A new species of Adactylidium (Acari: Heterostigmata: Acarophenacidae) associated with Phlaeothrips sp. (Thysanoptera: Phlaeothripidae) from European Russia
Khaustov, Alexander A.1 and Abramov , Vladimir V.2
1✉ Tyumen State University, Tyumen, Volodarskogo 6, 625003 Russia.
2Gagarin str. 12, Suvorov, Tula Region, 301430, Russia.
2021 - Volume: 61 Issue: 2 pages: 356-364
https://doi.org/10.24349/acarologia/20214435ZooBank LSID: E77F0742-DF53-4704-AA60-B17FFEBBBF6C
Original research
Keywords
Abstract
Introduction
The family Acarophenacidae includes species that are parasitoids of eggs of various insects, including beetles from the families Cerambycidae, Erotylidae, Tenebrionidae, Nitidulidae, Dermestidae, Curculionidae (Scolytinae), Mycetophagidae and thrips (Thysanoptera) (Goldarazena et al. 2001; Katlav et al. 2015; Ardjomandi et al. 2017; Walter and Seeman 2017; Khaustov and Abramov 2018; Xu et al. 2018). At present, the family includes about 40 described species (including three extinct species) from eight genera (Khaustov et al. 2021). Khaustov et al. (2021) provided the latest key to the genera of Acarophenacidae. Currently six species of acarophenacid mites have been recorded from Russia: Aethiophenax ipidarius (Redikortzev, 1947), Paracarophenax scolyti Khaustov, 1999 (both associated with bark beetles), P. bambergensis (Krczal, 1959) (associated with Nitidulidae), P. triplaxophilus Khaustov and Abramov, 2018 (associated with Erotylidae), Paradactylidium pyemotoformis Khaustov, 2007 and Adactylidium absurdum Khaustov, 2007 (both from unknown hosts) (Khaustov 1999, 2007; Khaustov and Abramov 2018, 2019).
The genus Adactylidium Cross, 1965 comprises 16 described species distributed in Europe, North and South Americas, and northern Africa (Goldarazena et al. 2001; Antonatos et al. 2011). All Adactylidium species are egg parasitoids of various thrips species (Thysanoptera). Therefore, they have been suggested to be potential biological control agents of thrips pests in agricultural systems (Antonatos et al. 2011).
During the study of insect associated mites, a new species of Adactylidium associated with Phlaeothrips sp. was revealed from the European Russia. The aim of this article is to describe this new species and provide an updated key to world species of Adactylidium.
Material and methods
Mites were collected from thorax of larvae of Phlaeothrips sp. by the junior author. The mites were mounted in Hoyer's medium. The terminology follows that of Lindquist (1986). All measurements are given in micrometers (μm) for the holotype and two female paratypes (in parentheses). For leg chaetotaxy the number of solenidia is given in parentheses. Differential interference contrast (DIC) micrographs were taken using the Carl Zeiss Axio Imager A2 compound microscope and Hitachi KP-HD20A and Olympus OM-D Em-10 digital cameras.
Abbreviations — ap1-ap5 – apodemes 1-5; appr – prosternal apodeme; appo – poststernal apodeme; apsej – sejugal apodeme; apgn – gnathosomal apodeme.
Results
Systematics
Family Acarophenacidae Cross, 1965
Genus Adactylidium Cross, 1965
Type species: Adactylidium beeri Cross, 1965, by original designation
Adactylidium europaeum n. sp.















ZOOBANK: 227447D6-8908-4C9C-8555-E92E102DC85A ![]()
(Figures 1–6)
Diagnosis — Apgn with median projections; genua I and III with three and one setae, respectively; tibiotarsus I with ventral subapical indentation; setae (tc) and (ft) of tibiotarsus I subequal in length; dorsal idiosomal setae with similar shape, elongate; apsej not fused with appr; setae 1b absent.
Description
Female – Length of idiosoma 130 (130, 135), width 100 (98, 105).
