House dust mites (Acari: Astigmata) from mattresses in Panama
Lezcano, Juan J.1 ; Murgas, Ingrid L.2 ; Barrera, Olga M.3 and Miranda, Roberto J.4
1✉ Instituto Conmemorativo Gorgas de Estudios de la Salud, Departamento de Investigación en Entomología Médica, Panamá.
2Instituto Conmemorativo Gorgas de Estudios de la Salud, Departamento de Investigación en Entomología Médica, Panamá.
3Instituto de Neumología y alergias, Clínica Hospital San Fernando, Ciudad de Panamá, Panamá.
4✉ Instituto Conmemorativo Gorgas de Estudios de la Salud, Departamento de Investigación en Entomología Médica, Panamá.
2020 - Volume: 60 Issue: 3 pages: 576-586
https://doi.org/10.24349/acarologia/20204386Original research
Keywords
Abstract
Introduction
Domestic mites are microscopic arachnids that occur in various households microhabitats and are traditionally grouped according to their ecological preferences as house dust mites (Pyroglyphidae), and storages mites (Acaridae, Glycyphagidae, Echimyopodidae and Chortoglyphidae (Arlian et al., 1992; Miranda et al., 2002; de Oliveira et al., 2003; Navarro et al., 2008; Colloff, 2009; Thomas, 2010). Abiotic factors such as high levels of humidity (above 75%), and temperatures (ranging from 25 to 30 °C) are ideal conditions for the proliferation of mite populations (Arlian, 1992; Colloff, 2009). Moreover, social practices or cleaning behavior may also affect the composition of the diversity of the mites that inhabit homes (Herbosa and García, 2008; Colloff, 2009).
Fecal pellets of mites are the main source of allergens in house dust and may cause allergic rhinitis, asthma, conjunctivitis and atopic dermatitis (Fernández-Caldas et al., 2014; Gandhi et al., 2013). The feces of these mites contain a wide variety of allergenic compounds, mainly proteins, that triggers allergic respiratory disease (Sánchez-Borges et al., 2017). Sensitization occurs in the respiratory tract and on mucous membranes when mite feces are inhaled.
It has been estimated that 2-10% of the global human population suffers some type of allergy to dust mites (Sánchez-Borges et al., 2017; Ling et al., 2018). Moreover, a global initiative to assess future trends in the prevalence and severity of allergic diseases through International Study of Asthma and Allergies in Childhood (ISAAC), estimates that cases of allergic rhinitis and rhinoconjunctivitis in Latin America are increasing each year, ranging from 12.7% for 6-7 year old to 18.5% for those over 14 years of age (Katelaris et al., 2012). In Panama, the impact of allergies to house dust is generally unknown and is typically reported as unspecified allergic rhinitis (Ministry of Health of Panama, MINSA, 2016).
Mattresses are recognized as one of the most important habitats for house dust mites (Sun and Lue, 2000; Nadchatram, 2005), since these structures provide ideal microenvironmental conditions for mite proliferation. Mites also feed upon skin scales, dander, and bodily fluids. In addition, the microclimate of the mattress is also affected during the time in which it is occupied, with an increase in temperature and relative humidity compared to the rest of the room (Colloff, 1988). Communities of mites vary between mattresses, with variation affected by characteristics of the homes (macroenvironment), quality of the mattress (microenvironment) as well as the habits of people who live in the households (Colloff, 2009).
The main Metropolitan Area in Panama includes the cities of Panama (Panama province, PA), Arraijan and La Chorrera (Panama Oeste province, PO), among other administrative areas. PO was created by law in December 2013, which has encouraged demographic and urban growth in the west side of the Panama Canal (Law Nº 119, 2013). This work was developed under the hypothesis that differences in mites community composition in mattresses between houses of PA (developed urban area) and PO (a growing urban center) depend on human movements that occurs between the two provinces. We also sought to compare our results with other published studies done in the bedrooms of houses in Panama.
Materials and methods
PA and PO comprise the principal cultural and economic center of Panama. PA has a total geographic area of 2,561 km2. The mean annual ambient temperature is 27 °C (21 to 33 °C) and the mean annual relative humidity is 83% (data from the Hydrometeorology Department of ETESA, the Electrical Transmission Company, 2015). PO is often known as ''dormitory towns'' and has a total area of 2,467.1 km2. The mean annual average is 28 °C, and ranges from 19-34 °C. The mean annual relative humidity is 80.3% (data obtained from the Hydrometeorology Department of ETESA, 2015). The average number of human inhabitants per dwelling in the PA is 3.6. However, since PO was created in 2013, this value is unknown (National Statistics and Census Institute, INEC, 2010).
