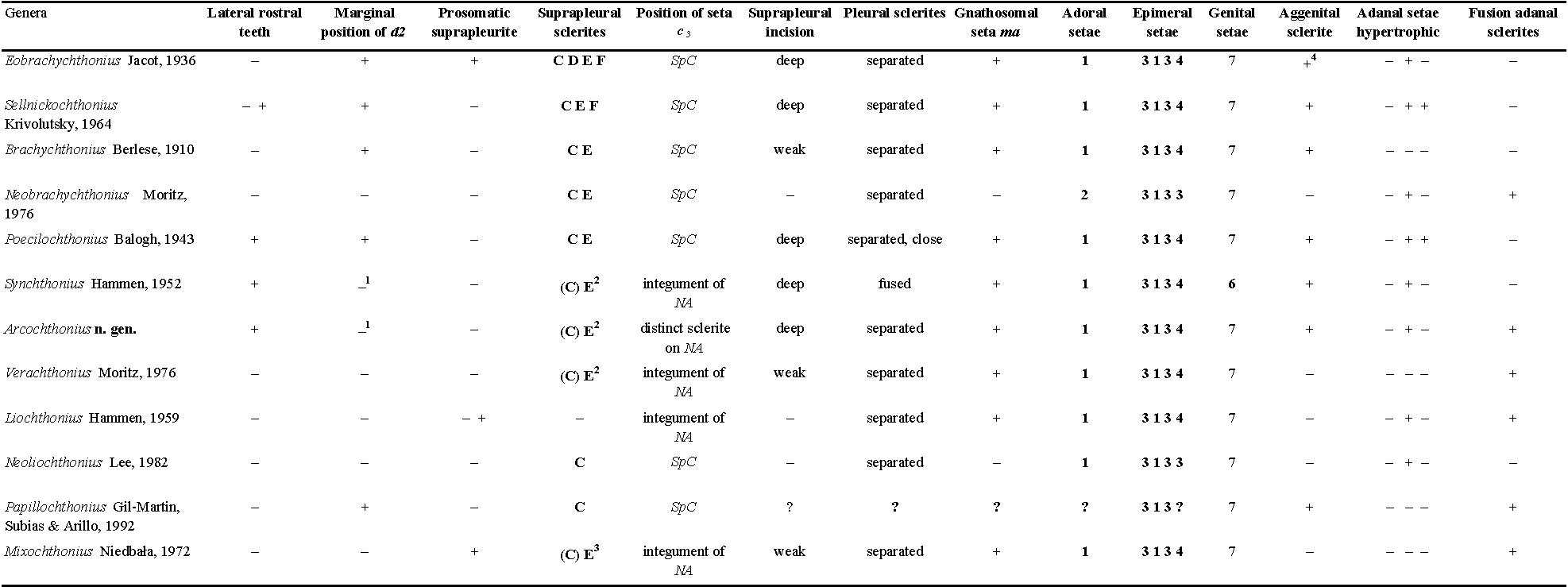


Many characters of Arcochthonius are shared with other Brachychthoniidae genera (Table 2). In addition to Synchthonius, species of Poecilochthonius Balogh, 1943 and Verachthonius Moritz, 1976 show most similarities with Arcochthonius n. gen.: Poecilochthonius: prodorsal and notogastral surface ornamented, lateral rostral teeth present, prosomatic suprapleurite absent, suprapleural sclerite SpE present, pleural sclerites separated, aggenital sclerite present; Verachthonius: prosomatic suprapleurite absent, d2 close to d1 , suprapleural sclerite SpE present, pleural sclerites separated, p2 posterior to p1 , adanal sclerites fused caudally.
The new species differs from the Poecilochthonius and Verachthonius species in the following: ornamentation of prodorsum and notogaster (vs. smooth in Verachthonius, different ornamentation pattern in Poecilochthonius), rostral apex with three larger teeth (vs. serrations without teeth in Poecilochthonius, smooth and without teeth in Verachthonius), lateral rostral teeth present (vs. absent in Verachthonius), seta d2 close to d1 (vs. d2 near notogastral margin in Poecilochthonius), suprapleural sclerite SpC fused with NA (vs. distinct with seta c3 in Poecilochthonius, fused with NA with distinct lateral lobe, seta c3 on unsclerotized integument in Verachthonius), seta p2 posterior to p1 (vs. p2 laterocaudal to p1 in Poecilochthonius), suprapleural incision deep and distinct (vs. short in Poecilochthonius and Verachthonius), aggenital sclerite present (vs. absent in Verachthonius), adanal sclerites fused caudally (vs. separated in Poecilochthonius), adanal seta ad2 hypertrophic (vs. ad2 not hypertrophic, setiform in Verachthonius, ad2 and ad3 hypertrophic in Poecilochthonius).
The morphological differences of Arcochthonius roynortoni n. sp. to species of these and other Brachychthoniidae genera justify the erection of a separate genus.
