Biological effects of three bacterial species on Tetranychus urticae (Acari: Tetranychidae) infesting eggplant under laboratory and greenhouse conditions
Al-Azzazy, Mahmoud M.1 ; Alsohim, Abdullah S.2 and Yoder, Carl E.3
1Department of Plant Production and Protection, College of Agriculture and Veterinary Medicine, Qassim University, Buraidah, Saudi Arabia & Agricultural Zoology and Nematology Department, Faculty of Agriculture, Al Azhar University, Cairo, Egypt.
2Department of Plant Production and Protection, College of Agriculture and Veterinary Medicine, Qassim University, Buraidah, Saudi Arabia.
3Department of Plant Sciences, Herbert College of Agriculture, University of Tennessee, 112 Plant Biotechnology Building, 2505 E.J. Chapman Drive, Knoxville, Tennessee 37996, USA.
2020 - Volume: 60 Issue: 3 pages: 587-594
https://doi.org/10.24349/acarologia/20204390Original research
Keywords
Abstract
Introduction
The two-spotted spider mite, Tetranychus urticae Koch, is one of the most damaging pests worldwide, causing severe loss to a wide range of agricultural crops (Migeon and Dorkeld, 2009). Tetranychus urticae is also the most phytophagous species belonging to the family Tetranychidae (de Carvalho Ribeiroa et al., 2019) as it infests 3,877 host plants in both greenhouse and field crops (Migeon and Dorkeld, 2009). In most agricultural crops, the use of synthetic pesticides is the main method to control T. urticae. However, because environmental adverse effects of these pesticides, the development of pesticide resistance in the target pest, and potential impacts on biodiversity and Human health (Roobakkumar et al., 2011; Dermauw et al., 2013), alternative methods should be developed.
Biological control agents like predatory mites, parasitoids and bacterial pathogens play a key role in the natural regulation of many plant pests (Al-Azzazy, 2002, 2005; Attia et al., 2013). As a group of important natural enemies of spider mites, pathogenic bacteria exhibit diverse modes of action. These include parasitizing; competition for nutrients; production of toxins, antibiotics or enzymes; induce of systemic resistance of plants (Alsohim et al., 2014; Alsohim and Fouly, 2015). Alternative crop protection strategies have been proposed based on the use of symbiotic bacteria (Christodoulou, 2011). In addition, many bacteria species are opportunistic pathogens that may be found in natural conditions, which may become pathogenic if conditions are favorable (Boucias and Pendland, 1998).
In Saudi Arabia, some works on the biological control of the date palm dust mite, Oligonychus afrasiaticus (McGregor), using two bacterial isolates of Pseudomonas fluorescens under laboratory conditions were developed (Alsohim and Fouly, 2015). However, no study to date has examined the potential of Acinetobacter sp., B. subtilis and B. qassimus on T. urticae. Acinetobacter sp., essentially found on plant roots, has been studied because of its effects on plant growth (Zamin et al., 2011). Until 1996, the role of Acinetobacter in pest control had only been demonstrated in some species, such as the stable fly, Stomoxys calcitrans (Linnaeus), and the Greater Wax Moth Larvae, Galleria mellonella Linnaeus (Lal and Khanna, 1996). These bacteria are able to rapidly penetrate tissue barriers and disperse throughout the whole body of the host (Favia et al., 2007; Crotti et al., 2009). It was reported that some strains of the genus Bacillus produce crystalline proteins with miticide properties, by stopping mite feeding and consequently leading to death of the mite Psoroptes cuniculi (Delafond) (Dunstand-Guzmán et al., 2015). The aims of the present study were to determine the effect of three bacterial species: Acinetobacter sp. (concentration 2.237×108 cfu/ml), B. subtilis (concentration 2.470×108 cfu/ml) and B. qassimus (concentration 3.320×108 cfu/ml) on T. urticae in both lab and greenhouse conditions (eggplants). In addition, two application methods were studied (direct sprays on mite individuals and residual activity by spraying only the leaves). Furthermore, effects of three pathogenic bacterial species were also assessed on a phytoseiid predatory mite, Phytoseius plumifer (Canestrini & Fanzago), under laboratory and greenhouse conditions.