In both provinces, we selected 25 houses, taking one mattresses sample per home. Sampling was carried out from 21-26 September 2015. An area of 1 square meter (1 m2) on the upper surface of the mattresses was vacuumed for 2 min (Colloff, 2009). Coffee filter paper was attached to the nozzle of the vacuum cleaner's hose. After vacuuming, the paper filter containing the dust was removed, placed in a sealed plastic bag, and labeled with the collection data. Subsequently, the nozzle was cleaned before collecting the next sample. Temperature and relative humidity data on the upper surface of the mattresses was recorded.
The samples were transported in coolers at 4 °C to the Medical Entomology Research Department of the Gorgas Memorial Institute for Health Studies (DIEM-ICGES). The method described by Fain and Hart (1986) was used to collect the mites from an aliquot of 0.1 g from each sample and expressed as mites/g of dust.
All data was tabulated in Microsoft Excel 2010. The communities of mites were described on basis of species composition and density of mites for each mattress. Diversity of mites includes species richness, relative abundance, and dominance. The comparison among mattresses by province were made using the Bray-Curtis similarity index and between provinces through the Mann Whitney-Wilcoxon's test for two independent group on Simpson dominance index. The indices were calculated, and statistical tests were carried out using the software Past version 4.03. Box plots of species richness, density of mites/g of dust were elaborated using ggplot2 in R environment. A literature review on domestic mite diversity in PA and PO was conducted.
This Project was approved for Bioethics Committee under No. 077/CBI/ICGES/14.
Results
We collected 10,225 mites with 41.54% recovered from PA samples and 58.4% obtained from PO samples. Fifteen genera and 22 morphospecies were identified, representing 15 families of mites (Table 1). Of these morphospecies, 13 were identified at the species level, five at the genus level, three at the family level, and one not identified.



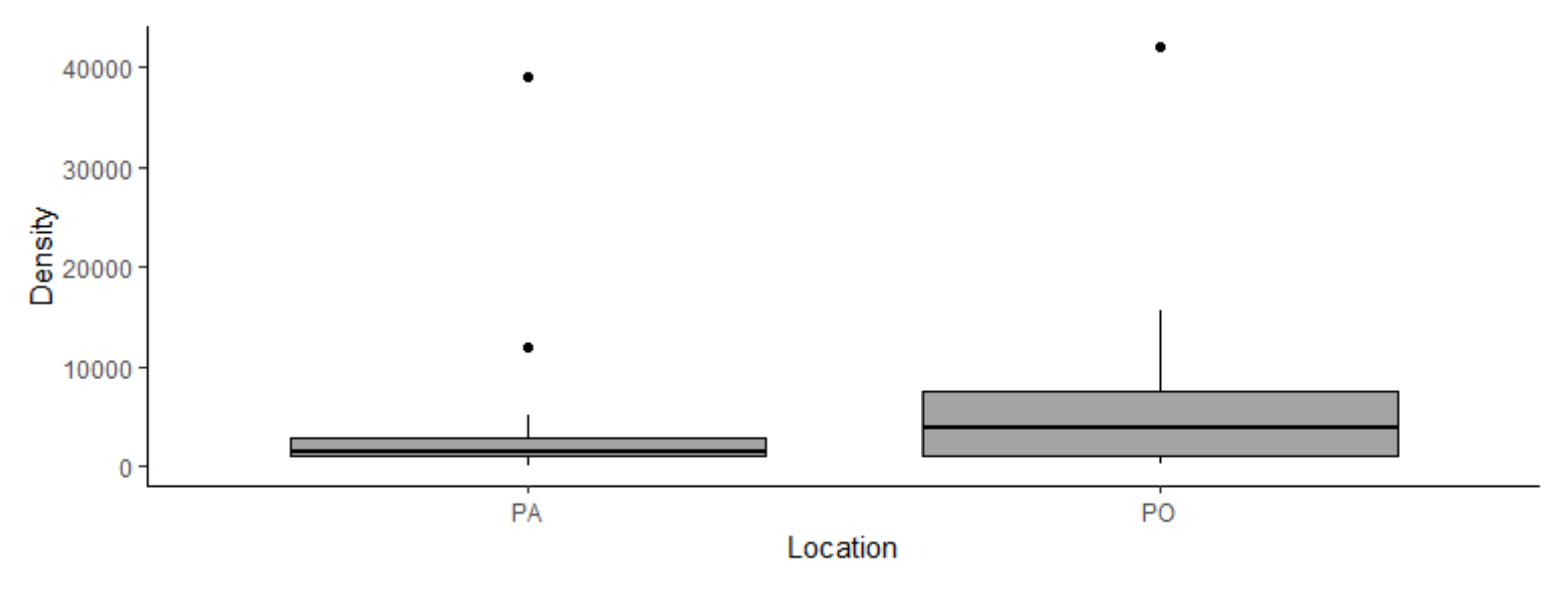
In PA mattresses, the median of species richness per mattress was 4, with a range of 1-6 species detected (Figure 1.A.). Density ranged from 150 to 39,000 mites/g of dust, with 92% of the mattresses with high densities (< 500 mites per gram of dust). The average was 3,577 ± 7,751 mites/g of dust, with a median of 1,390 mites/g of dust (Figure 2). The most abundant species in order of decreasing prevalence were Blomia tropicalis Bronswijk, Cock and Oshima, 1973, Dermatophagoides pteronyssinus (Trouessart, 1897), and Cheyletus malaccensis Oudemans, 1903 (Table 1).