Notes on ecology and biology
In the course of previous studies in the Obergurgl area, Central Alps, 12 Brachychthoniidae species were recorded (Fischer & Schatz 2013, Schatz 1979, 2004). Some species of this family were found in considerable abundances in raw soils (Schatz 2004). Remarkably, on the summit of the Festkogel mountain a further six Brachychthoniidae species beside Arcochthonius roynortoni n. sp. were encountered (Schatz in prep.). As mentioned above, this locality is only sparsely vegetated on the border between alpine grassland and subnival cushion plant formations with scarce pioneer vegetation. Numerous records of Brachychthoniidae species are reported from raw soil and lichens (e.g. Schweizer 1956, Moritz 1976a, b, Leonov 2020).
Arcochthonius roynortoni n. sp. seems to be a rare species. It was detected only in two soil samples in 1999 on the summit of the Festkogel mountain despite numerous additional sampling in the same spot. Also, this species was not found in the course of intensive collecting at lower elevations on the slope of the Festkogel (Schatz 1979). Possibly it survived as preglacial relict on a nunatak or it evolved in this altitude in isolation (cf. Schatz 2020).
Little is known on the feeding preferences of Brachychthoniidae species. Schuster (1956) demonstrated a relationship between relative size of chelicerae and feeding type. Based on the morphology of the chelicera, Brachychthoniidae species can be assumed to be ''microphytophagous'', consuming microorganisms as fungi, bacteria and algae (Lebrun 1971). Microphytophagous oribatid species are very abundant in alpine grassland and subnival cushion plants (Schatz 1979). These observations also apply to the locality of Arcochthonius roynortoni n. sp. This species has a small movable digit with weakly developed teeth and can also be classsified as microphytophagous in the sense of the above authors.
Acknowledgements
Sincerest thanks to the late Konrad Thaler, the renowned Arachnologist, who collected this material, and to Barbara Thaler-Knoflach for valuable information about the location. The institutes of Ecology and Zoology, University of Innsbruck gave logistic support. Thanks are also due to Valerie Behan-Pelletier, Irene Schatz and two anonymous reviewers for their valuable suggestions and comments on the manuscript.
References
- Akrami M.A. 2016. The oribatid mite family Brachychthoniidae of Iran (Acari: Oribatida), with description of a new species. Syst. Appl. Acarol., 21(4): 405-416. doi:10.11158/saa.21.4.3
- Alberti G., Moreno-Twose A.I. 2012. Fine structure of the primary eyes in Heterochthonius gibbus (Oribatida, Heterochthoniidae) with some general remarks on photosensitive structures in oribatid and other actinotrichid mites. Soil Org., 84(2): 391-408.
- Balogh J., Mahunka S. 1983. Primitive Oribatids of the Palaearctic Region. In: Balogh J., Mahunka S. (Eds.). The soil mites of the world. Akademiai Kiado Budapest vol. 1: pp. 372.
- Chinone S. 1974. Further contribution to the knowledge of the family Brachychthoniidae from Japan. Bull. Biogeogr. Soc. Jpn., 30: 1-29.
- Chinone S. 1978. Additional report on the soil mites of the family Brachychthoniidae from Japan. Bull. Biogeogr. Soc. Jpn., 33: 9-32.
- Chinone S., Aoki J. 1972. Soil mites of the family Brachychthoniidae from Japan. Bull. Nat. Sci. Mus., Tokyo, 15: 217-251.
- Cianciolo J.M., Norton R.A. 2006. The ecological distribution of reproductive mode in oribatid mites, as related to biological complexity. Exp. Appl. Acarol., 40: 1-25. doi:10.1007/s10493-006-9016-3
- Engelmann H.D. 1969. Beitrag zur Oribatidenfauna des Altvatergebirges (Jeseniky). Abh. Ber. Naturkundemus. Görlitz, 44(2): 95-100.
- Fischer B.M., Schatz H. 2013. Biodiversity of oribatid mites (Acari: Oribatida) along an altitudinal gradient in the Central Alps. Zootaxa, 3626 (4): 429-454. doi:10.11646/zootaxa.3626.4.2
- Forsslund K.H. 1956. Schwedische Oribatei (Acari) III. Entomol. Tidskr., 77(2-4): 210-218.
- Ghilarov M.S., Krivolutsky D.A. 1975. (Eds.). Identification keys of soil inhabiting mites, Sarcoptiformes. Nauka, Moscow, 491 pp. [in Russian]
- Grandjean F. 1958. Au sujet du naso et de son oeil infère chez les Oribates et les Endeostigmata (Acariens). Bull. Mus. Hist. Nat. (2), 30: 427-435.
- Grandjean F. 1963. Sur deux especes de Brachychthoniidae et leur developpement (Oribates). Acarologia, 5: 122-151.
- Hammen L. van der 1952. The Oribatei (Acari) of the Netherlands. Zool. Verh., Leiden, 17: 1-139.
- Krisper G., Schatz H., Schuster R. 2017. Oribatida (Arachnida: Acari). Checklisten der Fauna Österreichs, Nr. 9, Biosystematics and Ecology Series, Band 33. Verlag der Österreichischen Akademie der Wissenschaften, Wien: 25-90. doi:10.2307/j.ctvdjrr69.5
- Lebrun P. 1971. Écologie et biocénotique de quelques peuplements d'arthropodes édaphiques. Mem. Mus. r. hist. nat. Belg., 165: 1-203.
