Complementary description of Podocinella misella (Berlese, 1913) (Acari: Podocinidae) and a key to world species of the genus
Santos, Jandir C.1 ; de C. Mineiro, Jeferson L.2 and de Moraes, Gilberto J.3
1✉ CNPq Researcher, Departamento de Entomologia e Acarologia, ESALQ-Universidade de São Paulo, 13418-900 Piracicaba, São Paulo, Brazil & Present address: Biodiversity Institute of Ontario, University of Guelph, Guelph, Ontario, Canada.
2APTA, lnstituto Biológico, Centro Experimental do Instituto Biológico, Lab. Acarologia, Alameda dos Vidoeiros, nº 1097. CEP 13101-680. Gramado, Campinas, São Paulo. Brasil.
3CNPq Researcher, Departamento de Entomologia e Acarologia, ESALQ-Universidade de São Paulo, 13418-900 Piracicaba, São Paulo, Brazil.
2019 - Volume: 59 Issue: 2 pages: 181-187
https://doi.org/10.24349/acarologia/20194321ZooBank LSID: 211E4B2B-5758-4DDA-B410-F9B4AAE1562B
Original research
Keywords
Abstract
Introduction
Podocinidae sensu Lindquist et al. (2009) is a small family of mesostigmatic mites, comprising only two genera: Podocinum Berlese, 1882, with thirty-one species, and Podocinella Evans & Hyatt, 1958, with five species (Yan et al., 2012; Santos et al., 2017). Podocinidae has also been used by other authors to comprise a different set of taxa, as discussed by Sourassou et al. (2015).
Podocinids are frequently found in litter samples in Brazil (Mineiro & Moraes, 2001). In a recent publication, we reviewed the species of Podocinum occurring in the country, describing a new species (Santos et al., 2017). The objective of this paper is to provide morphological information about specimens of Brazilian populations identified as Podocinella misella (Berlese, 1913), revise the concept of Podocinella as to integrate additional morphological attributes observed in this study, and finally to present a key to world species of the genus.
Material and methods
Mites were extracted from litter samples using a Berlese funnel. They were then slide-mounted in Hoyer's medium and examined under phase (Leica, DMLB) and interference (Nikon, Eclipse 80i) contrast microscopes for species identification, using the world taxonomic literature (Ho & Wang, 2009). In addition to the specimens collected during this work, we also examined specimens of the same species previously collected by other people and deposited in the reference collection of Departamento de Entomologia e Acarologia, Escola Superior de Agricultura ``Luiz de Queiroz'', Universidade de São Paulo, Piracicaba, São Paulo.
Photos of taxonomically relevant structures were taken using a digital camera connected to the interference contrast microscope. Illustrations were produced using a digital tablet and the software Adobe Illustrator®. Measurements were done with a graded ocular. For each character, the average measurement followed (in parentheses) by the minimum and the maximum measurements are given in micrometers. Setal nomenclature is based on Lindquist & Evans (1965) and Lindquist (1994) as interpreted for the Podocinidae by Yan et al. (2012). However, that assignment is tentative, pending adequate ontogenetical study of this mite group, as for example done for Zerconidae by Lindquist & Moraza (1999). Nomenclature of opisthogastric setae is different from what was proposed by Santos et al. (2017) and in accordance with what was used by Lindquist & Moraza (1999), to better match what was reported in the latter publication as well as in other publications concerning the ontogenetic development of mesostigmatid mites. Leg setal notation and chaetotactic formulae are based on Evans (1963).
The key to species was constructed taking into account the original descriptions of all species, as well as the redescription of Podocinella misella (Berlese) by Evans & Hyatt (1958) and the examination of specimens of this species collected in Brazil.
Results
Podocinella misella (Berlese, 1913)
Podocinum misellum Berlese, 1913: 83.
Podocinella misella, Evans & Hyatt, 1958: 929.
Type depository — Istituto Sperimentale per la Zoologia Agraria, Florence, Italy.