In PO mattresses, the median of species richness was 3, with a range of 1-7 species detected (Figure 1.A.). The density ranged from 340 to 42,000 mites/g of dust, with an average of 6,273 ± 8,687 mites/g of dust, a median of 4,000 mites/g of dust (Figure 2). In 88% of the mattresses the density exceeded 500 mites/g of dust. The most abundant species were B. tropicalis, D. pteronyssinus, C. malaccensis, with the most prevalent species being D. pteronyssinus and B. tropicalis (Table 1).
Simpson's indices greater than 0.65 were considered positive for dominance by one species (Figure 1.B.). In seven mattresses in PA and five in PO, B. tropicalis and D. pteronyssinus were the dominant species.


A score above 0.65 in the Bray Curtis index corresponds to a high similarity between each pair of samples (mattresses). Similarities of the mattress samples are shown on dendrograms for each province (Figures 3, 4). The PA mattress samples are divided into two large groups, mainly due to the relative proportions of abundance of B. tropicalis and D. pteronyssinus. In contrast, the grouping of the samples in PO was influenced by the presence of rare taxa (less abundant and less prevalent).






Discussion
In our study, we found no significant differences between the mite communities inhabiting the mattresses of PA and PO. This is consistent with the fact that both provinces share similar climatic and microclimatic conditions, making both areas ideal for the proliferation of mites (Lafosse Marin et al., 2006; Navarro et al., 2008; see Table 2).
Regarding the density of mites/g of dust, our results indicated an average density higher in PO (6,273 mite/g of dust) than PA (3,577 mite/g of dust). PO continues to be a dormitory town, with large part of the human population working in the PA. It is possible that this difference is due to the short time that people spend at home or alternatively to poor housekeeping practices providing more homogeneous conditions for mattresses in PO. However, further studies will be necessary to test this hypothesis.
In addition, the average densities of mites in PA and PO mattresses were relatively high and were similar to those reported for tropical countries in different parts of the world (Table 2). In both provinces, 72% of mattresses had densities greater than 1,000 mites/g of dust. According to Platts-Mills and Chapman (1987), densities above 500 mites/g of dust have been associated with the development of allergic symptoms. In this sense, our measurements of mite densities represent significant findings that may have important implications for public health. The high risk of sensitization of people in these areas deserves further study.
Regarding diversity, communities of house dust mites and other species of mites were similar between mattresses, with 85% of Astigmata mites, the group of mites known to be associated with the development of allergies (Table 1). No significant difference was found when comparing the dominance values of Simpson's indices for the mite communities in PA and PO mattresses (Mann Whitney-Wilcoxon's test) for two independent groups: PA: 13.4; PO: 12.1; U= 280; p= 0.5345. Codominance of B. tropicalis and D. pteronyssinus observed in PA and PO, has been reported in other studies with mattress mite communities in Singapore and Colombia (Fernández-Caldas et al., 1993; Chew et al., 1999). This fact demonstrates that both species are common and important components of mite communities in mattresses in the tropics.



In public health, allergic studies using the skin prick test with mite extracts and a serum IgE measurement have determined that the humans may exhibit sensitization to both B. tropicalis and D. pteronyssinus (Fernández-Caldas et al., 1993; Kidon et al., 2011). Consequently, our study is important in identifying the relevance of both species as causing allergies in tropical cities (Thomas, 2010).
