Tiny mites on a great journey – a review on scutacarid mites as phoronts and inquilines (Heterostigmatina, Pygmephoroidea, Scutacaridae)
Baumann, Julia1
1✉ Institute of Biology, University of Graz, Universitätsplatz 2, 8010 Graz, Austria.
2018 - Volume: 58 Issue: 1 pages: 192-251
https://doi.org/10.24349/acarologia/20184238ZooBank LSID: B0E18B48-D388-4E7E-89FC-08224D78E42B
Keywords
Abstract
Phoresy is a phenomenon that can be found all over the animal kingdom. There are several definitions for this behavioural trait, ranging from strict to relatively loose interpretations. What they all agree about is that it is about a small individual, the phoront, being transported by a larger animal, the host. In contrast to parasitism, the phoront does not feed on its host. In fact, in strict definitions of phoresy like that stated by Farish and Axtell (1971), the phoront does not feed at all while it is attached to its host and does also cease any other activities. According to them, the only aim of phoresy is dispersal in order to reach new habitats which are favourable for reproduction (either for the phoront’s own reproduction or that of its progeny). More recent and loose definitions of phoresy like that of Walter and Proctor (1999) don’t exclude feeding during the phoretic stage anymore.
As phoresy is about dispersal, it is no wonder that this phenomenon gets more frequent the smaller the studied species are: small animals naturally don’t have the ability to move for large distances on their own, instead they have to rely on other dispersal mechanisms like anemochory (transport by air currents), hydrochory (transport by water) or said phoresy. Mites (Acari) are characterized by consistently small body sizes (Krantz and Walter 2009), and several groups of mites have indeed developed different strategies for dispersing through phoresy. In all these mites phoresy is restricted to one stage, either the (gravid) females or deutonymphs (Krantz and Walter 2009). Some mites, like gamasid deutonymphs, do not have any special morphological adaptations to phoresy at all, but freely move on their host’s body surface. Almost all other phoretic mites possess adaptations for attaching to their hosts, like ventral sucker plates in astigmatid deutonymphs or anal pedicels secreted by uropodid deutonymphs, both allowing the mites to adhere to smooth body surfaces (Ebermann 2004). Scutacarid mites, which are the main protagonists of the present paper, cling to their host by using enlarged claws on leg I.
The mite family Scutacaridae belongs to the superfamily Pygmephoroidea, Heterostigmatina, and is characterized by remarkably small body sizes of about 200μm, a strong sexual dimorphism and a shortened generation cycle with larvae being the only juvenile stage (Walter and Proctor 1999). All members of the family are fungivorous (Binns 1979; Ebermann 1991a; Ebermann and Goloboff 2002; Jagersbacher-Baumann and Ebermann 2013), and most of them are soil inhabiting mites occurring predominantly in decaying material, often in ephemeral habitats like manure or compost (Ebermann 1991a). Following the current taxonomy, the family Scutacaridae comprises some 24 genera (Zhang et al. 2011). In almost the half of these genera, species associated with other animals can be encountered, and the host taxa range from arachnids to insects and finally to mammals (e.g. Ebermann and Palacios-Vargas 1988, Eickwort 1990, Jagersbacher-Baumann and Ebermann 2012a). Most of the respective species have been encountered phoretic on their hosts, but there are also many species which only have been found as inquilines in the nests of their hosts. These inquilines are also included in the present review.
Although several studies about scutacarids associated with different host taxa have been published, a comprehensive overview about the known associations and all involved taxa was lacking until today and will be given in the present paper. Moreover, the morphological and behavioural adaptations of scutacarids to phoresy will be described.
Phoretic behaviour is presumably always induced by a reduction of the quality of food in the current habitat (Ebermann 2004). As soon as conditions get worse, mites have to be prepared to take action. In order to successfully perform phoresy, every phoretic mite instar, irrespective of the mite taxon, must fulfil two prerequisites: it has to 1) possess the sensory capability to identify suitable hosts and 2) have the mechanical ability to attach to the host (Eickwort 1994). While the sensory equipment responsible for host identification in scutacarids is not really known yet (see below, “Mounting and dismounting of hosts”), the morphological adaptations are well understood.
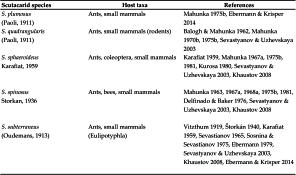
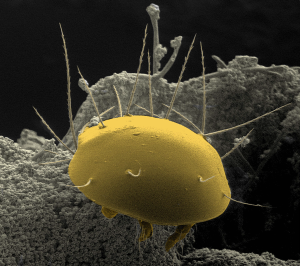
In Scutacaridae, only adult females perform phoresy, and their habitus is indeed predestined for this mode of dispersal (Figure 1). Their body is covered by curved dorsal tergits, which give them a tortoise-like appearance, and when attached to their hosts, their characteristic habitus offers practically no point of attack and thus it is difficult for the hosts to remove the mites (Ebermann 1991a). In regard of their general body shape, scutacarids are very similar to astigmatid hypopi (Anoetida, Acarida), and that is exactly what they had been mistaken for shortly upon their discovery in the 18th century (Michael 1884). In fact, a body plan similar to that described above can not only be found in Scutacaridae and astigmatid hypopi, but also in Tarsonemidae, Pyemotidae and some Uropodina (Lindquist 1975, Ebermann 2004). Scutacarids attach to their hosts by using large claws on leg I (Figure 2a) which can grasp setae or soft intersegmental skin (Ebermann 1991a). There are scutacarid species without these large claws, and these species are generally supposed not to perform phoresy. As will be seen below, there are also species in which both occur, morphs with and morphs without large claws on leg I. The detailed structure of the claws can also differ between scutacarid genera: for example, the claws of phoretic Lamnacarus females have long, thin tips which are not present in other genera (Ebermann 1991a).


In addition to their characteristic morphology, scutacarid mites can also avoid being removed by their hosts by choosing attachment sites which are difficult to reach for the (grooming) host, like under the elytra of beetles (e.g. Karafiat 1959). It is a general trend that phoretic mite taxa are not distributed randomly on their host, but gather highly localised in places that are not groomed (Eickwort 1994, Ebermann 2004). However, some scutacarid species on the contrary attach to exposed body parts of their hosts: for example, Thaumatopelvis reticulatus Ebermann, 1980 clings to setae on the head of ants (Ebermann 1980b). The exposed location demonstrates that the claws are powerful attachment structures as the mites can even withstand the grooming of the host.
In the genera Archidispus, Lamnacarus and Scutacarus, dimorphic species characterized by the occurrence of phoretic and non-phoretic females exist. This female dimorphism, which is also referred to as phoretomorphism, had firstly been suggested by Norton (1977) for Scutacaropsis baculitarsus agaricus (Norton and Ide, 1974) and had thereafter been demonstrated in other scutacarid species belonging to the genera Archidispus, Lamnacarus and Scutacarus through laboratory cultures by Ebermann (1991a,b). A similar dimorphism in connection with phoresy is also known from other mite taxa, either in deutonymphs or in adult females (Krantz and Walter 2009). To date, 11 scutacarid species are known to be dimorphic (Table 1). The main difference between the two morphs is the size of the claw on the tarsus of leg I, which is large in the phoretic morph and small or even absent in the non-phoretic morph (Figure 2). Moreover, the sclerotisation of phoretic females appears to be stronger (Ebermann 1991a). In the genus Archidispus, there are also distinct differences in the shape of particular setae between the two morphs (Ebermann 1991a,b; Figure 3). The large claws on leg I and the stronger sclerotization can easily be interpreted as adaptations to phoretic behaviour because they allow attachment and also protect the mites during their journey. The possible adaptive value of the differently shaped body setae in the genus Archidispus on the other hand remains unclear.









Laboratory cultures showed that females of both morphs can also produce female offspring belonging to both morphs. The mechanisms for morph determination are not totally clear yet, but Ebermann (1991a) convincingly hypothesized that the quality of nutrition of larvae might most likely be the determining factor. Behavioural experiments proofed that phoretic females readily attach to offered hosts, while non-phoretic females mount hosts only in exceptional cases (Ebermann 1991a).