Previous records — Indonesia (Berlese, 1913 and Evans & Hyatt, 1958), Jamaica (Evans & Hyatt, 1958), Tanzania (Hurulbutt, 1972).
Specimens examined — Alagoas State: three females collected by L.R. Santos on December 11, 2003 at Teotônio Vilela. Minas Gerais State: one female collected by J.C. Santos on March 4, 2013 at Inconfidentes. São Paulo State; two females collected by J.L. de C. Mineiro on June 13, 2011 at Campinas; seven females collected by J.C. Santos on November 27, 2013 at Jaboticabal; two females collected by A.R. Oliveira on July 11, 2000 at Pariquera-Açu; eight females collected by J.C. Santos on July 4, 2013 at Piracicaba; one female collected by J.L. Munhoz on January 5, 2014 at Ipaussu. All specimens collected from litter.
Adult female: (five specimens measured)
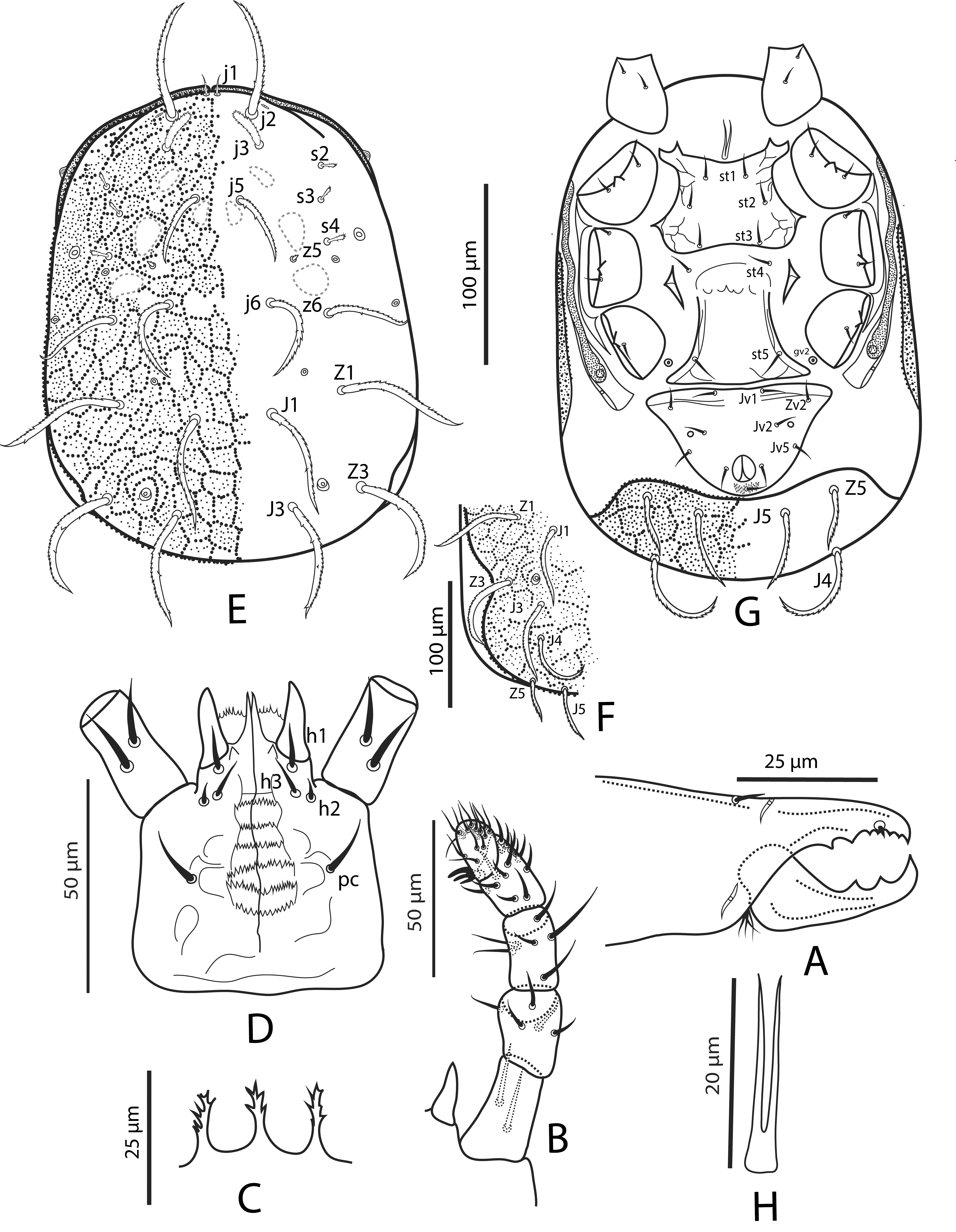


Gnathosoma — Chelicera with antiaxial and dorsal lyrifissures, and dorsal seta distinct (Fig. 1A); fixed digit 28 (28–29) long, with six teeth and a minute setiform pilus dentilis; movable digit 28 (28–29) long, with two teeth. Palp setae (Fig. 1B) (trochanter to tarsus): 2-5-6-14-15, regular for the Gamasina (Evans, 1964); all setae aciculate and smooth; apotele 3-tined. Anterior region of epistome with three distally spiculate extensions (Fig. 1C). Deutosternum with anteriormost transverse ridge smooth followed by six transverse rows of denticles, each with 12–21 denticles; corniculus horn-shaped, about 18 (18–20) long and 7 (7–8) wide basally (Fig. 1D). Measurements of setae: h1 11 (10–11), h2 6, h3 10 (9–10), pc 12 (12–13).
Dorsal idiosoma — Dorsal shield 292 (275–300) long and 212 (202–220) wide (Fig. 1E), covered by small protuberances aligned to constitute a polygonal network (Figs. 1E, 1F and 1G). Dorsal shield with a lateral notch at level of Z3 (often difficult to see when posterior end of dorsal shield is ventrally curved) (Fig. 1F). Podonotal region with ten pairs of setae (j1–j3, j5, j6, z5, z6 s2–s4) (z5 minute, usually difficult to discern), one pair of distinguishable lyrifissures (near j2) and three