Records of relative humidity inside the houses in PA have a range of 48-89%, with an average of 65.8%. In PO, the range is 50-76% and average of 65%. These values are optimal for the development and propagation of the mites. According to Mumcuoglu et al. (1999), humidity is the main limiting factor in the growth and development of mites. The average temperature of the houses studied in PA was 32 ± 3.04 °C and PO 32 ± 2.38 °C (Table 2), also ideal for mites such as D. pteronyssinus and B. tropicalis. In laboratory conditions, with the temperature of 25 °C and 75% relative humidity, B. tropicalis and D. pteronyssinus had generation times of 19.3 ± 2.5 and 22.9 ± 6.4 days, respectively (Arlian et al., 1990; Mariana et al., 1996). According to Puerta et al. (2008), the high humidity and temperature, characteristic of the tropics, are conducive for the proliferation of this species.
Mites mattresses vs bedroom floors in Panama
When comparing our results with those of previous studies conducted in Panama (Miranda et al., 2002; Murgas and Dutary, 2014), it appears that mattresses have a higher density of mites in comparison to bedroom floors. The average density in bedroom floors were 1,840 and 630 mites/g of dust in urban and rural areas of La Chorrera District (PO), respectively (Miranda et al., 2002). While, Murgas and Dutary, (2014) report a density 205 mites/g of dust in PA. The differences between mattresses and bedroom floors could also reflect differences in the methods of collection.
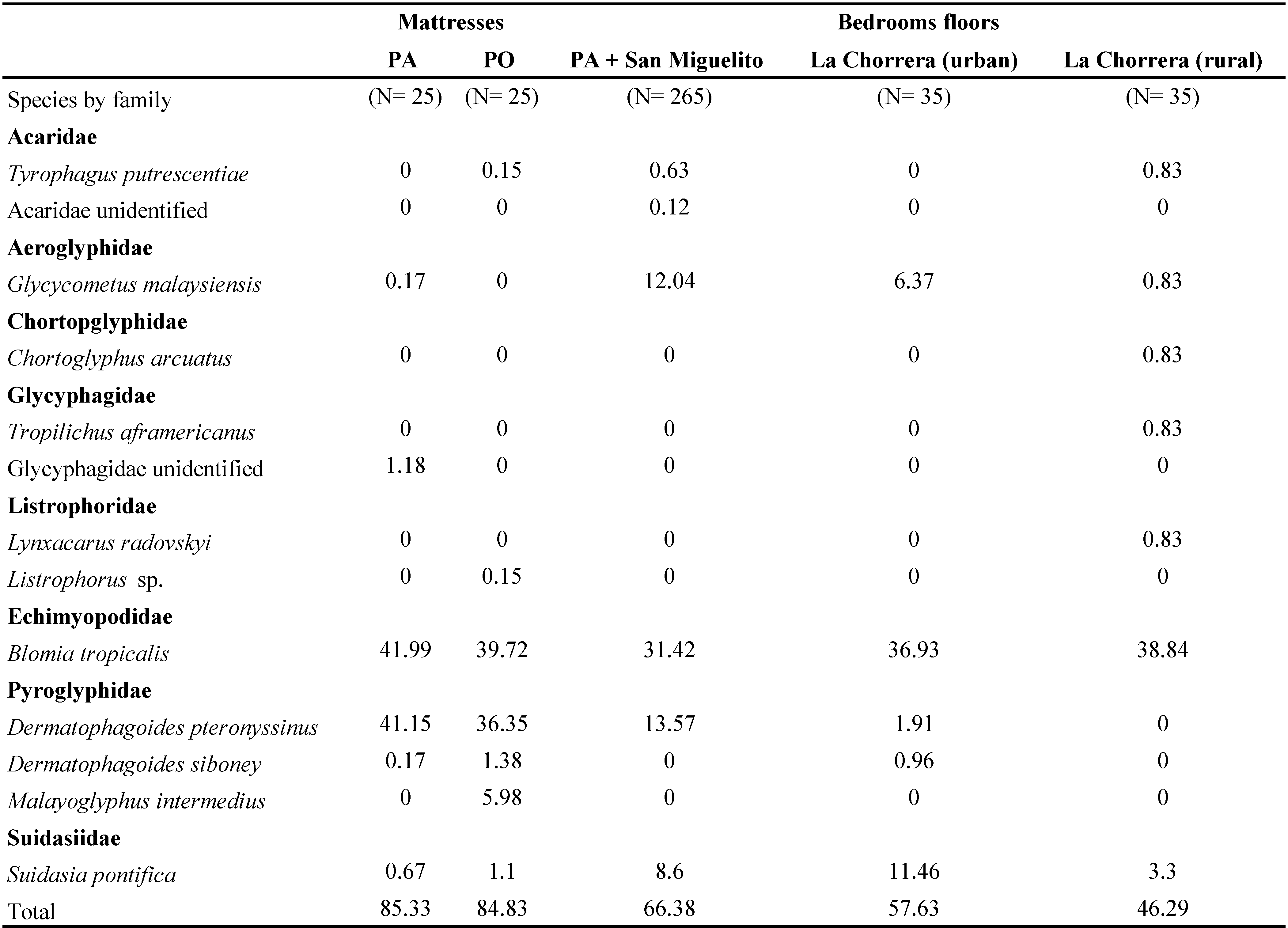
The species richness found in mattresses of PA and PO was lower than that reported by Murgas and Dutary (2014) and by Miranda et al., (2002). Even though the same Astigmata species are presented in these studies, the richness of predatory mites is higher in the bedroom floors, mainly represented by Prostigmata families: Cheyletidae, Cunaxidae, Stigmaeidae and Bdellidae. Additionally, these mites encompass almost one third of the total abundance of the individuals found in these environments. Whereas in mattresses, the predatory mites account for less than 15% of the total abundance, almost all belonging to the genus Cheyletus. Relative abundance of Astigmata mites in bedroom floors is fewer than mattresses (Table 3).


The same species were found in these microhabitat, mainly B. tropicalis and pyroglyphid mites, but they were less abundant than in mattresses, because the bedroom floors can be affected by other factors such as the frequency of cleaning the house and the type of floor in each room. Instead, the mattress it is a multi-layered structure that is not cleaned as frequently and represents an area that can retain humidity for longer periods. Thus, mattresses provide more opportunities to mites for feeding, favorable microclimates as shelters and act as reservoirs for allergens (Fernandez-Caldas et al., 2014; Wahongan et al., 2017).