Ebermann (1991a, 2004) stated that phoretomorphism most likely can be regarded as an adaptation to ephemeral or heterogeneous habitats. Accordingly, the phoretic morph is considered to be responsible for dispersal, and the usually smaller and weakly sclerotized non-phoretic morph is seen as an “energy saving version” specialized on reproduction: As long as conditions in the habitat are favourable, the non-phoretic females predominate and procreate quickly. Worsening conditions induce the increased development of phoretic females which mount on suitable hosts and thus disperse to new habitats. Ebermann (1992) hypothesized that differences in the degree of phoretomorphism reflect the selective pressure towards phoresy in scutacarids. This means that species with a strong degree of phoretomorphism depend on phoresy because they are specialized on certain habitat conditions, while species with a low degree of phoretomorphism do not exclusively rely on phoresy because they are also able to reproduce and thrive in suboptimal habitat conditions.
The detection of phoretomorphism had important consequences for scutacarid taxonomy as some taxa, which had been treated as separated species before, had to be merged into one single species. Accordingly, Ebermann (1990) demonstrated that Archidispus soosi (Mahunka, 1967) is the non-phoretic morph of A. armatus (Karafiat, 1959), Lamnacarus coprophilus Mahunka, 1968 is the non-phoretic morph of L. ornatus Balogh & Mahunka, 1963 and Scutacarus subfimetarius Momen & Bagoury, 1989 is the non-phoretic morph of S. longitarsus (Berlese, 1905). In the same work, Ebermann also discouraged from using Variatipes, a subgenus of the genus Scutacarus that is defined by the absence of claws on leg I. This absence of claws could indeed be a “good” character evolved by a gradual reduction of the claws, but specimens without claws could also be non-phoretic morphs of other species.
Most taxonomic problems caused by phoretomorphism arise in the genus Archidispus as the two female morphs differ extremely in this taxon. For several Archidispus species, only the non-phoretic or the phoretic morph have been described so far (Ebermann 1990), and further analyses may reveal that many of these species in reality represent the two morphs of one single species. Until now, cumbersome rearing experiments have been the only way to unequivocally identify the two morphs of a phoretomorphic species (Ebermann 1991a). Nowadays, molecular genetic methods are also available for investigating the status of different morphs, but so far no studies on the topic have been performed.
The detailed act of identifying and mounting hosts is unclear for most phoretic scutacarids. Most species that could be observed alive didn’t show any recognisable searching or appetence behaviour, but simply “adhered” to host individuals when being put together with them in plastic boxes in behavioural experiments (Schousboe 1986, Ebermann 1991a). Some scutacarids, however, display appetence behaviour: In order to locate potential new hosts, females of these species move to exposed positions and put themselves in an upright position, the “perching stance”: The first legs are held out with or without questing movements similar to what can be observed in ticks (Binns 1979). The mites stand on their extended legs IV which are normally trailed during locomotion, and they are apparently backed by their dorsal setae. The appetence behaviour described above has been reported for Scutacaropsis baculitarsus (Mahunka, 1968) by Binns (1979), for Archidispus amarae (Kurosa, 1970), A. armatus, A. bembidii (Karafiat, 1959), A. magnificus (Karafiat, 1959) and A. minor (Karafiat, 1959) by Ebermann (1991a,b), for Imparipes (Sporichneuthes) dispar Rack, 1964 by Ebermann (1995) and for the genus Lophodispus by Schlagbauer (1997) and Ebermann and Krisper (2014), and it has moreover been observed in Imparipes (I.) histricinus Berlese, 1903, Scutacarus longipes Rack, 1975 and S. longitarsus (Berlese, 1905) (Ebermann pers. comm.). In the genus Archidispus, Ebermann (1991a) noted that only phoretomorph females performed appetence behaviour, while non-phoretomorph females always fled whenever potential hosts approached.
Imparipes (S.) dispar, Lophodispus bulgaricus Dobrev, 1992 and L. irregularis (Mahunka, 1971) are the only scutacarid species which are known to mount its hosts by jumping, as has been reported by Ebermann (1995) for Imparipes and by Schlagbauer (1997) in his master thesis for Lophodispus. The jumping behaviour of I. dispar is provoked by tactile stimuli of the mite’s dorsal setae or even by air currents, and three types of jumping have been demonstrated: the aimed jump, the sideward jump and the high-and-long jump. While the latter two are interpreted as escape mechanisms for evading predators, the aimed jump is part of the mite’s phoretic behaviour as it permits I. dispar to jump onto fast-moving hosts. The two Lophodispus species show the same jumping types as I. dispar and additionally are able to jump backwards, but it is more difficult to provoke the jumping behaviour in them. Although observations indicate that leg IV in all cases plays the most important role in jumping, the detailed mechanisms of the movement are unknown: neither I. dispar nor the two Lophodispus species show clear (external) morphological differences compared to non-jumping scutacarids. Generally speaking, for successful host finding and identification, both mechanical as well as chemical stimuli are necessary. Mechanosensitive setae are located on the mites’ dorsal side as well as on their legs (Jagersbacher-Baumann 2007), and chemosensitive setae (solenidia) can be found on the legs, with legs I possessing the highest number of solenidia. The perching stance of some scutacarids, where legs I are outstretched, strongly points to a great importance of chemical cues, whereas experiments with I. dispar showed positive reactions to tactile stimuli (Ebermann 1995), suggesting that mechanical stimuli are more important here (Ebermann 2004). Different scutacarid species presumably follow different strategies in the act of host finding and identification, but detailed studies on this issue are lacking to date.
On their host, the mites usually are motionless (Karafiat 1959; pers. obs.). There is only one observation by Travis (1941) describing scutacarid mites (“Disparipedidae”) riding fire ants while standing “rather erect, bobbing up and down and tapping the ant with the first pair of legs”. Travis further describes that the mites feed on liquids on the mouthparts and occasionally also on the anal opening of the ants. The described behaviour rather resembles the appetence behaviour described above and does not fit to what is known about the feeding habits (fungivory) of scutacarids at all. As his descriptions are extraordinary, it is possible that Travis either misinterpreted the observations he made in the artificial environment of a laboratory or in fact saw mites of the family Antennophoridae (Mesostigmata) and mistook them for Scutacaridae. Another remarkable report is that of Schousboe (1986) who noted that Scutacarus acarorum (Goeze, 1780) specimens on overwintered Bombus queens discharged dark fecal pellets, something that has never been observed by other researchers afterwards.
Scutacarid mites are no parasites of their hosts, although this assumption had been stated at least in the case of the bumble bee associated S. acarorum (Chmielewski 1971) and since then has been circulating like an urban legend (Chmielewski and Baker 2008, Kontschán 2015, Kontschán et al. 2016). Indeed, Chmielewski never really observed parasitic behaviour (pers. comm. between Chmielewski and Ebermann). Experiments by Karafiat (1959) and Schousboe (1986) proofed that scutacarids are not parasitic, and the morphology of their gnathosoma with its delicate, stylet-like chelicerae also shows that they hardly could be parasites (Schousboe 1986).
The number of scutacarids phoretic on one host specimen usually is low and ranges around 10 mites per host individual (eg. Ebermann and Hall 2003, Hall and Ebermann 2005, Kurosa 2005). Some species, however, can also be found in higher numbers. Mites belonging to the genus Archidispus associated with different carabid beetles for example frequently occur in abundances of around 40 mite individuals per host specimen (e.g. Kurosa 1983, 1991, 2003). The highest numbers of scutacarids are known for A. bembidii: more than 100 individuals have been reported from under the elytra of different carabid species (Karafiat 1959, Ebermann 1991a). Generally, reports of more than 50 scutacarids on one host specimen are rather rare (e.g. 115 Scutacarus acarorum on Bombus terrestris queen (Schousboe 1986), 71 Imparipes apicola (Banks, 1914) on Andrena flavipes (Ebermann and Hall 2005)).
Dismounting of hosts has rarely been observed. Karafiat (1959) reported that Archidispus bembidii dismounted from beetles if the host was dead or if the mite was disturbed, and Binns (1979) noted that Scutacaropsis baculitarsus detached from anesthetized, decapitated flies. On the other hand, Schousboe (1986) was only in one exceptional case able to observe detachment of S. acarorum from bumble bees. He reported that apart from this case, the mites never dismounted, even if the host was dead. It is not clear which factors induce dismounting, but most probably the mites detect new habitats by chemical stimuli. “Negative stimuli” like disturbance or even death of the host seem to play a minor role in inducing dismounting. This assumption is supported by the fact that phoretic scutacarid mites can often be encountered on pinned, dried insects and also on animal material stored in ethanol (Ebermann pers. comm., pers. obs.).