pairs of distinguishable pores. Opisthonotal region with seven pairs of setae ( J1, J3–J5, Z1, Z3, Z5), and two pairs of distinguishable pores. Setal lengths: j1 14 (13–14), j2 66 (65–68), j3 22 (21–24), j5 36 (35–38), j6 52 (52–53), z5 3 (3–4), z6 46 (45–47), s2 11 (11–12), s3 11 (10–11), s4 14 (14–15), J1 64 (63–65), J3 63 (61–65), J4 70 (70–71), J5 42 (41–43), Z1 71 (70–72), Z3 70 (70–71), Z5 32 (31–32). All dorsal setae stout and serrate, except j1 slender and slightly serrate (Figs. 1E, 1F and 1G).
Ventral idiosoma (Fig. 1G) — Base of tritosternum indistinguishable; laciniae 19 (18–20) long, fused at their base for about 20% of their total length (Fig.1H). Sternal shield 54 (53–55) long at mid line and 53 (50–55) wide between setae st1 and st2, mostly smooth, except for lateral lines; posterior margin slightly concave. With three pairs of setae (st1–st3); seta st4 apparently inserted on soft cuticle; lyrifissures (iv1–iv5) indiscernible. Genital shield widened posteriorly; mostly smooth, except for lateral lines; hyaline anterior region rounded and brief, not overlapping sternal shield; posterior margin truncate. Distance st5-st5 47 (46–48). Pore gv2 on unsclerotised cuticle. Ventrianal shield subtriangular, 110 (110–111) long at mid-line and 77 (75–80) wide at widest level; with four pairs of setae (Jv1, Jv2, Jv5 and Zv2) in addition to circumanal setae, and a pair of pores laterad and transversely aligned with Jv3; para-anal and post-anal setae similar in length, the former inserted at about mid-length of anal opening. Unsclerotised opisthogastric cuticle without setae. Exopodal plate apparently not fused with peritrematic plate, usually fragmented into two small and triangular strips, but some specimens with a narrow line connection them; extending forward up to posterior end of coxa II. Measurements of setae: st1 11 (11–12), st2 11 (11–12), st3 11 (11–12), st4 14 (14–15), st5 20 (19–20); Jv1 14 (14–15), Jv2 10 (10–11), Jv5 14 (13–15), Zv2 10 (10–11); para-anal 12 (11–12), post-anal 14 (14–15). All setae aciculate and smooth.