In both Miranda et al. (2002), Murgas and Dutary (2014) and the present study, the most important families of mites were Echimyopodidae, Pyroglyphidae, Aeroglyphidae, Suidasiidae in the astigmatid mite communities (Table 3). D. pteronyssinus is known as the European house dust mite and has been studied extensively worldwide, since it has a wide distribution in both temperate and tropical zones (Arlian et al., 2002; Thomas, 2010). It has an important role as a sensitizing agent, besides presenting a very high cross-reactivity with other Pyroglyphidae (Ferrándiz et al., 1995; Colloff, 2009).
It is important to note that D. pteronyssinus was not the only species of Pyroglyphidae in our study, since we also found D. siboney Dusbabek, Cuervo and Cruz, 1982. This latter species has been reported in Cuba, Puerto Rico, Algeria, and Panama (Miranda et al., 2002; Colloff, 2009). Furthermore, we found Malayoglyphus intermedius (Fain, Cunnington and Spieksma, 1969), a species reported in houses in South America, Singapore, Malaysia and Indonesia (Fain et al., 1969; Mariana et al., 2000). Our results represent the first report of this species in Panama.
Regarding to B. tropicalis, despite being considered a storage mite, it is widely distributed within houses in tropical and subtropical areas and is often very abundant in house dust (Mariana et al., 2000). In addition, three other species of the so-called storage mites were found including Glycycometus malaysiensis (Fain & Nadchatram, 1980), Tyrophagus putrescentiae (Schrank, 1781) (both with a single individual), and Suidasia pontifica Oudemans, 1905. Together, these three species represented only 1% of the total identified mites.
Among predatory mites, Cheyletidae was the most common family in mattresses, represented mainly by C. malaccensis. In Mexico it was found that C. malaccensis and C. eruditus (Schrank, 1871) were frequent inhabitants in mattresses of allergic patients in six states (Fernández-Duro et al., 2013), while in Santa Marta, Colombia, Cheyletidae were the most abundant predatory mites in mattresses and pillows of allergic children (Navarro et al., 2008). Some species of Cheyletus are used as a biological control for pests in stored products (Cebolla et al., 2009). However, there are reports of cheyletid species (C. malaccensis and Chelacaropsis sp.) that are able to feed on human body fluids and cause papular urticaria (Yoshikawa, 1985; Htut, 1994).