Apparently the act of phoresy is risky for the mites because suitable environments cannot always be reached. Indeed, it is not too uncommon to find dead scutacarids on their host which obviously never arrived at their desired destination (Karafiat 1959, Ebermann 1991a).
As mentioned above, presumably all scutacarids are fungivorous. Only species of the subgenus Sporichneuthes of the genus Imparipes are known to feed on spores, all others suck the content of fungal hyphe (Ebermann 1998). While some species apparently are food generalists, others seem to be rather selective, feeding on only a few or even only on one fungal species (Ebermann et al. 2013). Some of the scutacarids with a constricted food spectrum developed a very rare phenomenon: they possess sporothecae. Sporothecae are defined as repositories inside the mites’ body which are used for storage and transport of fungal spores (Suski 1973, Ebermann and Hall 2003), and Jagersbacher-Baumann and Ebermann (2012a) moreover suggested that these repositories have to be filled actively by the mites. By transporting the spores of their preferred fungal species, the mites can secure food for their offspring: the spores are released and fungal hyphae start to grow as soon as the mites reach suitable habitats. Using this strategy, the mites avoid the risk of not finding their nutritional fungi when colonizing new environments (Ebermann et al. 2013).
Presumed sporothecae have been identified in four species belonging to the genera Heterodispus and Imparipes, and the two genera use different anatomical structures for transporting fungal spores (Figure 4): The African mite Heterodispus foveatus Jagersbacher-Baumann & Ebermann, 2012, associated with coleoptera and small mammals, possesses characteristic cavities in its posterior sternal plate which frequently are filled with spores and thus are interpreted as sporothecae (Jagersbacher-Baumann and Ebermann 2012a). Imparipes apicola, I. breganti Ebermann & Hall, 2004 and I. haeseleri Ebermann & Hall, 2003, all three phoretic on sphecids and wild bees, on the other hand use their genital atrium for fungal spore transfer (Ebermann and Hall 2003, 2004). So far it has not been possible to identify the fungal species transported in the sporothecae (Ebermann and Hall 2003, Ebermann et al. 2013).


Transport of fungal spores by scutacarids can also happen occasionally as the spores tend to adhere on the mites’ surface (Jagersbacher-Baumann and Ebermann 2012a), which can frequently be observed in microscopic slides. Of course, this mode of transport does neither include specialised anatomical structures nor active collection of spores on the part of the mites, so this random occurrence of spores on the mites should not be confused with real sporothecae.
Scutacarid mites occur in associations with other animal taxa because it is beneficial for them. Either the mites find favourable environmental conditions in the nests of their hosts (most likely, these conditions simply mean “availability of appropriate nutritional fungi”), or they mount their hosts in order to take advantage of their dispersal abilities so that they can reach new habitats, or both factors are true.
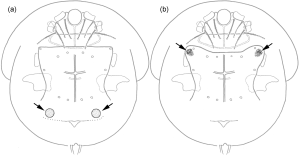
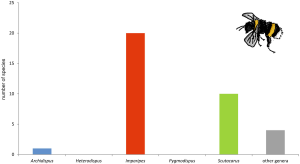
The following chapter will give an overview on (to my knowledge) all reports of associations between scutacarids and other animal taxa published until May 2017. In total, 414 species and subspecies in 17 genera have been reported from some kind of associations with other animals (reports of species listed as “near” or “cf” have been counted as separate species and are thus included in this number). Most species (n = 162) belonged to the genus Scutacarus, followed by Imparipes (125) and Archidispus (76; Figure 5). In the last published comprehensive summary of the taxonomic richness within the order Trombidiformes by Zhang et al. (2011), the family Scutacaridae is indicated to contain about 800 species in 24 genera, so it seems safe to estimate that approximately the half of all scutacarid species live in some kind of association with other animal taxa.



The following subchapters are divided into the reported host taxa, starting with Arachnida, moving to various taxa within the large group of Hexapoda, then to Aves and finally to Mammalia. Due to clarification reasons, the groups of hosts that are used in the graphs do not represent consistent taxonomic taxa, they rather are groups which combine hosts of different taxonomic levels using their common names (e.g., “ants” only describes members of one family while “mammals” covers the members of one whole class).
Anticipatory it can be stated that by far most scutacarid species can be encountered in association with ants (Formicidae) (Figure 6) and that the prevailing types of associations vary between the different host groups (Figure 7). The subchapters give information on the number of associated scutacarid species of the host taxa and, if available, details on the associations and possible biological backgrounds. The included annex is ordered according to the hosts, giving information on the associated scutacarid species, the type of association (inquiline, phoront, or both), the geographic range and occasionally other interesting remarks (for example, other known habitats of the mites, like soil or moss). In another table available in the online supplementary material, the same information is organized the other way round, listing the scutacarid species first and adding the reported host taxa for each species afterwards.






Associations between mites and other arachnids in general are rare (Ebermann 2004), thus it is no surprise that scutacarid mites also can only rarely be found in association with other arachnids (Annex I.). So far, two Scutacarus species have been reported phoretic on spiders, Araneae, of the family Nemesiidae from Argentina (Ebermann and Goloboff 2002). Another species, the usually soil inhabiting Imparipes tocatlphilus Ebermann & Palacios-Vargas, 1988 from Central and South America, has also been found phoretic on Ricinulei (Ebermann and Palacios-Vargas 1988). On spiders and ricinuleids, the scutacarids attach to the soft integument of the legs (Ebermann and Palacios-Vargas 1988, Ebermann and Goloboff 2002).
Even other mites, Acari, have been reported as phoresy hosts for scutacarids. Scutacarus talpae (Oudemans, 1913) was found by Oudemans (1913) inside of a mole’s nest, and all five encountered individuals were attached to deutonymphs of the mesostigmatid Haemogamasus ambulans. In the respective publication, Oudemans expressed his surprise as the scutacarids could neither be found on protonymphs or adults of H. ambulans nor on any other gamasid genus, all of which were available in enormous numbers. Other scutacarid species using mites as phoresy host are S. acarorum and S. deserticolus Mahunka, 1969. Both are common phoronts on gamasid Parasitellus deutonymphs (mostly P. fucorum), which are in turn phoretic on various bumble bee (Bombus) species (Rack 1964, Chmielewski 1971, Schousboe 1986, Ebermann 1991a, 2004, Schwarz and Huck 1997, Khaustov 2008). On the Parasitellus deutonymphs, the scutacarids cling to setae on body and legs (e.g. Schousboe 1986). The association between S. acarorum/S. deserticolus and Parasitellus sp. is the only example of frequent hyperphoresy in Scutacaridae. Hyperphoresy generally is an uncommon phenomenon that has apart from this example been demonstrated in mesostigmatid mites (Bajerlein and Błoszyk 2003, Szymkowiak et al. 2007) and accidentally in astigmatid hypopi (Athias-Binche 1994). Ebermann (2004) described one exceptional finding of hyper-hyperphoresy from a bumble bee: he found a scutacarid phoretic on a Parasitellus deutonymph, and the scutacarid in turn also had a hypopus of Anoetidae (Astigmata) attached.
Only one scutacarid species, Imparipes intentatus Khaustov, 2008, has been reported from Entognatha, all other hexapod hosts belong to Ectognatha. The respective host is a member of the Diplura, genus Campodea (Khaustov 2008; Annex I). As diplurans occur frequently in soil and don’t have strong dispersal abilities due to their small size, the found association between mite and diplurans may have been by chance. When extracted through Berlese-Tullgren funnels and kept alive in boxes filled with plaster of Paris, some scutacarids appear to be stressed and for short periods of time tend to attach to whatever host available (pers. obs.). In these artificial environments scutacarid individuals have even been observed to “desperately” cling to other conspecifics. Bearing this behaviour in mind, unusual reports of phoresy like that on diplurans should be taken with a grain of salt.