- Leonov V.D. 2020. The first report on the oribatid mites (Acari: Oribatida) in tundra of the Chunatundra Mountains on the Kola Peninsula, Russia. Acarologia, 60(4): 722-734.
- Mahunka S. 1995. Two new Brachychthoniid species (Acari: Oribatida) from the Comoro Islands. Acarologia, 36(3): 241-245.
- Moritz M. 1976a. Revision der europäischen Gattungen und Arten der Familie Brachychthoniidae (Acari, Oribatei). Teil 1. Allgemeiner Teil: Brachychthoniidae Thor, 1934. Spezieller Teil: Liochthonius v.d.Hammen, 1959, Verachthonius nov. gen. und Paraliochthonius nov. gen. Mitt. Zool. Mus. Berlin, 52: 27-136. doi:10.1002/mmnz.19760520104
- Moritz M. 1976b. Revision der europäischen Gattungen und Arten der Familie Brachychthoniidae (Acari, Oribatei). Teil 2. Mixochthonius Niedbala, 1972, Neobrachychthonius nov. gen., Synchthonius v.d.Hammen, 1952, Poecilochthonius Balogh, 1943, Brachychthonius Berlese, 1910, Brachychochthonius Jacot, 1938. Mitt. Zool. Mus. Berlin, 52: 227-319. doi:10.1002/mmnz.4830520205
- Norton R.A., Behan-Pelletier V. 2009. Chapter 15, Oribatida. In: Krantz G.W., Walter D.E. (Eds). A Manual of Acarology 3rd Edition. Lubbock. Texas Tech University Press. p. 421-564. doi:10.1653/024.092.0323
- Norton R.A., Palmer S.C. 1991. The distribution, mechanisms and evolutionary significance of parthenogenesis in oribatid mites. In: Schuster R., Murphy P.W. (Eds.). The Acari - Reproduction, development and life-history strategies. London - New York. Chapman and Hall. p. 107-136. doi:10.1007/978-94-011-3102-5_7
- Reeves R.M., Marshall V.G. 1971. Redescription and chaetotaxie of Brachychthonius lydiae adults and nymphs (Acarina: Oribatei). Ann. Entomol. Soc. Am., 64(2): 317-325. doi:10.1093/aesa/64.2.317
- Schatz H. 1979. Ökologische Untersuchungen an Wirbellosen des zentralalpinen Hochgebirges (Obergurgl, Tirol). II. Phänologie und Zönotik von Oribatiden (Acari). Veröffentlichungen der Universität Innsbruck (Alpin-biologische Studien 10), Innsbruck, 117: 15-120.
- Schatz H. 2004. Die Hornmilbenfamilie Brachychthoniidae (Acari: Oribatida) in Tirol (Österreich). Denisia, Linz, 12: 343-355.
- Schatz H. 2008. Hornmilben (Acari: Oribatida) im Naturpark Schlern Rosengarten (Südtirol, Italien). Gredleriana, 8: 219-254.
- Schatz H. 2020. Oribatid mites (Acari: Oribatida) from the Sella massif (Dolomites, Trentino, Italy) with description of Trichoribates valeriae n. sp. (Ceratozetidae). Acarologia, 60(4): 842-862. doi:10.24349/acarologia/20204405
- Schatz H., Behan-Pelletier V.M., OConnor B.M., Norton R.A. 2011. Suborder Oribatida van der Hammen, 1968. In: Zhang Z.-Q. (Ed). Animal biodiversity: An outline of higher-level classification and survey of taxonomic richness. Zootaxa, 3148: 141-148. doi:10.11646/zootaxa.3148.1.26
- Schuster, R. 1956. Der Anteil der Oribatiden an den Zersetzungsvorgängen im Boden. Zeit. Morphol. Oekol. Tiere, 45: 1-33. doi:10.1007/BF00699814
- Schweizer, J. 1956. Die Landmilben des Schweizerischen Nationalparkes. 3. Teil, Sarcoptiformes Reuter 1909. Ergebnisse der wissenschaftlichen Untersuchungen des schweizerischen Nationalparks, Neue Folge, Liestal, 5(34): 213-377.
- Subías L.S. 2004. Listado sistemático, sinonímico y biogeográfico de los ácaros oribátidos (Acariformes, Oribatida) del mundo (1758-2002). Graellsia, 60 (número extraordinario): 3-305 (electronic updates 2017, 2020). doi:10.3989/graellsia.2004.v60.iExtra.218
- Travé J., André H.M., Taberly G., Bernini F. 1996. Les Acariens Oribates. Wavre, Belgium: AGAR Publishers. pp. 110.
- Travé J., Vachon M. 1975. François Grandjean. 1882-1975 (Notice biographique et bibliographique). Acarologia, 17(1): 1-19.
- Weigmann G. 2006. Hornmilben (Oribatida). Die Tierwelt Deutschlands, 76. Teil. Keltern: Goecke & Evers. pp. 520.



2021-03-08
Date accepted:
2021-04-25
Date published:
2021-04-29
Edited by:
Baumann, Julia

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Schatz, Heinrich
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)