Materials and methods
Mite's colonies
The mite colonies (T. urticae and P. plumifer) were maintained at the Acarology laboratory of Qassim University. Tetranychus urticae specimens were collected in a conventional unsprayed eggplant (Solanum melongena L.) greenhouse in the Qassim region (26.3489° N, 43.7668° E), Saudi Arabia, and reared in the laboratory at 30±2 °C and 50±5% RH for three generations on eggplant seedlings before experiments. Phytoseius plumifer specimens were collected in unsprayed (for the previous 3 months) eggplant greenhouses in the Melida district, Qassim region, Saudi Arabia. For rearing, P. plumifer specimens were placed on detached lima bean leaves (Phaseolus lunatus L.) infested with T. urticae, placed upside-down on cotton wool in Petri dishes (4.5 cm in diameter × 1.5 cm high), moistened daily. To prevent the predators from escaping, the margin of bean leaves was covered with strips of wet cotton. Contaminated fresh leaves were added to provide a continuous food supply for T. urticae.
Bacteria cultures
Bacteria were obtained from the biotechnology lab, Department of Plant Production and Protection, College of Agriculture and Veterinary Medicine, Qassim University, KSA. They were cultured in Luria-Bertani (LB) medium for 24 h at 26 °C. Afterwards, a single colony was re-cultured in LB medium overnight. All treatments were maintained in a media consisting of LB medium (10 g/L tryptone, 5 g/L NaCl and 5 g/L yeast) and incubated at 27±1 °C with shaking (250 rpm) overnight (Alsohim et al., 2014). Optical density was measured using a standard spectrophotometer. The number of colony forming units (CFU) was calculated using the formula: cfu/ml = (no. of colonies x dilution factor) / volume of culture plate.
Laboratory trials – residual effects on T. urticae
The host plant used in these bioassays was eggplant (Solanum melongena L.). Eggplants (three to seven-leaf stage) were first sprayed directly with 5 ml of the bacterial preparations of Acinetobacter sp. (concentration 2.237×108 cfu/ml), B. subtilis (concentration 2.470×108 cfu/ml) and B. qassimus (concentration 3.320×108 cfu/ml) using a hand sprayer. In the control treatment, only water was sprayed. Randomly selected eggplant leaves were removed from the plants and let at air conditions during 30 minutes for drying. Leaf discs of 5 cm diameter were cut and placed on saturated cotton wool (0.5 cm thick) in covered Petri dishes (4.5 cm in diameter × 1.5 cm high). Ten adult females of T. urticae were added on each disc. To prevent T. urticae escape, a ring of Vaseline was placed around the leaves. Water was added daily to the Petri dishes to keep cotton moist. The Petri dishes were placed in transparent plastic boxes (40 x 25 x 14 cm), covered with lids and placed in an incubator at 30±2 °C, 65±5% RH and a photoperiod of 16 H light: 8 H dark. Dead mites within 24 h after their transfer were removed and not included in the statistical analyses. Leaf discs were checked daily and the number of dead mites recorded during seven days. Bacteria occurrence was confirmed by stereomicroscope (SD30, Olympus, Japan) examination of mite cadavers.
Laboratory trials – direct effects on P. plumifer
To obtain same aged cohorts of eggs, fifty gravid females of P. plumifer taken from the stock culture were placed in the rearing units with about 1,000 spider mites as prey. These mite rearing units were maintained at 30±2 °C and 50±5% RH. After 24 hours, eggs laid by P. plumifer were transferred individually to new arenas, and the newly hatched larvae were supplied with T. urticae as prey; appropriate densities of prey nymphs (20 individuals of T. urticae) were added daily to the rearing units. Twenty adult females of the same age of P. plumifer were transferred on a lima bean leaf disc (5 cm. in diameter), placed on moist cotton in a Petri dish (Tree Tangle foot® Insect Barrier or a ribbon of moist cotton was put on the rim of leaf discs to prevent escape) and sprayed directly with 5 ml of Acinetobacter sp. (concentration 2.237×108 cfu/ml), B. subtilis (concentration 2.470×108 cfu/ml) and B. qassimus (concentration 3.320×108 cfu/ml) using a hand sprayer. Three replicates were performed for each bacterial treatment and 20 adult females were considered per replicate. Female mortality was recorded 72 h after spraying. A control was included (females were sprayed with water only). Bacteria occurrence was confirmed by stereomicroscope examination of predatory mite cadavers.