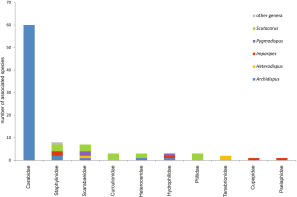
In the taxon Coleoptera, by far the most scutacarid species (62 spp.) can be detected on ground beetles, Carabidae (Figures 8-9; Annex II). All of the mites associated with Carabidae belong to the genus Archidispus, no other scutacarid genera have been found so far (e.g. Karafiat 1959, Kurosa 1977, 1980, 1989, 2005, Khaustov 2008; for other references see Annex II). Moreover, Archidispus can only rarely be seen on beetles belonging to families other than Carabidae. Only two species, A. armatus and A. bembidii, can apart from Carabidae also be encountered on Heteroceridae, Hydrophilidae and Staphylinidae (Karafiat 1959, Mahunka 1972b, Khaustov 2008), and A. irregularis Katlav & Hajinqanbar, 2016 was found only on Staphylinidae. Generally speaking, different scutacarid genera can be found on other beetle families, but among them, the genus Scutacarus is the most prominent (Figure 10).







In the following, the beetle families reported to be hosts for scutacarids will be discussed.
Within the family Carabidae, most associated Archidispus species are rather opportunistic regarding their host choice: the majority of the reported species have been found on different species of one carabid genus, and about the half of the scutacarids occurred on several different carabid genera. The scutacarids have only been reported from imagines, they have never been found on the beetles’ soil living larvae or pupae (Karafiat 1959). On their carabid hosts, the mites can be found on four typical attachment locations: the cervical membrane between head and prothorax, the intersegmental membrane between pro- and mesothorax, between thorax and abdomen, and finally under the beetle’s elytra (e.g. Karafiat 1959, Kurosa 1970, 1972a, Khaustov 2008). Some Archidispus species show no preference for special attachment sites (Kurosa 1984), while others prefer certain regions. For example, A. magnificus preferably attaches on the anterior part of the prothorax of its carabid hosts and A. sugiyamai Kurosa, 1991 on the posterior part of the prothorax (Ebermann et al. 2011). These two species can also simultaneously be found on the same host, each one on its preferred attachment site. It is common to encounter more than one Archidispus species on one host (e.g. Kurosa 2009). Only five Archidispus species found on Carabidae have also been reported from soil samples, and all of them display phoretomorphism (Table 1). The carabid beetles used for phoresy usually occur in moist environments, which are often inhabited by scutacarid species as they favour the growth of fungi. The moist habitats don’t offer stable environmental conditions as they are prone to frequent flooding, and thus the respective scutacarids strongly depend on phoresy in order to escape from flooded regions (Ebermann 1991a).
Additional to Carabidae, scutacarids in moist habitats also use ripicolous Heteroceridae and Hydrophilidae as hosts (Annex III). On these two beetle families, species belonging to the genera Archidispus, Imparipes, Pygmodispus and Scutacarus can be found (Karafiat 1959, Kurosa 1980, Messner 2001, Khaustov 2008, Ebermann et al. 2016). The respective mites have been reported from under the elytra or attached randomly on the beetles’ surface (Karafiat 1959).
Members of Staphylinidae also often occur in moist habitats, moreover on dung and on carcasses (Zahradník 1985). Mites belonging to five scutacarid genera have been found phoretic on staphylinid beetles from the Holarctic and from Sudan: Archidispus armatus, A. irregularis, Imparipes dispar, I. histricinus, Lamnacarus ornatus and 3 Scutacarus species (e.g. Karafiat 1959, Ebermann 1991a, Khaustov 2008, Truck 2012, Ebermann et al. 2016; for other references see Annex III). Most of these scutacarid species have also been recorded from other habitats like soil, moss, manure or Fragaria plants. On their host, the mites can be found rather randomly on different attachment sites like the legs, the lateral abdomen or between caput and prothorax (Karafiat 1959, Ebermann 1991a).
Scutacarids can also be found on beetle families which occur in other than moist habitats which may be suitable for the mites as well: fungus-infested wood, manure, carcasses and different types of decaying material in general.
Beetle species with an affinity to fungus-infected wood can be found in the families Curculionidae and Cupepidae (Annex III). In Curculionidae, Scutacarus scolyti Mahunka & Moser, 1980 and S. cf. culmusophilus Sevastianov, 1975 have been reported from four species of wood borer and bark beetles from Eurasia (Mahunka and Moser 1980, Khaustov 2008, Knapp 2008). The mite occurs in the galleries made by the beetles (Khaustov 2008) and also phoretic on setae on the base of the coxae (Mahunka and Moser 1980). As the galleries made by these beetles are known to contain fungi (e.g. Vega & Blackwell 2005, Okabe 2013), it is probable that the scutacarids feed on them. Moreover, dead wood and microorganisms are available as possible food sources (Okabe 2013). The red palm weevil Rhynchophorus ferrugineus is another curculionid beetle which has been reported as host for a not nearer identified species of the genus Scutacarus in Egypt (El-Sharabasy 2010). The beetle is an important pest of palms in Asia and Europe which is known to be a host to several mite species (Al-Deeb et al. 2011), but associated scutacarids had never been reported before.
One species of Cupepidae, the North American Tenomerga cinerea, has been given as host of the scutacarid Imparipes cupes Delfinado & Baker, 1978 (Delfinado and Baker 1978). Larvae of T. cinerea are known to thrive in fungus-infested wood (Hörnschemeyer 2005), which probably is a suitable habitat for the scutacarids as well.
Scarabaeidae, which often are coprophilous and as such visit habitats that are commonly used by scutacarid species, have also been reported as phoresy hosts for several scutacarids: Archidispus sacculiger (Mahunka, 1968), Heterodispus near elongatus (Trägardh, 1904), two Pygmodispus species and three Scutacarus species have been found phoretic on Scarabaeidae from Brazil, Canada, Chile, Iran, Java and the USA (Paoli 1911, Mahunka 1968a, Norton 1973, Ebermann and Rodrigues 2001, Rodrigues et al. 2001, Ebermann et al. 2003, Loghmani et al. 2014; Annex III). Not all of the scarabaeid species used as hosts are dung beetles: imagines of Osmoderma eremicola feed on sap and damaged fruits, but the juvenile stages thrive in decaying wood (Norton 1973), and the juveniles of Bothynus striatellus are crop pests in South America (SiNAViMo 2015). As attachment sites on the beetles, the underside of the elytra, hairs on the ventral side and the suture between forelegs and head have been reported (Mahunka 1968a, Ebermann and Rodrigues 2001, Ebermann et al. 2003).
Another family of beetles occurring in different types of decaying material is the family Ptiliidae. These mycophagous beetles are characterized by very small body sizes (Zahradník 1985), still three different Scutacarus species have been reported from one single specimen of Ptiliidae which has not been identified to genus- or species level (Mahunka and Zaki 1992, Truck 2012; Annex III). The mites attach to thick hairs on the abdomen or legs of the beetles (Truck 2012).
Members of the family Tenebrionidae also are used as hosts by scutacarids (Annex III). Beetles of this family are generalists (Zahradník 1985), and thus it is very likely that they often visit habitats that are suitable for scutacarids, too. Two Heterodispus species from Ukraine and Africa have been reported phoretic on tenebrionid beetles, whereas the concrete attachment sites are unknown (Khaustov 2008, Jagersbacher-Baumann and Ebermann 2012a).
The last family of Coleoptera which has been reported as host for Scutacaridae is the Pselaphidae. Only one scutacarid species phoretic on Pselaphidae is known to date, namely Imparipes pselaphidorum Ebermann, 1988 from individuals of the genus Centrophthalmus in Tanzania (Ebermann 1988; Annex III). Many Pselaphidae are known to be myrmecophilous (Zahradník 1985), which could be an explanation for why the scutacarids can be found on with them: the mites could be primarily commensals of ants which also can move to the syntopically occurring beetles. However, the pselaphids found to bear I. pselaphidorum are not myrmecophilous and thus the reasons for the associations between mites and beetles remain unclear (Ebermann 1988).
Finally, there are also reports of scutacarid mites phoretic on not nearer identified “coleoptera” from Australia and Ghana (Annex III). They comprise Archidispus cornutus (Mahunka, 1973), Diversipes horridolatus Mahunka, 1975, Lamnacarus expansus Mahunka, 1973, 2 Scutacarus species, 3 Symbolacrasis species and two species of Thaumatopelvis (Mahunka 1973a, 1973b, 1975c, Ebermann 1980b). The three species Symbolacrasis acutimera Mahunka, 1973, S. hypostigma Mahunka, 1973 and S. synmixta Mahunka, 1973 are the only known members of Symbolacrasis, which means that the entire genus is known exclusively phoretic on coleopterans.