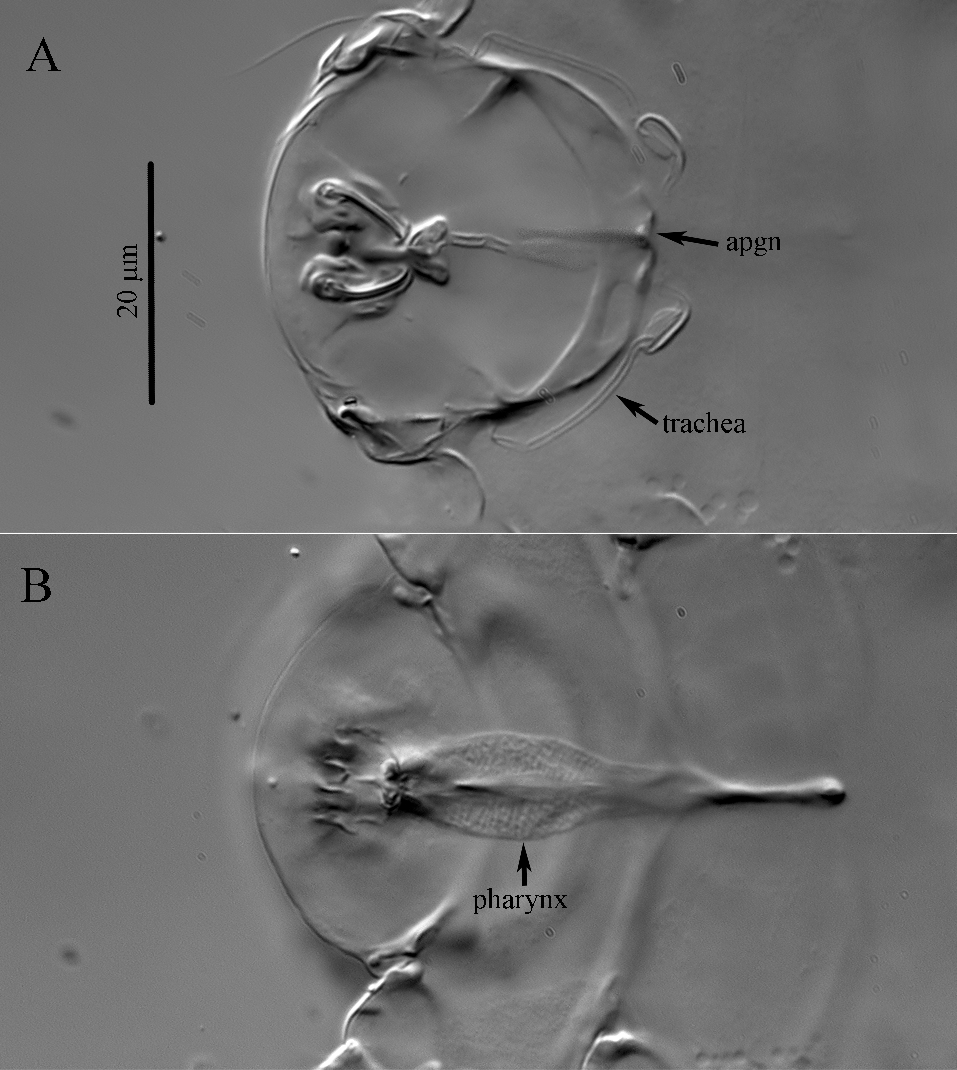
Gnathosoma ‒ Apgn with distinct median projections (Fig. 6 A). Gnathosoma concealed dorsally by prodorsum, ventrally indistinguishable from idiosoma; dorsum with one pair of setae anterolaterally, probably representing postpalpal seta pp; venter with mouth flanked by paired, semi-ovular smooth membranous areas; one pair of rod-like ventral setae of unknown homology situated at anterior ends of membranous areas; one pair of blunt-ended subcapitular setae m located posteriad membranous areas. Pharynx large, slightly oval, thin walled, distinctly punctate (Fig. 6B).