Greenhouse trial – effects on T. urticae
A greenhouse trial was conducted from March to April, 2018 at the Agricultural Research Station, College of Agriculture and Veterinary Medicine, Qassim University, Al-Mulida district at 34-36±2 °C and 45-65±5 % RH. The experiment was conducted on young eggplant seedlings (at the seven leaf stage). Plants were cultivated in a mixture of vermiculite and soil in plastic pots (9.2 cm diameter top, 6.3 cm diameter base and 9 cm depth; capacity: 0.37 liter). About 800 individuals of T. urticae were collected from the stock culture and 50 adults were placed on each plant seedlings. Four plant seedlings were considered for each modality (three bacterial applications and control treated with water). Just after the infestation, bacterial pathogens were applied (with approximately 10 ml of bacterial suspension) using a hand sprayer at the following concentrations: 2.237×108 cfu/ml, 2.470×108 cfu/ml and 3.320×108 cfu/ml for Acinetobacter sp., B. subtilis and B. qassimus, respectively. Observations were made, every day for seven days, by taking randomly 3 leaves, one from the top, one from middle and one from the bottom levels of each plant. All motile stages of T. urticae were counted on each leaf under a stereomicroscope and the average number of mites found on the three leaves (per plant) was considered as the number of mites/leaf/plant. Observations with stereomicroscope (SD30, Olympus, Japan) were carried out to detect bacteria on mite cadavers.
Greenhouse trials – effects on P. plumifer
Under the same conditions as those mentioned above, 240 adult females of the same age of P. plumifer were transferred to eggplant seedlings infested with T. urticae as food source (20 females transferred per seedling; three seedlings/replicate for each treatment) and sprayed directly with 10 ml of Acinetobacter sp. (concentration 2.237×108 cfu/ml), B. subtilis (concentration 2.470×108 cfu/ml) and B. qassimus (concentration 3.320×108 cfu/ml) using a hand sprayer. Female mortality was recorded 72 h after spraying. A control was included using the same number of females and sprayed by water only.
Statistical analysis
The reduction percentages of the population density of T. urticae were calculated using the equation proposed by Henderson & Tilton (1955):
Mortality % = 1- (n in C before treatment × n in T after treatment/ n in C after treatment × n in T before treatment) ×100. Where: n = pest mite population, T= treated, C = control
Statistical differences in mortality mean rates between the treatments were assessed using ANOVA followed by Duncan's Multiple Range test (DMRT).
Results and Discussion
Laboratory efficacy on T. urticae
Bacillus subtilis and B. qassimus caused similar reductions in mite populations, seven days post treatment (72.22 and 70.74%, respectively), whereas mortality was significantly higher when mites were sprayed with Acinetobacter sp. The same trend was observed during the successive days. Acinetobacter sp. reached the highest effect on T. urticae three days after treatment (mite mortality of 87.15%) (Table 1). It thus seems that Acinetobacter sp. caused significantly higher mortality on the two- spotted spider mites infesting eggplant plants than B. subtilis and B. qassimus.