In addition to beetles, different Hymenoptera belonging to the suborder Apocrita are important hosts for Scutacaridae. Apocrita divide into the monophyletic subclade Aculeata (containing ants, bees and wasps) and the artificial, paraphyletic group Terebrantia/ “Parasitica”. Members of Terebrantia have only been reported as hosts of Scutacaridae in one case: Imparipes dispar has been found phoretic on specimens of Eucoilidae, Proctotrupoidea and Pteromalidae (Ebermann et al. 2016; Annex I). On the other hand, a larger number of scutacarids (35 spp.) is associated with different social, but also with solitary Aculeata, as will be described below.
In the superfamily Apoidea, scutacarid mites occur together with Apidae, Andrenidae, Colletidae, Crabronidae, Halictidae and Sphecidae (Annex IV). In fact, Scutacaridae are amongst the most frequent and diverse associates of bees (Eickwort 1994). Most of the associated scutacarids belong to the genera Imparipes, followed by Scutacarus (Figure 10).
In Apidae, scutacarid mites play no large role as inquilines of the economically important honey bee Apis mellifera: only two species, Imparipes apicola and Scutacarus acarorum, have been reported from nests of honey bees, and neither of them is a frequent associate (Banks 1914, Schousboe 1986). More scutacarids can be found as common inquilines and phoronts of bumble bees (genus Bombus). The scutacarid acarofauna of bumble bees consists of 6 Scutacarus species, Parascutacarus indicus Baker & Delfinado, 1975 and Imparipes degenerans Berlese, 1904 (e.g. Karafiat 1959, Cross and Bohart 1969, Larsson 2007, Jagersbacher-Baumann 2015; for other references see Annex IV). Imparipes degenerans, however, has only been reported once from Bombus and can normally be found on ants and in their nests and also in rodents’ nests. Among the Scutacarus species associated with bumble bees, S. acarorum is particularly worth mentioning as it is one of the most common “bumble bee mites” (Chmielewski 1971), as it performs hyperphoresy on phoretic mesostigmatid deutonymphs (e.g. Schousboe 1986) and as it was one of the first mite species to be described in history by Goeze in 1780 (Goeze 1780). On bumble bees, scutacarid mites can be found on the base of the forewings, on the thorax or between thorax and abdomen, and they are most frequent on hibernated queens (e.g. Chmielewski 1971, Schousboe 1986). In the nests, the scutacarids can mostly be found in the outer portions, and they are much less frequent in artificial hives than in natural nests (Chmielewski 1971). As workers emerge constantly in bumble bee nests, the life cycles of most associated mites are expected not to be well synchronized with that of their hosts (Okabe 2013). Indeed, the developmental cycle of S. acarorum takes around 9 days (Jagersbacher-Baumann and Ebermann 2013), being much shorter than that of its host.
In nests of Apidae, flower nectar, pollen, microorganisms and nest debris are present, although microorganisms and nest debris lack in honey bees (Okabe 2013). Dead brood, provisions and parts of the debris can become moldy and then offer suitable food for saprophagous mites like Scutacaridae (Eickwort 1994). As there is not much substrate which can become moldy in the nests of honey bees (Okabe 2013), this may explain the scarceness of associated scutacarids.
Solitary bees belonging to the families Andrenidae and Colletidae generally have few associated mite genera (Eickwort 1994). However, they serve as hosts for four scutacarid species of the genus Imparipes (e.g. Eickwort 1979, Ebermann and Hall 2003, 2004, Ebermann et al. 2013; for other references see Annex IV). The two bee families offer different habitat conditions: members of Andrenidae are ground-nesting, while Colletidae nest in dead wood. Imparipes breganti and I. burgeri, which use bees of both families as hosts, are thus considered to be pronounced generalists (Ebermann and Hall 2004, Ebermann et al. 2013).
Imparipes apicola is associated with different species of Andrenidae, but also with Halictidae, which are another family of soil-nesting and rather primitive bees that exhibit a great diversity of mite associates (Eickwort 1979, 1994). Eickwort (1979) gives a thorough description of the life history of I. apicola inside the cells of a laboratory reared Lasioglossum host, showing that, in contrast to species associated with bumble bees (see above), the mite’s life cycle follows that of its host. Halictid bees are also hosts for 15 other Imparipes species, moreover for two Nasutiscutacarus and two Scutacarus species (e.g. Beer and Cross 1960, Delfinado and Baker 1976, Ebermann and Hall 2005; for other references see Annex IV). Most of these scutacarids have been found phoretic on the bees, but some have also been reported from their nests. Like in Apidae, nests of Halictidae provide flower nectar, pollen, microorganisms and nest debris as potential food sources for different mites (Okabe 2013). Accordingly, Ordway (1964) reported mites of the genus Imparipes from halictid nest cells containing pupae, near or on the fecal deposit, and Eickwort and Eickwort (1971) also found larvae of I. eickworti Mahunka in cell contents of its halictid host.
Sphecoid wasps of the families Crabronidae and Sphecidae have been identified as phoresy hosts for five scutacarids, one Archidispus (A. sphecis Mahunka, 1977) and four Imparipes species (e.g. Kuhlmann 1998, Ebermann and Hall 2003, 2004, Ebermann et al. 2013; for other references see Annex IV). Among these mites, only A. sphecis (Lang and Mahunka 1977) is exclusively known from sphecids. The reported host species build nests in different substrates like sand, mud, dead wood or stalks. After deposition of the eggs, the nests are supplied with different paralyzed insects or spiders which serve as food for the offspring (Witt 1998). The nests probably are suitable habitats for mites as they contain enough material which could become moldy. However, no scutacarids have been reported from nests so far, all of the reported species were phoretic on the sphecoid wasps. Lang and Mahunka (1977) described the coxal region as favored attachment site on the hosts.
Associations between Vespoidea (exclusive Formicidae!) and Scutacaridae are extremely rare. Species of Mutilidae and Pompilidae have been found to carry phoretic specimens of Imparipes burgeri Ebermann & Jagersbacher-Baumann, 2013 (Ebermann et al. 2013) and Scutacarus subquadratus Khaustov & Chydyrov, 2004 (Loghmani et al. 2014). Wasps of these families build their nests in sand, are kleptoparasites of other wasps or parasitoids on halictid bees (Witt 1998). Another scutacarid, I. haeseleri, has been reported from a member of Vespidae (Ebermann and Hall 2003, Hall and Ebermann 2005). The respective host species, Symmorphus bifasciatus, builds its nests in stalks, reed or dead wood (Witt 1998) and animal meat, microorganisms, nest debris and dead larvae are available as food in its nests (Okabe 2013). Finally, Vitzthum (1927) reported random findings of S. acarorum on wasps, not without hypothesizing that these associations may be the rare results of occasional contact between wasps and bumble bees on flowers.
The majority of scutacarid mites living in associations with other animals can be found with another family of Vespoidea, the Formicidae, or ants (214 spp.). Most associated scutacarids belong to the genera Scutacarus and Imparipes (Figures 11, 12; Annex V-VII). A great variety of different mite taxa can be found as guests of ants (e.g. Vitzthum 1919, Campbell et al. 2013), and it is also common to encounter more than one scutacarid species within one single ant nest (e.g. Friedl 2000). Scutacarids are either phoretic on the ants or they live in ant nests, or both. Inside the ants’ nests, nest debris and (in army ants) temporary provisions are available (Okabe 2013), both of which can become moldy and then serve as food sources for scutacarids. On the ants, scutacarids can be found on the thorax, between the coxae, but also on rather exposed parts like the ant’s legs or on the head (e.g. Paoli 1911, Rettenmeyer 1961a, Ebermann 1982, Elbadry et al. 1976).