Peritreme and peritrematic plate (Figs. 1E, 1G) — Apices of peritremes nearly contiguous between setae j1. Peritrematic plate fused with dorsal shield anteriad of s2, extending posteriorly slightly behind coxa IV, and apically truncate; with a distinguishable pore next to coxa II; section posteriad of stigma with a pore and no distinct lyrifissures.
Spermathecal apparatus. Indistinct.
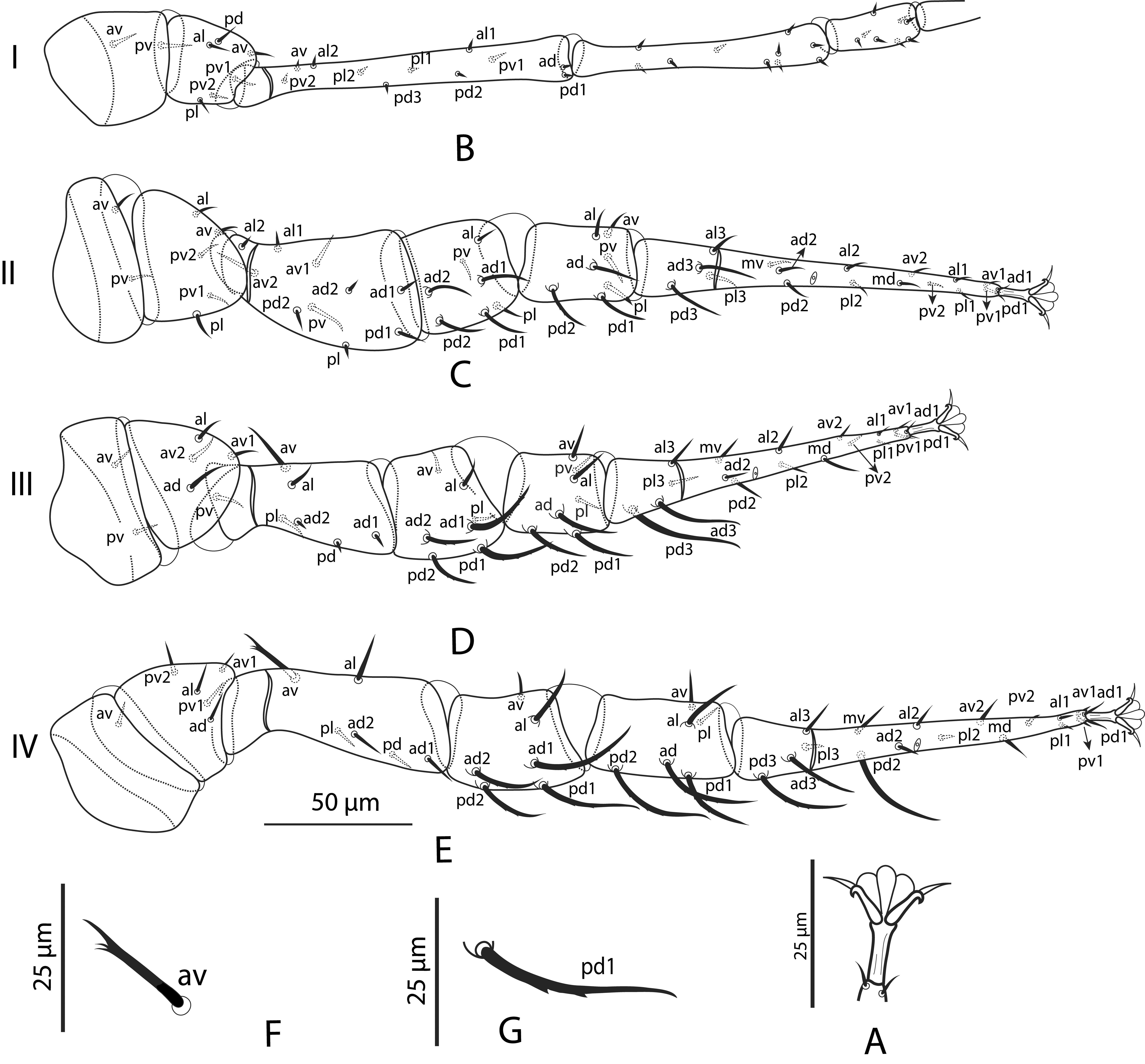
Legs (Figs. 2A–2G) — Median section of pulvilli of legs II–IV rounded; paradactyli of these legs elongate and acuminate (Fig. 2A). Lengths: I: 408 (400–415); II: 312 (300–325); III: 268 (265–270); IV: 332 (330–335). Chaetotaxy – Leg I coxa: I: 0,0/1,0/1,0; trochanter: 1,0/1,1/2,1; femur: 2, 3/1,1/2,2. Leg II coxa: 0,0/1,0/1,0; trochanter: 1,0/1,0/2,1; femur: 2,2/2,2/1,1; genu: 1,2/0,2/1,1; tibia: 1,1/1,2/1,1. Leg III coxa: 0,0/1,0/1,0; trochanter: 1,1/2,0/1,0; femur: 1,2/1,1/0,1 genu: 1,2/1,2/0,1; tibia: 1,1/1,2/1,1. Leg IV coxa: 0,0/1,0/0,0; trochanter: 1,1/2,0/1,0; femur: 1,2/1,1/0,1; genu: 1,2/1,2/0,0; tibia: 1,1/1,2/0,1. Numbers of setae on other leg I segments: genu 10 and tibia 8; given their reduce diameter, chaetotaxy could not be reliably accounted for. Number of setae on tarsus I not counted; number of setae on other tarsi: 18. Most setae aciculate and smooth (except av femur IV clearly 3-4 tined); some dorsal setae of genu, tibia and basi-tarsus of legs II-IV discreetly barbed (usually difficult to discern).