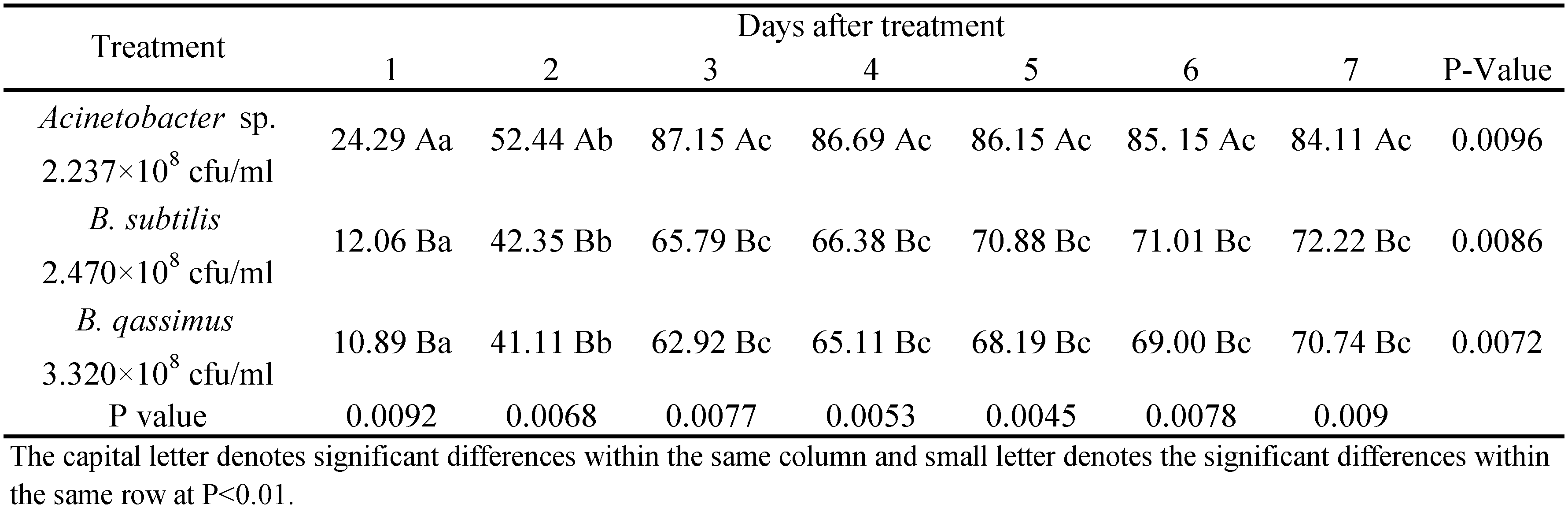


Greenhouse efficacy on T. urticae
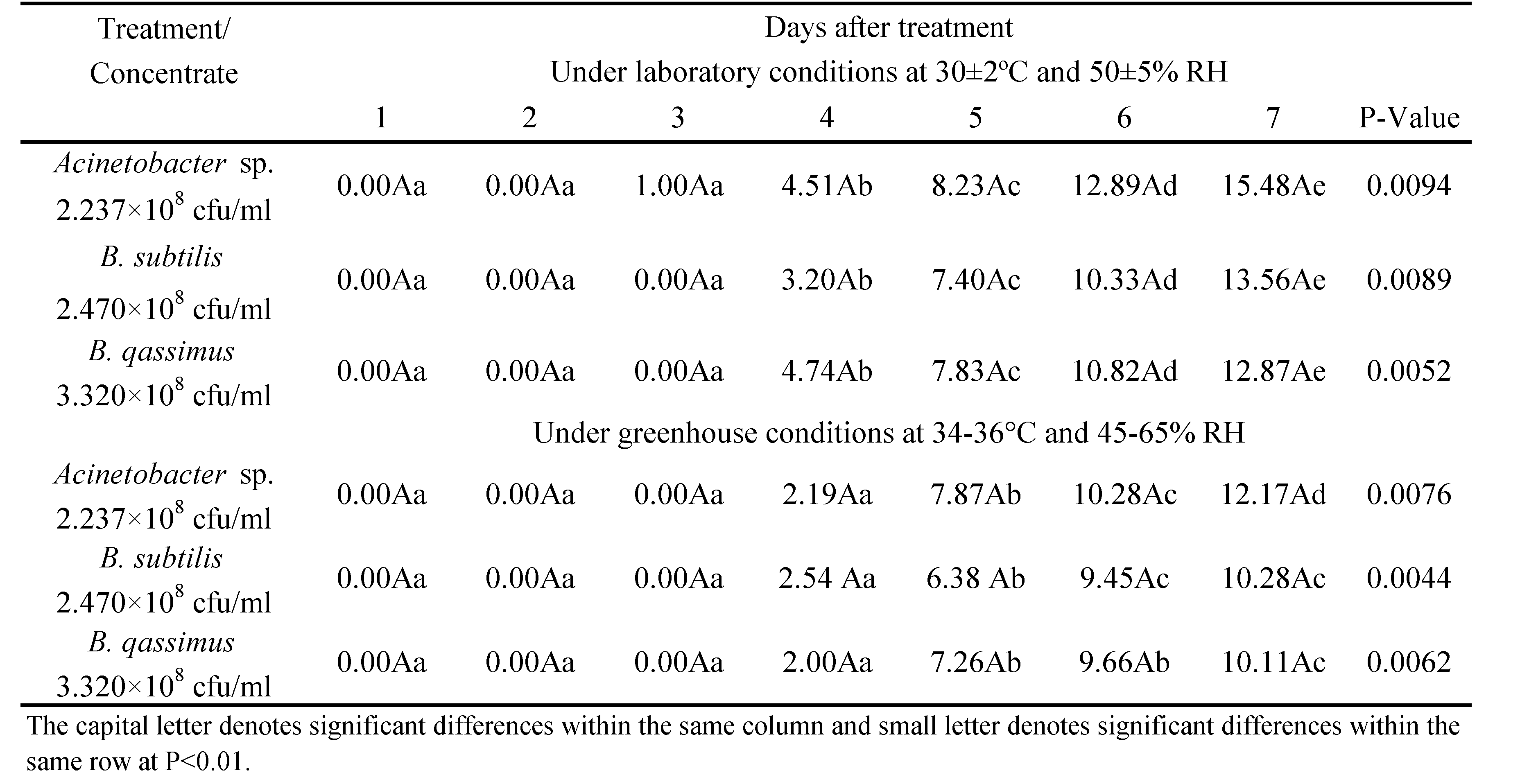
The number of mobile stages was counted every day for seven days to monitor the accumulative reduction of mites' population. The mortality rates increase till the day 3 for Acinetobacter sp treatment, whereas for the B. subtilis and B. qassimus treatments, an increase in mortality rate was observed till the day 7. Statistical analysis demonstrated that the reduction percentages of T. urticae populations were significantly different among the three treatments (P < 0.001). Results indicate that the application of Acinetobacter sp. might be considered a promising control agent against T. urticae as it caused a population reduction of 77.29% during a three days' period following the application. B. subtilis and B. qassimus caused remarkable less reduction compared to Acinetobacter sp. (67.11% for B. subtilis and 65.19% B. qassimus, seven days after the treatment) (Table 2).
Comparable results were obtained by Alsohim and Fouly (2015), who stated that the direct application of Bacillus thuringiensis and Pseudomonas fluorescens on the date palm dust mite, O. afrasiaticus, caused a mite mortality of 93.8% and 85.8%, respectively. Moreover, Hall et al. (1971) mentioned that the β-exotoxin fraction of B. thuringiensis caused high mortality in adult mites of the citrus red mite, Panonychus citri (McGregor). Eggs and immature stages were similarly affected. The findings of Hornsey et al. (2013) and Andrea et al. (2019) are also similar to the present results, in suggesting that the pathogenic bacteria Acinetobacter has high virulence potential on Galleria mellonella. In the present study, we can hypothesize that the high mortality caused by Acinetobacter sp., B. subtilis and B. qassimus could possibly provide solutions for pests' resistance to pesticides and decrease pesticide use. However, further studies on field efficacy have to be conducted for defining the application conditions allowing a successful biocontrol of T. urticae. Growers in the Qassim region, Saudi Arabia, where this pest causes high losses (up to 90% on eggplant crops), spend as much as \$100 per acre to manage this pest. It would thus also be interesting to analyze the cost of bacterial applications compared to other solutions.