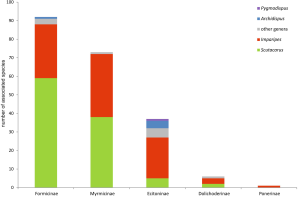


From not further determined “ants”, 51 scutacarid species have been reported: Archidispus haarloevi (Karafiat, 1959), 13 Imparipes species, Pygmodispus calcaratus Paoli, 1911 and 36 Scutacarus species (e.g. Karafiat 1959, Mahunka 1981, 1986, Dobrev 1992; for other references see Annex V). For all other reports of associations between ants and Scutacaridae, at the least the subfamily of the host was given. Most scutacarid species occur together with Formicinae and Myrmicinae (Figure 12; Annex VI, VII).
In the subfamily Formicinae, Archidispus intermissus (Karafiat, 1959), 28 Imparipes species, 2 Lophodispus species, 58 Scutacarus species and Thaumatopelvis reticulatus are present as associates (e.g. Karafiat 1959, Mahunka 1972b, 1977c, Khaustov 2008, Ebermann and Krisper 2014; for other references see Annex VI). The scutacarids could be detected in 5 ant genera, and the majority of all mite species (52 species) was present in the genus Lasius. Ants of this genus can occur in high population densities (e.g. L. alienus or L. niger), they frequently are social parasites of other ants and often feed through trophobiosis in symbiosis with aphids (Seifert 1996). Scutacarids can also often be found in associations with zoophagous Formica ants, further with the genera Camponotus and Paratrechina. There are even species associated with desert ants of the genus Cataglyphus, which are highly thermophilic scavengers feeding mostly on dead arthropods (Lenoir et al. 2009).
Thirty-four Imparipes species, Lophodispus irregularis and 38 Scutacarus species have been reported from the subfamily Myrmicinae (e.g. Ebermann 1979, 1980a, b, Khaustov 2008, Ebermann and Krisper 2014; for other references see Annex VII). The mites have been found in association with ants belonging to 10 genera, and the largest number of mites (25 species) was present in the genus Myrmica. The genera accepted as hosts are characterized by different life styles, ranging from harvester ants feeding on seeds (e.g. Messor) to zoophagous (e.g. Myrmica) and omnivorous ants (e.g. Solenopsis). The colony sizes of the hosts also vary, ranging from 40-120 workers in Stenamma to some thousand workers in Aphenogaster (Seifert 1996).
Scutacarids can also be associated with army ants of the subfamily Dorylinae (following the taxonomic classifaction by Brady et al. 2014). These ants are carnivorous, they raid for food in large groups, build extremely large colonies and the whole colonies emigrate periodically (Rettenmeyer et al. 2011, Okabe 2013). Colonies of Dorylinae are home to several animals belonging to a variety of taxa, but their most abundant guests are mites (Gotwald 1996, Rettenmeyer et al. 2011). Accordingly, Dorylinae also serve as hosts for scutacarids: 39 scutacarid species belonging to seven genera have been reported from five army ant genera (Annex V). The respective mites are 4 Archidispus species, 24 Imparipes species, Pygmodispus dorylini Mahunka, 1977, Rettenmeyerella petropolitana solenifera Mahunka, 1977, 2 Scutacaropsis species, 5 Scutacarus species and 2 Thaumatopelvis species (Rettenmeyer 1961a, Mahunka 1977a,b, Ebermann 1980b, Berghoff and Franks 2007, Berghoff et al. 2009, Rettenmeyer et al. 2011). Within ants, Dorylinae display the highest diversity of scutacarids on genus level.
Six scutacarid species (3 Imparipes, Lophodispus tapinoma Sobhi & Hajiqanbar, 2017 and 2 Scutacarus) were found in association with ants of the subfamily Dolichoderinae, either with members of the genus Tapinoma or with members of Liometopum (Mahunka 1977c, 1982, Khaustov 2008; Annex V). Tapinoma ants are omnivorous and not sedentary, instead they often change the location of their nests, while Liometopum ants are sedentary, associated with trees and shrubs and they can also be minor pests in housing areas (Hoey-Chamberlain et al. 2013).
Only one scutacarid, Imparipes malus Khaustov, 2008, has been reported from Ponera coarctata of the subfamily Ponerinae (Khaustov 2008; Annex V). The concealed life style of this thermophile ant species makes it difficult to sample (Seifert 1996, Wagner 2014), so intensified collections may reveal a higher number of associated scutacarids.
Different other insect taxa beside Coleoptera and Aculeata also serve as hosts for a variety of Scutacaridae (Annex I, Figures 6, 7, 13). There are unique reports of scutacarid species found on earworms (Dermaptera) and on bugs (Heteroptera), respectively (Trägårdh 1905, Paoli 1911, Khaustov 2008). The mite species reported from earworms is Imparipes histricinus, which is a pronounced generalist and which has been reported from ants and beetles, from mammals’ nests and from soil (Table 2). As will be discussed below, I. histricinus might in fact be a cryptic species complex. On the other hand, the scutacarid associated with the heteropteran family Reduviidae, Imparipes nikitensis Khaustov, 2005, has not been reported from any other host (Khaustov 2008).
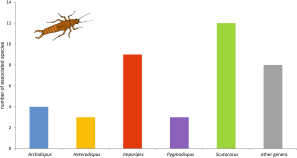





Reports of Scutacaridae associated with Orthoptera are quite rare (Annex I). Two Heterodispus species have been found on crickets (Mahunka 1964a, Khaustov 2008), and Imparipes rectangulatus Mahunka, 1977 has been reported from grasshoppers (Mahunka 1977a). The latter species is another generalist, occurring also together with South American army ants (e.g. Berghoff et al. 2009).
Nine scutacarid species and subspecies can be found phoretic on 12 families of flies (Diptera) from the Holarctic and South America (Annex I): 2 Imparipes species, Scutacaropsis baculitarsus, Scutacaropsis baculitarsus agaricus, 3 Scutacarus species, Rettenmeyerella petropolitana Vitzthum, 1928 and Rettenmeyerella petropolitana solenifera. Most of these scutacarids have also been reported from other environments like soil or from other hosts (e.g. Norton and Ide 1974, Binns 1979). On their host, the scutacarids can be found on caput, thorax, abdomen, wings or on the metacoxae (Norton and Ide 1974, Binns 1979, Zaki et al. 1987, Ebermann et al. 2016).
As has been shown above, a vast number of scutacarids can be found in the nests of social hymenopterans. One could assume that the abundance of scutacarid species in another taxon of social insects, namely termites (Isoptera), should be high as well. After all, fungal cultures and microorganisms are available as food sources in their nests (Okabe 2013). However, a surprisingly low number of scutacarids has been reported in association with termites so far (Annex I). The respective species are Archidispus brevisetus (Mahunka, 1964) from Africa, Coronipes samsinaki (Mahunka, 1966) from China (Mahunka 1966) and C. sperati Khaustov et al., 2016 from South Korea, Gerdalbertia elongata Khaustov, Hugo-Coetzee & Ermilov, 2017 from South Africa, 4 Imparipes species from Africa, Europe and the USA, and Scutacarus mirabilis Mahunka, 1964 also from Africa (Mahunka 1964a, Delfinado and Baker 1976, Khaustov et al. 2016, 2017, Baumann and Ferragut unpubl.). Moreover, not nearer identified Scutacaridae have been reported by Wang et al. (2002) from China.
Some Scutacaridae have been found in fecal pellets of eagles (Accipitridae) and owls (Strigiformes), in the debris of mutton bird (Procellariidae) nests, on the feathers of “Antarctic birds” and in the nests of other not identified birds (Annex VIII). Strictly speaking, the occurrence of scutacarid mites in birds’ fecal pellets does not indicate any further association between the two animals; the respective mites have still been included in the present review because reports of scutacarids in connection with birds are scarce. The mites reported in association with birds (Figure 14) comprise 7 Scutacarus species, Heterodispus longisetosus (Womersley, 1955), Imparipes obsoletus Rack, 1966 and Pygmodispus abestus Mahunka & Philips, 1978 and unidentified Scutacaridae (Womersley 1955, Delfinado et al. 1976, Delfinado and Baker 1976, 1978, Mahunka and Philips 1978, Philips et al. 1988, Krivolutsky et al. 2004). Curiously, one of the Scutacarus species, S. meansi Delfinado & Baker, 1978, has not only been found in bird’s nests’ debris, but also in dog food (Delfinado and Baker 1978). Another species, S. imitans Delfinado & Baker, 1976, has been reported from bird’s nests, but also from nests of shrew and from the coat of grey squirrels (Delfinado and Baker 1976), suggesting that this mite is a pronounced generalist. Apart from the mites from Antarctic birds, all scutacarids associated with birds have been reported from North America.