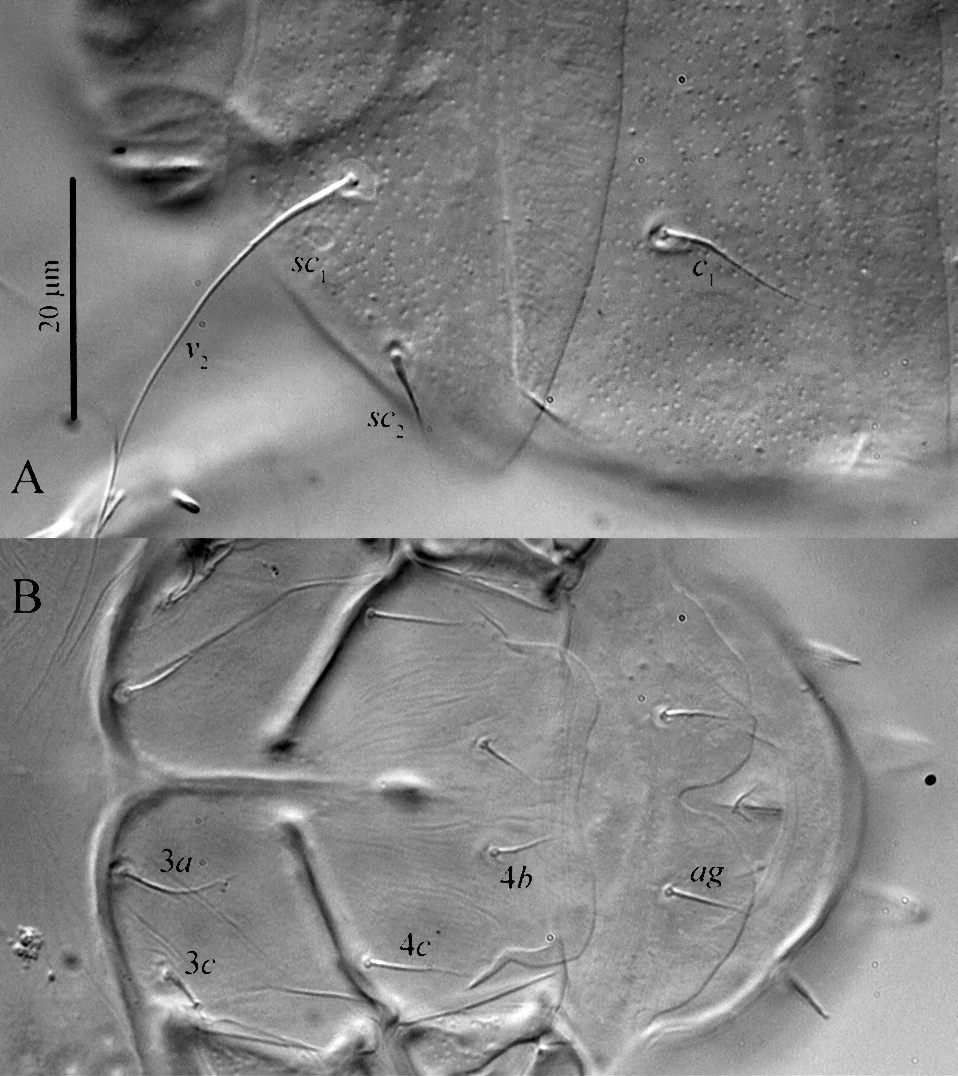
Idiosomal dorsum (Figs 1A, 4A, 5A) ‒ Ovate. Prodorsal shield delineated into primary plate and prodorsal projection, encapsulating gnathosoma. Stigmata on prodorsal projection; tracheal trunks long, with small atria located posteriad posterior margin of apgn. Prodorsal shield and plates C, D, EF, H distinctly punctate (Fig. 5A). Alveolar pits sc1 situated anterolaterad bases of setae v2. Setae v2, sc2, c1 and c2 thin, smooth and pointed; setae d, e and f with few weak barbs in basal half; setae h1 and h2 located ventrally, slightly thickened, blunt-ended and weakly barbed. Cupuli ia small, round, located laterad or anterolaterad bases of setae d; cupuli im situated anteromesad bases of setae e; cupuli ih not evident. Lengths of dorsal setae: v2 38 (38, 43), sc2 18 (18, 19), c1 17 (18), c2 17 (18), d 17 (18, 20), e 15 (17, 18), f 17 (18), h1 7 (8, 9), h2 6 (7, 8). Distances between setae: v2–v2 34 (34, 38), sc1–sc1 43 (42, 47), v2–sc2 17 (16–18), sc2–sc2 66 (65, 71), c1–c1 43 (42, 45), c1–c2 22 (23, 24), d–d 52 (49, 58), e–f 16 (15, 16), f–f 20 (19, 26), e–e 52 (48, 52), h1–h1 13 (11), h2–h2 8 (11, 12).