Remarks. The number and position of setae on femur I of this species is essentially the same as reported by Santos et al. (2017) for Podocinum, based on the examination of P. bengalensis Bhattacharyya, P. pacificum (Berlese), P. sagax (Berlese) and P. tupinamba Santos, Martins, Britto & Moraes, except that al2 is distinctly more anteriad of av and that pl1 and pl2 migrate ventrally.


A comparison of the specimens collected with type specimens of P. misella was not possible, because the type specimens could not be located; according Castagnoli & Pegazzano (1985) the types are missing. Evans & Hyatt (1958) provided a complementary description of this species based on specimens collected in Tomohon, Sulawesi (mentioned as N.E. Celebes), Indonesia and Jamaica. Hurlbutt (1972) presented a new complementary description based on specimens collected from Morogoro river (Tanzania) and reviewed the specimens used by Evans & Hyatt (1958) collected in Celebes.
The specimens that we examined from Brazil agree well with the redescription of Podocinella misella by Hurlbutt (1972). While his description was rather detailed, it included no setal measurements, leg chaetotaxy or details of the hypostome. Despite the missing information, we concluded that the specimens we collected belong to P. misella, because other features are sufficiently detailed, in addition to the fact that approximate setal length can be inferred from the illustrations provided and that setations of femur, genu and tibia (most variable segments between species) are provided.
New characterization of Podocinella (adult female)
Podocinella Evans & Hyatt, 1958: 926
Type species — Podocinum plumosa Evans & Hyatt, 1958, by original designation.