Scutacarids have been reported from several “small mammals” (52 spp.), and in all cases where host species have been nearer identified in literature, they turned out to be soil dwelling and to belong either to the group of Eulipotyphla or Rodentia. Apparently, the nests of these mammals offer a favourable environment for mites (Ebermann 2004). Like in birds, most associated scutacarids belong to the genus Scutacarus (Figure 15). All in all, the following scutacarids have been reported: Diversipes exhamulatus (Michael, 1886), 5 Heterodispus species, 15 Imparipes species, Lophodispus irregularis, Pygmodispus zicsii Mahunka, 1964, 28 Scutacarus species and Reductacarus singularis Mahunka, 1963 (e.g. Mahunka 1967a, 1972b, 1975b, 1977d, Sosnina and Sevastianov 1975, Sevastyanov and Uzhevskaya 2003; for more references see Annex VIII). Most species occur in the nests of rodents, only 12 Scutacarus species and Reductacarus singularis were found in the nests of Eulipotyphla. While most scutacarids live inside their mammal hosts’ nests, five species, namely H. foveatus, I. cupes, I. spickai Mahunka, 1977, S. geomyi Mahunka, 1977, S. imitans and S. missourensis Mahunka, 1977, were found phoretic on their hosts (Drummond 1957, Basolo and Funk 1974, Delfinado and Baker 1976, Mahunka 1977d, Spicka 1981, Estébanes-González and Cervantes 2005, Jagersbacher-Baumann and Ebermann 2012a). Amongst these, only I. cupes has not been reported from the coat of its host, the pocket mouse Chaetodipus spinatus lambi, but from its dorsal skin, where the mite apparently lives in small cavities that appear as scabs (Estébanes-González and Cervantes 2005). This report is extraordinary as it is the only one describing the occurrence of scutacarid mites “inside” its host and not attached to external structures like setae, hairs or feathers.
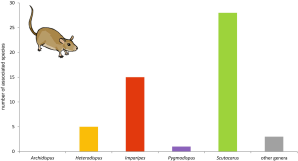


All of the scutacarids associated with mammals except the African H. foveatus and the Mexican I. cupes have been reported from the Holarctic. Unidentified scutacarids have also been reported from mammal carrion (Early and Goff 1986) and scutacarid mites can frequently be found in mammal dung (e.g. Mahunka 1964b, Truck 2012). It would have exceeded the scope of the present review to include all of these dung inhabiting species as, in contrast to scutacarids in bird’s faeces, there is a high number of them and as they cannot be regarded as real associates of mammals.
Last but not least, there are even studies in which Scutacaridae have been found in connection with humans. Of course these encounters are no examples of associations and should only be considered as random or accidental, but they still are worth mentioning in this paper: Paik et al. (1992) found not identified Scutacaridae in low numbers in house dust in Seoul, Korea, and Modak (1991) found members of the genera Imparipes and Scutacarus in house dust in West Bengal, India. Modak even described a new species “Imparipes tropicus” from house dust, but as the description is only available in a PhD thesis and has not been published properly, it has to be considered a nomen nudum.
Some scutacarid genera show clear preferences towards special host taxa, while other genera are evenly distributed amongst a variety of hosts (Figure 16). Strong preferences can be detected in the genera Archidispus, Imparipes and Scutacarus.



The vast majority of all known Archidispus species (85%) occurs phoretic on beetles, and within this taxon predominately on ground beetles (Carabidae). Less than 10% of the known Archidispus species have been reported in association with other hosts- ants, “insects”, bees and termites. The term “insects” has been adopted from the original literature and indicates that the hosts had not been identified in detail. “Insects” might therefore also include beetles, meanining that the number of Archidispus associated with Coleoptera may even be higher than given here. There are no reports of Archidispus in association with birds or mammals.
Both Imparipes and Scutacarus clearly prefer ants as hosts: 63% of all known Imparipes and 58% of all known Scutacarus species have been found on ants or in ant nests. Both genera have also been reported from associations with beetles, bees, termites, other insects, small mammals and birds. Within these other host taxa, most species of both genera can be found on mammals and bees (or in their nests), with Imparipes showing a trend towards preferring bees and Scutacarus preferring mammals as host. Scutacarus is, moreover, the only scutacarid genus occurring in associations with other acari.
In the genus Heterodispus, no comparable clear trend towards host preference is recognizable. However, most species of this genus have been reported as inquilines of small mammals (42%), 25% were found on beetles and “insects”, respectively, and less than 10% as inquilines of birds.
Pygmodispus can be found on ants, beetles, other insects, small mammals and birds, and there is a tendency towards “insects” as hosts since most species (63%) were found phoretic on this taxon.
The genera Diversipes, Lamnacarus, Lophodispus, Nasutiscutacarus, Parascutacarus, Reductacarus, Rettenmeyerella, Scutacaropsis, Symbolacrasis and Thaumatopelvis are poor in species, so it does not make much sense to talk about “trends” regarding their host taxa. However, some of these genera have been reported from diverse hosts: Diversipes (3 species) can be found together with beetles, bees and small mammals; Reductacarus (2 species) with “insects” and mammals; Rettenmeyerella (2 subspecies) with ants and diptera; and finally Thaumatopelvis (6 species) with beetles, ants and other “insects”. The other genera apparently are restricted to one host taxon: Lamnacarus (2 species) and Symbolacrasis (3 species) have been reported from beetles, Nasutiscutacarus (2 species) and Parascutacarus (1 species) from bees, and Lophodispus (2 species) and Scutacaropsis (2 species) from ants.
In most of cases, the host range of single scutacarid species apparently is rather narrow. One species usually uses hosts belonging to one genus or family, and several species have been reported from only one host species (Annex). A similar trend can also be recognized in other phoretic mite taxa. For example, when analyzing mites associated with bees, Eickwort (1994) reported that few mite species can be found on only one host species, but that there also few occurring on hosts belonging to more than one subfamily; the majority of mite species are in association with many congeneric host species which are available in the range of the mite.
Examples for scutacarids that have been reported only from one host species are Imparipes adleri Delfinado & Baker, 1976 found on the termite Reticulitermes virginicus (Delfinado and Baker 1976), Pygmodispus zicsii from the rodent Cricetulus migratorius (Sevastyanov and Uzhevskaya 2003) or Scutacarus sabinaesimilis Khaustov & Chydyrov, 2004 from the ant Tapinoma simrothy (Khaustov 2008). However, Scutacaridae in general are weakly studied and several reports of phoretic scutacarids are random findings. Comprehensive or more focused studies on the mites’ hosts like the one by Ebermann et al. (2016) on I. dispar might thus reveal more host species.
Some scutacarid species, however, already seem to be pronounced generalists and can be found on a variety of hosts belonging to very different taxa (Table 2). For most of these host taxa, one important ecological similarity can be encountered: a lifestyle strongly connected to the substrate “soil”. Because of this shared habitat, it probably is rather easy for mites with a generalist life style to switch between different hosts. For other host taxa, like flies and ants, it is more difficult to find clear points of contact between them and thus to explain the occurrence of a mite species on both groups.
While some of the presumed generalists have been investigated thoroughly and thus their species status is quite certain (e.g. Jagersbacher-Baumann and Ebermann 2012a, Ebermann et al. 2016), the status of other generalists is questionable. As will be discussed below, the respective species could have been misidentified or might be complexes of cryptic species.
The present work shows that most animal taxa that are accepted as host by scutacarid mites have a strong connection to the habitat “soil”: either they are highly mobile, epigaeic species, or they are endogaeic species building nests in the ground. As Scutacaridae are also soil inhabiting mites, it seems probable that associations between them and their host evolved through the shared habitat. The genus specific trends towards different taxa also indicate that the associations between scutacarids and their hosts evolved independently several times within the family, and most probably also within the genera.
Exceptions from typical soil living hosts are bark beetles and some Apoidea and Vespoidea that thrive in dead wood, reeds and stalk; moreover not ground dwelling birds which can harbour scutacarids in their nests. These hosts, however, still offer favourable conditions in form of mouldy debris in their dwellings which contains the mites’ nutritional fungi.