Parasitic mites were found on mattresses, including the tick Rhipicephalus sanguineus s. l. (Latreille, 1806) known as the dog tick, a common ectoparasite found in domestic dogs and a vector of human diseases which can be found often inside of houses (Bermúdez and Miranda, 2011). Other non-parasitic species include the hair mite Listrophoridae which feeds from sebaceous gland secretions on its host hair and is explained by the presence of pets (dog and cats) inside the bedrooms (Miranda et al., 2002).
In summary, the results obtained in the present study indicates a strong presence of mites belonging to the order Astigmata which occurred at high densities in almost all mattresses reviewed. Predatory mites, mainly C. malaccensis (Order Prostigmata), only accounted for less than 15% of the total and parasitic species amounted to less than 1%.
Conclusions
Our results point to mattresses as a microhabitat that has a higher density of mites/g of dust, but a lower species richness than bedroom floors. Furthermore, the relative humidity and temperature at the regional level and even on the mattress surface indicate optimal conditions for development of mites in both PA and PO, with no significant difference recorded between mite communities in both provinces.
In the provinces of PA and PO, B. tropicalis and D. pteronyssinus were the most abundant and prevalent mites. These species are recognized for their relevance in the etiology of allergic diseases within houses. Consequently, it is clear that the exposure to the house dust mite allergens is almost inevitable inside houses, thus, it is necessary carry out studies of skin prick test or specific IgE blood test for the diagnosis and treatment of allergic symptoms in Panama. This study represents the first examination of mites in mattresses in Panama. Further research is needed to evaluate diversity of mites inside houses.
Acknowledgments
To House-dust mite project funded by the Ministry of Economy and Finance (MEF), code SINIP: 9044.043. To Ana Aguilar and Milagros Vargas for the fieldwork and Sergio Bermúdez for his suggestions and comments on the manuscript. To Victor Townsend for English review.
References
Arlian L. G. 1992. Water balance and humidity requirements of house dust mites. Exp. Appl. Acarol. 16(1-2): 15-35. doi:10.1007/BF01201490 ![]()
Arlian L. G., Rapp C. M., Ahmed S. G. 1990. Development of Dermatophagoides pteronyssinus (Acari: Pyroglyphidae). J. Med. Entomol. 27(6): 1035-1040. doi:10.1093/jmedent/27.6.1035 ![]()
Arlian L. G., Bernstein D., Bernstein I. L., et al. 1992. Prevalence of dust mites in the homes of people with asthma living in eight different geographic areas of the United States. J. Allergy Clin. Immunol. 90(1): 292-300. doi:10.1016/S0091-6749(05)80006-5 ![]()
Arlian L. G, Morgan M. S, Neal J. S. 2002. Dust mite allergens: ecology and distribution. Curr Allergy Asthma Rep. 2(5): 401-411. doi:10.1007/s11882-002-0074-2 ![]()
Bermúdez S. E., Miranda R. J. 2011. Distribution of ectoparasites of Canis lupus familiaris L. (Carnivora: Canidae) from Panama. Rev. MVZ Córdoba. 16(1): 2274-2282. doi:10.21897/rmvz.285 ![]()
Cebolla R., Pekár S., Hubert, J. 2009. Prey range of the predatory mite Cheyletus malaccensis (Acari: Cheyletidae) and its efficacy in the control of seven stored-product pests. Biological Control. 50(1): 1-6. doi:10.1016/j.biocontrol.2009.03.008 ![]()
Chew F. T., Zhang L., HO T. M., Lee B. W. 1999. House dust mite fauna of tropical Singapore. Clin. Exp. Allergy. 29(2): 201-206. doi:10.1046/j.1365-2222.1999.00493.x ![]()
Colloff M. J. 1988. House dust mite ecology in my bed. In: De Weck A.L. and Todt A.L. (eds), Mite Allergy, a Worldwide Problem. UCB Institute of Allergy, Brussels, pp. 51-54.
Colloff M. J. 2009. Dust mites. CSIRO Publishing. Australia. pp. 600. doi:10.1007/978-90-481-2224-0 ![]()
de Oliveira C. H., Binotti R. S., Muniz J. R., dos Santos J. C., do Prado A. P., de Pinho A. J 2003. Comparison of House-dust mites found on different mattress surfaces. Ann. Allergy Asthma Immunol. 91(6): 559-62. doi:10.1016/S1081-1206(10)61534-7 ![]()
Etesa. 2015. Available from: http://www.hidromet.com.pa/datos_diarios.php ![]() [15 august 2016].