Idiosomal venter (Figs 1B, 4B, 5B) ‒ Ventral plates smooth; small subtriangular areas posteriad trochanters II with rough microsculpture; coxisternal fields IV with weak longitudinal striation (Fig. 5B). All ventral setae smooth and pointed; setae 1b absent. Aggenital plate with deep incision posteriorly. Ap1 weakly sclerotized, fused with well sclerotized appr; ap2 well sclerotized and fused with appr; apsej well sclerotized, not fused with appr; ap3 well developed, fused with well-developed appo; ap4 well sclerotized, not fused with appo; ap5 absent. Lengths of ventral setae: 2a 11 (12–13), 3a 11 (12, 13), 3c 11 (12), 4b 276 (7, 8), 4c 9 (8, 10), ag 7 (7, 8).
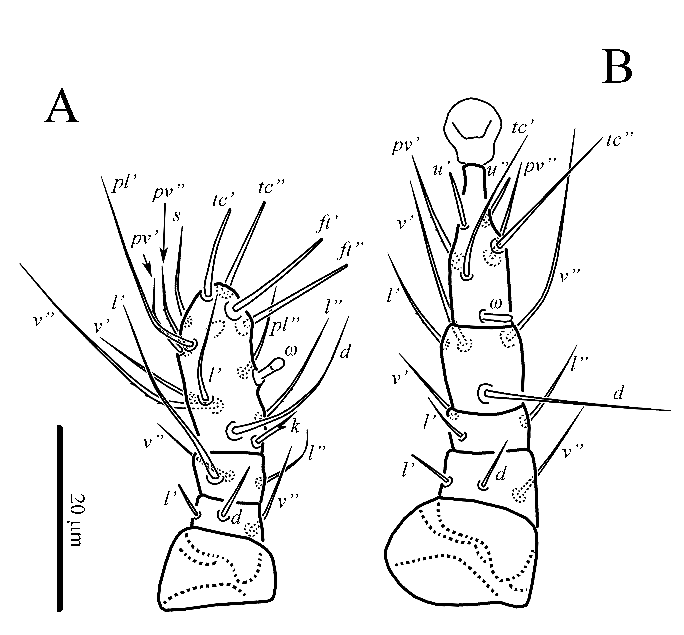
Legs (Figs 2, 3) ‒ Leg I (Fig. 2A) distinctly shorter than other legs. Leg setation: Tr 0, Fe 3, Ge 3, TiTa 15(1). Tibiotarsus with ventral subapical indentation. Solenidion ω 4 (3, 4) clavate, situated in the middle of segment. Seta k bifurcate; eupathidia (tc) and (ft) subequal in length. At least setae l'', v'' and pv'' of tibiotarsus weakly barbed, other leg setae smooth; setae d, l' of femur and eupathidia (tc) and (ft) blunt-ended, other leg setae pointed. Leg II (Fig. 2B). Leg setation: Tr 0, Fe 3, Ge 3, Ti 4, Ta 6(1). Solenidion ω 4 (4, 5) digitiform. Setae l' of femur and (u) of tarsus blunt-ended, other leg setae pointed; at least setae l' of tibia and (pv) of tarsus weakly barbed, other leg setae smooth. Leg III (Fig. 3A). Leg setation: Tr 0, Fe 2, Ge 1, Ti 4, Ta 4. Setae (u) of tarsus blunt-ended, other leg setae pointed; at least setae v' of femur, v' of genu, l', v'' of tibia and pv' of tarsus weakly barbed, other leg setae smooth. Leg IV (Fig. 3B). Leg setation: Tr 0, Fe 1, Ge 1, Ti 4, Ta 4. Setae (u) of tarsus blunt-ended, other leg setae pointed; at least setae v' of genu, l', v'' of tibia weakly barbed, other leg setae smooth; seta tc'' of tarsus ultralong.
Type material — Female holotype, slide ZISP T-Acaroph-001, Tula Region, vicinity of Suvorov town, 54°07' N, 36°30' E, 10 November 2020, on thorax of larva of Phlaeothrips sp. on the bark of thin dead branches of birch, coll. V.A. Abramov. Paratypes: two females, same data, 14 November 2020.
Type deposition — The holotypes of the new species is deposited in the acarological collection of the Zoological Institute of RAS, St. Petersburg, Russia; two female paratypes are deposited in the mite collection of the Tyumen State University Museum of Zoology, Tyumen, Russia.