Gnathosoma — Anterior region of epistome with three distal extensions, spiculate or not. Hypostome with h2 much shorter than h1, h3 and pc. Deutosternum delimited by lateral lines (P. misella) or not visible (P. plumosa), usually with smooth anteriormost transverse line followed by five (P. plumosa) or six (P. misella) rows of 12–23 denticles each. Palp tarsal apotele 3-tined. Cheliceral digits similar in length; fixed and movable digits with 5–6 and two teeth, respectively.
Idiosomal dorsum. Podonotal and opisthonotal shields fused, covered with small protuberances aligned to constitute a polygonal network (not discernible in P. meghalayaensis Bhattacharyya), with or without lateral notch between Z1 and Z3. Dorsal shield with 17–23 pairs of setae (11 in the illustration of P. meghalayaensis; a few setae not discernible, according to Bhattacharyya, 1994), without supernumerary setae; seta j1 usually present (absent in P. alstoni Evans & Hyatt and P. plumosa). Soft lateral cuticle of idiosoma without setae. Dorsal setae serrate (also referred to as ciliate, plumose or pilose by various authors), stout or slender.
Idiosomal venter. Sternal shield with three pairs of setae of similar lengths (st1–st3); seta st4 inserted on metasternal plate (apparently on soft cuticle in P. misella); lyrifissures (iv1–iv3) indistinct. Genital shield bearing st5, posteriorly truncate to slightly convex. Pore gv2 (P. misella) on unsclerotised cuticle. Without metapodal plates. Ventrianal shield with three or four pairs of setae in addition to circumanal setae; para-anal and post-anal setae similar in length. soft opisthogastric cuticle with 0–3 pairs of setae.
Peritreme and peritrematic plate — Peritreme about as wide as stigma, usually extending forward beyond level of j1.
Spermathecal apparatus — Indistinct.
Legs — Leg I much longer than legs II-IV, antenna-like, terminating in a whip-like seta, without pretarsi. Legs II–IV with median section of pulvilli rounded, paradactyli elongate and acuminate.
Male — Unknown.
Remarks — The indistinct sperm access system in Podocinella and the apparent absence of males in this genus lead to the assumption that species of this genus could reproduce by thelytoky. This type of parthenogenesis has been proven by Wong (1967) for Podocinum pacificum.
Key to the world species of Podocinella (based on females)
1. Ventrianal shield with three pairs of setae (Jv5 absent) in addition to circumanal setae
...... 2
— Ventrianal shield with four pairs of setae (Jv5 present) in addition to circumanal setae
...... 4
2. Seta j1 present; with three pairs of opisthogastric setae on soft cuticle
...... Podocinella meghalayaensis Bhattacharyya 1994; India
— Seta j1 absent; without opisthogastric setae on soft cuticle
...... 3
3. Dorsal shield with 23 pairs of setae; ventrianal setae of approximately similar lengths
...... Podocinella plumosa Evans & Hyatt, 1958; Indonesia
— Dorsal shield with 18 pairs of setae; ventrianal shield with Jv5 distinctly longer than the other opisthogastric setae
...... Podocinella alstoni Evans & Hyatt, 1958; Indonesia
4.Seta j1 slightly pilose; j2 at least 4x the length of j1; z5 present
...... Podocinella misella (Berlese, 1913); Indonesia
— Seta j1 strongly pilose; j2 at least 3x the length of j1; z5 absent
...... Podocinella poetica Ishikawa, 1976; Malaysia
Acknowledgments
We appreciate very much the help of Dr. Roberto Nannelli in trying to locate the type specimens of P. misella. We thank the anonymous reviewers for their suggestions to improve the quality of this publication. This research was partially supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Projeto. 401557/2017-0 - Proc. 150755/2017).
References
Berlese A. 1913. Acari nuovi. manipoli VII–VIII. Redia, 9: 77–111.
Bhattacharyya S.K. 1994. On a collection of Podocinidae (Acarina: Mesostigmata). Records of the Zoological Survey of India, 94: 55–61.