[15 august 2016].
Fain A., Hart J. 1986. A new, simple technique for extraction of mites, using the difference in density between ethanol and saturated NaCl (Preliminay note). Acarologia. 27(3): 255-256.
Fain A., Cunnington A. M., Spieksma F. T. 1969. Malayoglyphus intermedius n.g., n.sp., a new mite from House-dust in Singapore and Djakarta (Pyroglyphidae: Sarcoptiformes). Acarologia. 11(1): 121-126.
Fernández-Caldas E., Puerta L., Mercado D., Lockey R. F., Caraballo L. R. 1993. Mite fauna, Der p I, Der f I and Blomia tropicalis allergen levels in a tropical environment. Clin. Exp. Allergy. 23(4): 292-297. doi:10.1111/j.1365-2222.1993.tb00325.x ![]()
Fernández-Caldas E., Puerta L., Caraballo L. 2014. Mites and allergy. In: Bergmann K-C, Ring J (Eds): History of Allergy. Chem. Immunol. Allergy. 100: 234-242. doi:10.1159/000358860 ![]()
Fernández-Duro B. I., Cuervo-Pineda N., Rodríguez-Alvízar J.A., Celio-Murillo R., Juárez-Anaya D., Pérez-Ortiz T. M. 2013. Identificación de la fauna de ácaros del polvo doméstico en colchones de pacientes alérgicos de seis estados mexicanos. Rev. Alergia Méx. 60(3): 87-92.
Ferrándiz R., Casas R., Dreborg S., Einarsson R., Fernández B. 1995. Crossreactivity between Dermatophagoides siboney and other House-dust mite allergens in sensitized asthmatic patients. Clin. Exp. Allergy. 25(10): 929-934. doi:10.1111/j.1365-2222.1995.tb00394.x ![]()
Gandhi V. D., Davidson C., Asaduzzaman M., Nahirney D., Vliagoftis H. 2013. House dust mite interactions with airway epithelium: Role in allergic airway inflammation. Curr. Allergy Asthma Rep. 13(3): 262-270. doi:10.1007/s11882-013-0349-9 ![]()
Hammer Ø., Harper D. A. T., Ryan. P. D. 2001. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontologia Electronica 4(1): 9 pp.
Herbosa R. O., García M. M. 2008. Alergias: Los ácaros del polvo doméstico. OFFARM. 27(4): 56-66.
Htut T. 1994. A Case Study of Bite Reactions in Man and Domestic Dust Samples that Implicate the House-dust Mite Predator Cheyletus malaccensis Oudemans (Acari: Cheyletidae). Indoor Environ. 3(2): 103-107. doi:10.1177/1420326X9400300208 ![]()
INEC. 2010. Censo poblacional. Available from: https://www.contraloria.gob.pa/INEC/busqueda/default.aspx?ID_PROVINCIA=08 ![]() [10 January 2016].
[10 January 2016].
Katelaris C. H., Lee B. W., Potter P. C. et al. 2012. Prevalence and diversity of allergic rhinitis in regions of the world beyond Europe and North America. Clin. Exp. Allergy. 42(2): 186-207. doi:10.1111/j.1365-2222.2011.03891.x ![]()
Kidon M. I., Chin C. W., Kang L. W., et al. 2011. Mite component-specific IgE repertoire and phenotypes of allergic disease in childhood: The tropical perspective. Pediatr Allergy and Immunol. 22(2): 202-210. doi:10.1111/j.1399-3038.2010.01094.x ![]()
Lafosse Marin S., Iraola V., Merle S., Fernández-Caldas E. 2006. Étude de la faune acarologique des matelas de l'île de la Martinique. Rev. Fr. Allergol. 46(2): 62-67. doi:10.1016/j.allerg.2006.01.008 ![]()
Law Nº 119. Official Gazette of the Republic of Panama, Panama, 30 december 2013.
Ling L., Jun Q., Ying Z., Yubao C. 2018. Domestic mite-induced allergy: Causes,
diagnosis, and future prospects. Int. J. Immunopathol. Pharmacol. 32: 1-8. doi:10.1177/2058738418804095 ![]()
Mariana A., Ho T. M., Sofian-Azirun M., Wong A. L. 2000. House-dust mite fauna in the Klang Valley, Malaysia. Southeast Asian J. Trop. Med. Public Health. 31(4): 712-721.