Etymology — The name of the new species refers to distribution in European part of Russia.
Differential diagnosis — The new species is most similar to Adactylidium ficorum Goldarazena and Ochoa, 2001, described from Puerto Rico and Egypt (Goldarazena et al. 2001) in having three setae on genu I, one seta on genu III, and well developed appo. The new species differs from A. ficorum in having apsej not fused with appr (apsej fused with appr in A. ficorum), setae 1b completely absent (alveoli of setae 1b present in A. ficorum) and median projections present on apgn (absent in A. ficorum).
Key to world species of Adactylidium (females)
1. Tibiotarsus I with solenidion
...... 2
— Tibiotarsus I without solenidion
...... A. absurdum Khaustov, 2007
2. Tarsus III without solenidion
...... 3
— Tarsus III with solenidion
...... A. nicolae (Krczal, 1959)
3. Setae 4b present
...... 5
— Setae 4b absent
...... 4
4. Genu I with three setae; genu II with two setae; dorsal hysterosomal setae short, thickened and distinctly barbed
...... A. brasiliensis Goldarazena and Ochoa, 2001
— Genu I with four setae; genu II with three setae; dorsal hysterosomal setae long, thin and weakly barbed in basal part
...... A. flechtmanni Goldarazena and Ochoa, 2001
5. Genu I with three setae
...... 6
— Genu I with four setae
...... 8
6. Poststernal apodeme well developed
...... 7
— Poststernal apodeme absent
...... A. irregularis Goldarazena and Ochoa, 2001
7. Sejugal apodeme fused with prosternal apodeme; setae 1b present
...... A. ficorum Goldarazena and Ochoa, 2001
— Sejugal apodeme not fused with prosternal apodeme; setae 1b completely absent
...... A. europaeum n. sp.
8. Genu III with one seta
...... 9
— Genu III with two setae
...... 10
9. Coxisternal fields I with tiny setae 1b
...... A. morazae Goldarazena and Ochoa, 2001
— Coxisternal fields I without setae
...... A. beeri Cross, 1965
10. Femur II with three setae
...... 11
— Femur II with two setae
...... A. rumanicus Jordana and Goldarazena, 2001
11. At least setae f similar in shape and length to d
...... 12
— Setae e and f short, thick and distinctly barbed, much shorter than d
...... A. gynaikothripsi Antonatos et al., 2011
12. Setae e much shorter than f
...... 14
— Setae e and f subequal
...... 13
13. Bases of setae e located distinctly anteriad setae f
...... A. lindquisti Jordana and Goldarazena, 2001
— Bases of setae e located slightly posteriad setae f
...... A. moundi Goldarazena et al., 1997
14. Aggenital plate striated
...... 15
— Aggenital plate smooth
...... 16
15. Gnathosomal apodeme with a distal notch; tergite С longer than wide; setae h1 as long as h2
...... A. costarricensis Goldarazena et al., 1997
— Gnathosomal apodeme smooth distally; tergite С wider than long; setae h1 three times longer than h2
...... A. mooniensis Goldarazena and Jordana, 2001
16. Setae v2 and sc2 subequal; apodemes 3 not fused with poststernal apodeme
...... A. smileyi Goldarazena and Ochoa, 2001
— Setae v2 three times longer than sc2; apodemes 3 fused with poststernal apodeme
...... A. crespii Goldarazena and OConnor, 2001
Acknowledgements
The authors thank Dr. V.I. Rozhina (Section of Research of Quarantine Materials, Kaliningrad Interregional Veterinary Laboratory, Kaliningrad, Russia) and Dr. Halina Kucharczyk (Department of Zoology and Nature Protection, Maria Curie-Skłodowska University, Lublin, Poland) for the identification of host thrips larvae. The present research was supported by the Russian Science Foundation, grant No. 20-64-47015.
References
- Antonatos S.A., Kapaxidi E.V., Papadoulis G.Th. 2011. Adactylidium Gynaikothripsi n. sp. (Acari: Acarophenacidae) associated with Gynaikothrips Ficorum (Marshal) (Thysanoptera: Phlaeothripidae) from Greece, Internat. J. Acarol., 37: sup1, 18-26. doi:10.1080/01647954.2010.531763
- Arjomandi E., Hajiqanbar H., Joharchi O. 2017. Aethiophenax mycetophagi sp. nov. (Acari: Trombidiformes: Acarophenacidae), an egg parasitoid of Mycetophagus quadripustulatus (Coleoptera: Mycetophagidae) from Iran. Syst. Appl. Acarol., 22: 541-549. doi:10.11158/saa.22.4.9
- Cross E.A. 1965. The generic relationships of the family Pyemotidae (Acarina, Trombidiformes). Univ. Kans. Sci. Bull., 45: 29-215.