Symptoms of T. urticae with a bacterial infection
Bacterial pathogens invade their hosts mostly through the mouth and alimentary canal. Less often, they enter through peritremes and wounds in the integument (Poinar and Poinar, 1998). Bacterial infections may be classified as toxaemia, when bacteria are confined in the alimentary canal where they produce toxins, bacteremia, when bacteria multiply in the hemolymph of the mite without producing toxins (apparently unaffected by the mite immune system), or septicaemia, when bacteria multiply in the hemocoel, producing toxins and killing the mite (Tanada and Kaya, 1993). In the present study, many of these factors might have contributed to the mortality of the two-spotted spider mite. Diagnostic features considered as general characteristics of bacterial infection were recorded in the current study: distinct color changes (dark brown color with a black tinge), degeneration of internal tissues, discontinuation of feeding, inability to move, weakening of the outer skin, females failing to oviposit, excretion of diarrhoea-like faeces, occasionally causing mites to become glued to the substrate, vomiting and cadavers becoming black due to the bacterial infection (Figure 1).In another study (Wilson et al., 2002) reported that bacterial hemolysins are exotoxins that attack blood cell membranes and cause cell rupture and body swelling. These hemolysins may also be involved in the pathogenicity of the three bacterial species on T. urticae, especially given the rapid mortality observed.



We can hypothesize that Acinetobacter sp. was found to be a better bacterial agent under greenhouse and lab experiments, because of its higher colonizing capacity in small-bodied mites by providing very strong adhesion to the cuticle (Van der Geest et al., 2000). Future studies would be needed to understand interactions between mites and these bacterial pathogens.
Effect of three bacterial species on the predatory mite Phytoseius plumifer
The three pathogenic bacteria studied have no different significant effects on P. plumifer (Table 3). The corrected mortality percentages were 15.48, 13.56 and 12.87% under laboratory conditions and 11.17, 10.28 and 10.11% under greenhouse conditions after one week of exposure. The pathological manifestations in P. plumifer recorded during this study are as follows: the predators became very pale and so thin that they became translucent. Those results, in general, agree with the studies carried out by other authors (i.e. Hess and Hoy, 1982). Some adult females were plumb and had a cream-color to pink rectal plug that extruded from their posterior end and occasionally caused mites to become glued to the substrate.


Conclusion
The present results show that although Acinetobacter sp., B. subtilis and B. qassimus were harmful to T. urticae, they were relatively harmless to the predatory mite P. plumifer. We hypothesize that the three pathogenic bacteria studied might affect T. urticae via ingestion, via contact while crawling on sprayed leaves or by direct reception of spray droplets. Higher activity of pathogenic bacteria towards T. urticae can be explained by the ability of pathogenic bacteria to penetrate their thin body wall, inter-skeletal membranes or through body openings. In the case of P. plumifer, the thicker exoskeleton (Duso and Fontana, 2002; Zannou et al., 2007) may prevent penetration of pathogenic bacteria into body tissues. Furthermore, predatory mites usually do not eat on plants, which can have limit bacteria entrance (McMurtry et al., 2013).
Many vegetable crop growers throughout the world are now exploring bacterial potential applicability due to ongoing pesticide restrictions. The results of the present study indicated that although all three bacterial species lead to significant mortality to T. urticae, the pathogenic bacteria of Acinetobacter sp. seems to be the most promising candidate for biocontrol of the two-spotted spider mite. Furthermore, high selectivity towards T. urticae versus P. plumifer was remarked and can be considered as a positive aspect for integrated pest management (IPM). Future research should, however, be carried out for using more bacterial species, especially species that are similar to Acinetobacter and to determine how the predators can be affected if fed with infested preys. This may help in developing an IPM program that can minimize environmental pollution. This study is a preliminary step for future work which will help policy decision makers meet agricultural product safety standards and maintain ecological balance.
References
Al-Azzazy M.M. 2002. Studies on mites associated with olive trees [M Sc Thesis]. Cairo. Faculty of Agriculture, Al-Azhar University. pp. 84.
Al-Azzazy M.M. 2005. Integrated management of mites infesting mango trees [Phd Thesis]. Cairo. Faculty of Agriculture, Al-Azhar University. pp. 122.