Castagnoli M., Pegazzano F. 1985. Catalogue of the Berlese Acaroteca. Istituto Sperimentale per la Zoologia Agraria. pp. 490.
Evans G.O., Hyatt K.H. 1958. The genera Podocinum Berl. and Podocinella gen. nov. (Acarina: Mesostigmata). Annals and Magazine of Natural History, 10: 913–932. doi:10.1080/00222935708656095 ![]()
Evans G.O. 1963. Observations on the chaetotaxy of the legs in the free–living Gamasina (Acari: Mesostigmata). Bulletin of the Natural History Museum Zoology, 10: 277–303.
Evans G.O. 1964. Some observations of the chaetotaxy of the pedipalps in the Mesostigmata (Acari). Annals and Magazine of Natural History, Series 13, 6: 513–527. doi:10.1080/00222936308651393 ![]()
Hurlbutt H.W. 1972. Ascinae and Podocinidae (Acarina: Mesostigmata) from Tanzania. Acarologia, 13: 280–300.
Ishikawa K. 1976. Taxonomic investigation on mesostigmatid mites (Acarina) from Pasoh Forest Reserve, Malay Peninsula. Nature and Life in Southeast Asia, 7: 231–252.
Lindquist E.E. 1994. Some observations on the chaetotaxy of the caudal body region of gamasine mites (Acari: Mesostigmata), with a modified notation for some ventrolateral body setae. Acarologia, 35: 323–326.
Lindquist E.E., Evans G.O. 1965. Taxonomic concepts in the Ascidae, with a modified setal nomenclature for the idiosoma of the Gamasina (Acarina: Mesostigmata). Memoirs of the Entomological Society of Canada, 47: 1–64. doi:10.4039/entm9747fv ![]()
Lindquist E.E., Moraza, M.LO. 1999. Observations on homologies of idiosomal setae in Zerconidae (Acari: Mesostigmata), with modified notation for some posterior body setae. Acarologia, 39: 203–226.
Lindquist E.E., Krantz G.W., Walter D.E. 2009. Order Mesostigmata. In: Krantz G.W., Walter D.E. (Eds). A Manual of Acarology, Third Edition, Texas Tech University Press, Lubbock, Texas. pp. 124–232.
Mineiro J.L.C., Moraes G.J. 2001. Gamasida (Arachnida: Acari) edáficos de Piracicaba, Estado de São Paulo. Neotropical Entomology, 30: 379–385. doi:10.1590/S1519-566X2001000300007 https://doi.org/10.1590/S1519-566X2001000300007 ![]()
Santos J.C., Martins J.P.I., Britto E.P.J. Moraes G.J. de. 2017. A new species of Podocinum (Acari: Podocinidae) from Brazil, and supplementary descriptions of three species of this genus. Zootaxa, 4290, 3: 444–458. doi:10.11646/zootaxa.4290.3.2 ![]()
Sourassou N.F., Moraes G.J. de, Delalibera Jr. I., Correa A.S. 2015. Phylogenetic analysis of Ascidae sensu lato and related groups (Acari: Mesostigmata: Gamasina) based on nuclear ribosomal DNA partial sequences. Systematic & Applied Acarology, 20: 225–240. doi:10.11158/saa.20.3.1 ![]()
Wong C.L. 1967. A study of the biology of two species of Podocinidae (Acarina: Mesostigmata). University of Kansas Scientific Bulletin, 47: 575–600.
Yan Y., Jin D.C. Wu, D. Guo J.J., Guo X.G. 2012. A revised checklist and key to the genus Podocinum Berlese (Acari: Podocinidae) with description of a new species from Tibet, southwest China. Zootaxa, 3194: 35–48. doi:10.11646/zootaxa.3194.1.2 ![]()



2018-04-10
Date accepted:
2019-03-31
Date published:
2019-04-08
Edited by:
Faraji, Farid

This work is licensed under a Creative Commons Attribution 4.0 International License
2019 Santos, Jandir C.; de C. Mineiro, Jeferson L. and de Moraes, Gilberto J.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