- Goldarazena A., Jordana R., Zhang Z.-Q. 1997. Adactylidium moundi and Adactylidium costarricensis, two new species of Acarophenacidae (Acari: Tarsonemida) parasitic on Thysanoptera. Internat. J. Acarol., 23: 261-268. doi:10.1080/01647959708683575
- Goldarazena A., Ochoa R., Jordana R., OConnor B.M. 2001. Revision of the genus Adactylidium Cross (Acari: Heterostigmata: Acarophenacidae), mites associated with thrips (Thysanoptera). Proc. Entomol. Soc. Wash., 103: 473-516.
- Katlav A., Hajiqanbar H., Talebi A.A. 2015. First record of the genus Aethiophenax (Acari: Acarophenacidae) from Asia, redefinition of the genus and description of a new species. J. Asia-Pac. Entomol., 18: 389-395. doi:10.1016/j.aspen.2015.03.011
- Khaustov A.A. 1999. Redescription of <Pediculoides> ipidarius Redikortsev, 1947, and a description of a new species from the genus Paracarophenax (Acari: Heterostigmata: Acarophenacidae). Acarina, 7: 57-59.
- Khautsov A.A. 2007. Two new species of mites of the family Acarophenacidae (Acari, Heterostigmata) from Crimea (Ukraine). Vestn. Zool. 41: 549-553.
- Khaustov A.A., Abramov V.V. 2018. A new species of Paracarophenax (Acari: Heterostigmata: Acarophenacidae) associated with Triplax scutellaris (Coleoptera: Erotylidae) from European Russia. Acarologia, 58: 332-341. doi:10.24349/acarologia/20184245
- Khaustov A.A., Abramov, V.V. 2019. First record and redescription of Paracarophenax bambergensis (Acari: Heterostigmata: Acarophenacidae) associated with Amphotis marginata (Coleoptera: Nitidulidae) from European Russia. Acarina, 27: 45-51. doi:10.21684/0132-8077-2019-27-1-45-51
- Khaustov A.A., Vorontsov D.D., Perkovsky E.E., Lindquist E.E. 2021. Review of fossil heterostigmatic mites (Acari: Heterostigmata) from late Eocene Rovno Amber. I. Families Tarsocheylidae, Dolichocybidae and Acarophenacidae. Syst. Appl. Acarol., 26: 33-61. doi:10.11158/saa.26.1.3
- Krczal H. 1959. Systematik und Ökologie der Pyemotiden. Beitr. Syst. Ökol. Mitteleurop. Acarina, 1: 385-625.
- Lindquist E.E. 1986. The world genera of Tarsonemidae (Acari: Heterostigmata): a morphological, phylogenetic, and systematic revision, with a reclassification of family-group taxa in the Heterostigmata. Mem. Entomol. Soc. Can., 118: 1-517. doi:10.4039/entm118136fv
- Redikortzev V.V. 1947. The mite Pediculoides ipidarius sp. n. (Acari: Pediculoididae), a parasite of bark beetles. Entomol. Obozr., 29: 247-249. [In Russian]
- Walter D.E., Seeman O.D. 2017. A new species of Paracarophenax (Acariformes: Acarophenacidae) with a new means of phoretic attachment. Internat. J. Acarol., 43: 329-335. doi:10.1080/01647954.2017.1287216
- Xu Y., Li Y.-C., Huang B.-R., Cai M.-L., Wu J.-Q., Wu S.-Q., Zhang, F.-P. 2018. First record of the genus Paracarophenax (Acari: Acarophenacidae) from China, with description of a new species. Syst. Appl. Acarol., 23: 2411-2419. doi:10.11158/saa.23.12.10



2021-02-05
Date accepted:
2021-04-25
Date published:
2021-04-29
Edited by:
Baumann, Julia

This work is licensed under a Creative Commons Attribution 4.0 International License
2021 Khaustov, Alexander A. and Abramov , Vladimir V.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