Alsohim A.S., Fouly A.F. 2015. Biological effects of two bacterial isolates and mutants of Pseudomonas fluorescens on date palm red spider mite Oligonychus afrasiaticus (Acari: Tetranychidae). Egypt. J. Biol. Pest. Co., 25: 513-518. http://www.esbcp.org/index.asp ![]()
Alsohim A.S., Taylor T.B., Barrett G.A., Gallie J., Zhang X.X., Junqueira A.E., Johnson L.J., Rainey P.B., Jackson R.W. 2014. The bio surfactant viscosin produced by Pseudomonas fluorescens SBW25 aids spreading motility and plant growth promotion. Environ. Microbiol., 16: 2267-2281. doi:10.1111/1462-2920.12469 ![]()
Andrea A., Krogfelt K.A., Jenssen H. 2019. Methods and Challenges of Using the Greater Wax Moth (Galleria mellonella) as a Model Organism in Antimicrobial Compound Discovery. Microorganisms, 7: 85. doi:10.3390/microorganisms7030085 ![]()
Attia S., Grissa K.L., Lognay G., Bitume E., Hance T., Mailleux A.C. 2013. A review of the major biological approaches to control the worldwide pest Tetranychus urticae (Acari: Tetranychidae) with special reference to natural pesticides. J. Pest. Sci., 86: 361-386. doi:10.1007/s10340-013-0503-0 ![]()
Boucias D.G., Pendland J.C. 1998. Principles of insect pathology. Kluwer Academic Publishers, Boston, USA. pp. 7. doi:10.1007/978-1-4615-4915-4 ![]()
Christodoulou M. 2011. Biological vector control of mosquito-borne diseases. Lancet Infect Dis., 11: 84-85. doi:10.1016/S1473-3099(11)70017-2 ![]()
Crotti E., Damiani C., Pajoro M. et al., 2009. Asaia, a versatile acetic acid bacterial symbiont, capable of cross-colonizing insects of phylogenetically distant genera and orders. Environ. Microbiol., 11: 3252-3264. Epub 2009 Sep 4. doi:10.1111/j.1462-2920.2009.02048.x ![]()
de Carvalho Ribeiro N., da Camara C.A.G., de Melo J.P.R., de Moraes M.M.2019. Effect of the essential oil from the latex of the fruit Mangifera indica L. on Tetranychus urticae Koch (Acari, Tetranychidae). Acarologia, 59 (3): 335-347. doi:10.24349/acarologia/20194333. ![]()
Dermauw W., Wybouw N., Rombauts S., Menten B., Vontas J., Grbic M., Clark R.M., Feyereisen R., Van Leeuwen T. 2013. A link between host plant adaptation and pesticide resistance in the polyphagous spider mite Tetranychus urticae. Proc. Natl. Acad. Sci. USA, 110(2): E113-22. doi:10.1073/pnas.1213214110 ![]()
Dunstand-Guzmán E., Peña-Chora G., Hallal-Calleros C., Pérez-Martínez M., Hernández-Velazquez V.M., Morales-Montor J., Flores-Pérez F.I. 2015. Acaricidal effect and histological damage induced by Bacillus thuringiensis protein extracts on the mite Psoroptes cuniculi. Parasites & Vectors., 8:285. doi:10.1186/s13071-015-0890-6 ![]()
Duso C., Fontana P. 2002. On the identity of Phytoseius plumifer (Canestrini & Fanzago) (Acari: Phytoseiidae). Acarologia, 2: 127-136. https://www1.montpellier.inra.fr/CBGP/acarologia/article.php?id=98 ![]()
Favia G., Ricci I., Damiani C., Raddadi N., Crotti E, Marzorati M., Rizzi A, Urso R., Brusetti L., Borin S., Mora D., Scuppa P., Pasqualini L., Clementi E, Genchi M., Corona S., Negri I., Grandi G., Alma A, Kramer L., Esposito F., Bandi C., Sacchi L., Daffonchio D. 2007. Bacteria of the genus Asaia stably associate with Anopheles stephensi, an Asian malarial mosquito vector. Proc. Natl. Acad. Sci. USA, 104: 9047-9051. doi:10.1073/pnas.0610451104 ![]()