Mariana A., Ho T. M., Heah S. K. 1996. Life-cycle, longevity and fecundity of Blomia tropicalis (Acari: Glycyphagidae) in a tropical laboratory. Southeast Asian J. Trop. Med. Public Health. 27(2): 392-395.
Minsa. 2016. Dirección de Planificación en Salud, Departamento de Registros y Estadística en Salud. Available from: http://www.minsa.gob.pa/informacion-salud ![]() [10 January 2016].
[10 January 2016].
Miranda R. J., Quintero A. D., Almanza A. 2002. House-dust mites from urban and rural houses on the lowland Pacific slopes of Panama. Syst. Appl. Acarol. 7(1): 23-30. doi:10.11158/saa.7.1.3 ![]()
Mumcuoglu K. Y., Gat Z., Horowitz T., et al. 1999. Abundance of House-dust mites in relation to climate in contrasting agricultural settlements in Israel. Med. Vet. Entomol. 13(3): 252-258. doi:10.1046/j.1365-2915.1999.00149.x ![]()
Murgas D., Dutary S. 2014. Diversidad de ácaros (Arachnida: Acari) en habitaciones de personas alérgicas en Ciudad de Panamá, Panamá. [BSc Thesis]. Universidad de Panamá. pp. 85.
Nadchatram M. 2005. House-dust mites, our intimate associates. Tropical biomedicine. 22(1): 23-37.
Navarro J. M., Luz D., Meza M. 2008. Identificación de ácaros del polvo casero en colchones y almohadas de niños alérgicos de Santa Marta, Colombia. Duazary. 5(1): 24-31.
Platts-Mills T. A. E., Chapman M. D. 1987. Dust mites: Immunology, allergic disease, and environmental control. J. Allergy Clin. Immunol. 80(6): 755-775. doi:10.1016/S0091-6749(87)80261-0 ![]()
Puerta L., Fernández-Caldas E., Caraballo L. 2008. Ácaros domesticos. En: Asma. Ed Elizabeth Garcia y Luis Caraballo. Editorial Medica Internacional LTDA Bogota, Colombia. pp. 123 -132.
R Core Team. 2018. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available from: https://www.R-project.org/ ![]()
Sanchez-Borges M., Fernandez-Caldas E., Thomas W.R., et al. 2017. International consensus (ICON) on: clinical consequences of mite hypersensitivity, a global problem. World Allergy Organ J 10(1):14. doi:10.1186/s40413-017-0145-4 ![]()
Sun H. L., Lue K. H. 2000. Household distribution of House-dust mite in central Taiwan. J. Microbiol. Immunol. Infect. 33(4): 233-6. doi:10.1016/S0022-1759(99)00183-0 ![]()
Thomas W. R. 2010. Geography of House-dust mite allergens. Asian Pac. J. Allergy Immunol. 28(4): 211-224. doi:10.1016/j.alit.2015.05.004 ![]()
Valdivieso R., Iraola V., Estupiñán M., Fernández-Caldas E. 2006. Sensitization and exposure to House-dust and storage mites in high-altitude areas of Ecuador. Ann. Allergy Asthma Immunol. 97(4): 532-538. doi:10.1016/S1081-1206(10)60946-5 ![]()
Wahongan G. J. P., Sembel D. T., Tulung M., Satoto T. B.T. 2017. Types and density of dust mites found among different habitats in houses in North Sulawesi Province, Indonesia. 5(2): 681-685.
Wickham H. 2016. ggplot2: Elegant Graphics for Data Analysis. Springer-Verlag New York. ISBN 978-3-319-24277-4. Available from: https://ggplot2.tidyverse.org/ ![]()
Wilson J. M., Platts-Mills T. A. E. 2018. Home environmental interventions for house dust mite. J Allergy Clin. Immunol. 6(1): 1-7. doi:10.1016/j.jaip.2017.10.003 ![]()
Yoshikawa M. 1985. Skin lesions of papular urticaria induced experimentally by Cheyletus malaccensis and Chelacaropsis sp. (Acari: Cheyletidae). J. Med. Entomol. 22(1): 115-117. doi:10.1093/jmedent/22.1.115 ![]()



2019-10-29
Date accepted:
2020-07-23
Date published:
2020-08-06
Edited by:
Roy, Lise

This work is licensed under a Creative Commons Attribution 4.0 International License
2020 Lezcano, Juan J.; Murgas, Ingrid L.; Barrera, Olga M. and Miranda, Roberto J.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