Hall I.M., Hunter D.K., Arakawa K.Y. 1971. The effect of the β-exotoxin fraction of Bacillus thuringiensis on the citrus red mite. J. Invertebr. Pathol., 18: 359-362. doi:10.1016/0022-2011(71)90038-3 ![]()
Henderson C.F., Tilton E.W. 1955. Test with acaricides against the brown wheat mite. J. Econ. Entomol., 48: 157-161. doi.org/10.1093/jee/48.2.157. doi:10.1093/jee/48.2.157 ![]()
Hess R.T., Hoy M.A. 1982. Microorganisms associated with the spider mite predator Metaseiulus (=Typhlodromus) occidentalis: electron microscope observations. J. Invertebr. Pathol., 40: 98-106. doi:10.1016/0022-2011(82)90041-6 ![]()
Hornsey M., Phee L., Longshaw C., Wareham D.W. 2013. In vivo efficacy of telavancin/colistin combination therapy in a Galleria mellonella model of Acinetobacter baumannii infection. Int. J. Antimicrob. Agents., 41: 285-287. doi:10.1016/j.ijantimicag.2012.11.013 ![]()
Lal B., Khanna S. 1996. Degradation of crude oil by Acinetobacter calcoaceticus and Alcaligenes odorans. J. Appl. Bacteriol., 81: 355-362. doi:10.1111/j.1365-2672.1996.tb01926.x ![]()
McMurtry J.A., Moraes G.J. De, Sourassou N.F. 2013. Revision of the lifestyles of phytoseiid mites (Acari: Phytoseiidae) and implications for biological control strategies. Syst. Appl. Acarol., 18(4): 297-320. doi:10.11158/saa.18.4.1 ![]()
Migeon A., Dorkeld F. Spider Mites Web: a comprehensive database for the Tetranychidae [Internet]. [15 June 2009]. Montpellier: INRA/CBGP; [25 Sept 2009]. Available from: http://www1.montpellier.inra.fr/CBGP/spmweb ![]()
Poinar R., Poinar G. 1998. Parasites and pathogens of mites. Annu. Rev. Entomol., 43: 449-469. doi.org/10.1146/annurev.ento.43.1.449 doi:10.1146/annurev.ento.43.1.449 ![]()
Roobakkumar A., Babu A., Kumar D.V., Rahman V.J., Sarkar S. 2011. Pseudomonas fluorescens as an efficient entomopathogen against Oligonychus coffeae Nietner (Acari: Tetranychidae) infesting tea. Int. J. Nematol. Entomol., 1:117-120.
Tanada Y., Kaya H.K. 1993. Insect Pathology. Academic Press, San Diego, Publisher. pp. 666. Available from: https://www.elsevier.com/books/insect-pathology/tanada/978-0-08-092625-4. ![]()
Van der Geest L.P.S., Elliot S.L., Breeuwer J.A.J., Beerling E.A.M. 2000. Diseases of mites. Exp. Appl. Acarol., 24: 497-560. doi:10.1023/A:1026518418163 ![]()
Wilson M., McNab R., Henderson B. 2002. Bacterial Disease Mechanisms: An Introduction to Cellular Microbiology. Cambridge University Press, UK, Publisher. pp. 656. doi:10.1017/CBO9781139164665 ![]()
Zannou I.D., Moraes G. J., Ueckermann E.A., Oliveira A.R., Yaninek J.S., Hanna R. 2007. Phytoseiid mites of the subtribe Amblyseiina (Acari: Phytoseiidae: Amblyseiini) from sub-Saharan Africa. Zootaxa.,1550: 1-47. doi:10.11646/zootaxa.1550.1.1 ![]()
Zamin F.R., Sachdev D., Pour N.K. Chopada, A.B., 2011. Characterization of plant growth promoting traits of Acinetobacter species isolated from rhizosphere of Pennisetum glaucum. J. Microbiol. Biotechnol., 21: 556-566. doi:10.4014/jmb.1012.12006 ![]()



2020-06-08
Date accepted:
2020-08-17
Date published:
2020-09-01
Edited by:
Tixier, Marie Stephane

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Al-Azzazy, Mahmoud M.; Alsohim, Abdullah S. and Yoder, Carl E.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