Based on the compiled information on scutacarids and their hosts given in the present review, their associations can be grouped into three categories with typical ecological characteristics:
1. Phoresy only
Host within this group have high dispersal abilities and are only used for phoresy. They can transport the scutacarids to new, suitable habitats, but they themselves do not provide theses respective habitats (that is, they do not build nests that offer favourable conditions for the mites). Typical for this group are epigaeic, fast running Carabidae and several other beetles like Staphylinidae or Hydrophilidae, moreover flying insects like Orthoptera and Diptera.
2. Shared habitat only
This group includes soil-dwelling hosts whose nests have been invaded randomly by the scutacarid mites through the surrounding soil (as described by Okabe 2013). The mites live in the dwellings of their hosts as facultative commensals, or, if the association is already more evolved, as obligate inquilines. In this category, hosts are not used for dispersal. This type of association is not very common; typical hosts are mammals and ant species with large and temporarily stable nests, like some members of the genus Crematogaster.
3. Shared habitat and phoresy
In this category, the hosts build nests which are more or less temporary. The scutacarids live in the hosts’ nest as obligatory commensals and when the time is right they use their hosts for phoresy in order to reach new habitats, that is, new nests. Examples for hosts in this group are ant species with fissioning colonies, bumble bees with annual colonies or wild bees which build single brood cells. In fact, the majority of associations between scutacarid mites and their hosts belong to this category.
Although many associations between various species of Scutacaridae and other animals have been reported, most times also including information about the type of association (inquilinism, phoresy, or both), few details are known about the behaviour of the mites and the interactions with their hosts. At has been mentioned in the introduction, the mechanisms of host finding, host identification, host switch and identification of suitable habitats are only speculative today and should be subject of future studies. Behavioural experiments which could shed a light on this issue can, however, be rather complicated because of the mites’ extremely small size which makes them more difficult to handle.
The cohabitation of mites and their hosts also raises questions: What fungi exactly do the mites feed on in the shared habitat, are they food specialists or generalists? The nutritional fungal species have only been identified for very few scutacarid species (Jagersbacher-Baumann and Ebermann 2013). Have the scutacarids any influence, positive or negative, on their hosts? Most likely, the tiny mites do not bother or harm their hosts in any way. A strong support for this assumption is the fact that hosts usually do not remove their mites (Eickwort 1994). It is possible that Scutacaridae even have a positive effect on their hosts as they could play a sanitary role by feeding on potentially harmful fungi. Such sanitary roles have been hypothesized for other mite taxa before (Okabe 2013), but so far only one study provides statistically supported evidence for it: Biani et al. (2009) observed significant correlations between the presence of Laelaspoides mites (Mesostigmata) in a bee nest, the absence of fungi in brood cells and a decrease in bee mortality. Studies on scutacarids about this topic are still lacking to date. Preliminary observations revealed that scutacarid mites can consume considerable amounts of fungal hyphe in laboratory cultures (Baumann unpublished), so a sanitary effect indeed seems probable.
In scutacarid species with a broad host spectrum and/or a wide geographical distribution, doubts about the status of the respective species should arise for two reasons, as has already been discussed in Ebermann et al. (2016).
(1) The frequent occurrence of a species on certain hosts tempts to neglect a proper identification. For example, S. acarorum is widely distributed and is also the most common scutacarid associated with bumble bees. Comparisons between presumed S. acarorum individuals from spatially distinct populations (European localities and New York) confirmed their conspecificity even after thorough morphometric analyses (Jagersbacher-Baumann 2015). Because of its abundance, apparently all scutacarids encountered on bumble bees often are being identified as S. acarorum without any closer inspection. However, there are at least three other morphologically similar scutacarid species associated with bumble bees: S. deserticolus, S. mendax and S. occultatus Sevastianov, 1975. They all belong to the acarorum species-complex as they share a very similar phenotype and can also be encountered syntopically in the nests of large bumble bee species (Jagersbacher-Baumann 2014). Re-inspections of presumed S. acarorum individuals thus already revealed incorrect classifications: for example, specimens of “S. acarorum” from Hamburg, Germany, were identified as S. deserticolus (Ebermann 1991a), and “S. acarorum” reported from bumble bees in Argentina turned out to be S. mendax or a new species close to S. mendax (Revainera et al. 2014 and pers. comm.). A not nearer identified scutacarid from Brazilian bumble bees (Guerra et al. 2012) also turned out to be a variation of S. mendax (Baumann unpublished).
(2) Thorough morphological and molecular genetic analyses of scutacarids will most likely reveal the existence of several “cryptic” species. The term cryptic species describes species which are impossible or extremely difficult to distinguish by traditional (morphological) means, and due to advanced techniques, many of them have been described in the recent years in mites (Knee et al. 2012, Skoracka et al. 2015). Although it had been stated before that host-race formation of mites might be a main driving force for cryptic speciation, this hypothesis could not be supported by Skoracka et al. (2015). They showed that not only strong host relationships can induce speciation, but also abiotic or other host-independent environmental factors can do so. An example for a cryptic species complex could be present in the scutacarid I. histricinus, which apparently is one of the most frequent guests of ants (Annex V-VII; Karafiat 1959, Okabe 2013). Closer inspections of the mites identified as I. histricinus might reveal the existence of several new species with a similar phenotype. In fact, there are already several reports of scutacarids only being similar to I. histricinus: for example, Mahunka (1967b, 1970b) reported I. cf. histricinus from ants of the genus Formica. The respective specimens have not yet been described as separate species.
Additional, all other scutacarid species with a broad host spectrum (Table 2) or species with a wide geographic distribution also could in reality be complexes of cryptic species.
The factors determining the morphs in dimorph/phoretomorph scutacarid genera like Archidispus or Scutacarus are another point which needs further clarification. Ebermann (1991a) already showed that morph determination might happen in the larval stage and might be induced by the quality and/or quantity of available food, but detailed and statistically supported studies are still pending.
Last but not least, the knowledge of the scutacarid fauna in different countries is very fragmentary, depending on the individual interests of different researchers and made difficult by the generally low abundance and small size of the scutacarids. While some countries like Austria, Hungary, Iran or Russia have been and/or are still investigated intensely by researchers like Ebermann, Hajiqanbar, Khaustov, Loghmani and Mahunka, others, like Spain or Portugal (Ferragut 2015) are practically ``terra incognita" when it comes to Scutacaridae. From other countries, soil living scutacarids have been reported, but few or no associations with other animals are known yet. For example, although several scutacarid mites are known from Australia (e.g. Mahunka 1967c, 1974d), only three species have been reported from associations with other animals (Womersley 1955, Mahunka 1975c, Seastedt et al. 1986). Investigating Scutacaridae from new geographical areas as well as closer looks on other possible host taxa will most likely bring more associated/phoretic scutacarid species to light, several of which might be new to science.
Because of their size, scutacarids may often simply be overlooked, and moreover, they can easily be mistaken for astigmatid deutonymphs (hypopi) by non-specialists when they are attached to their hosts. With the present review, the awareness of Scutacaridae and their manifold associations with other animals will hopefully rise.
I am forever indebted to my ``Doktorvater" (doctoral supervisor) Ernst Ebermann for introducing me to the fascinating family Scutacaridae, for passing on his invaluable collection of mites and literature to me and for still supporting me even after his retirement. ``Muito obrigada" to Carlos Flechtmann for sharing his information of associations between mites and other arthropods with me.
Al-Deeb M.A., Muzaffar S.B., Abuagla A.M., Sharif E.M. 2011 — Distribution and abundance of phoretic mites (Astigmata, Mesostigmata) on Rhynchophorus ferrugineus (Coleoptera: Curculionidae) — Fla. Entomol., 94(4): 748-755. doi:10.1653/024.094.0403 ![]()
Athias-Binche F. 1994 — La phoresie chez les acariens. Aspects adaptatifs et evolutifs — Perpignan: Editions du Castillet. pp. 178.
Bajerlein D., Błoszyk J. 2003 — Two cases of hyperphoresy in mesostigmatic mites (Acari: Gamasida: Uropodidae, Macrochelidae) — Biol. Lett., 40(2): 135-136.
Baker E.W., Delfinado M.D. 1975 — A new genus of Scutacaridae (Acarina) on a bumble bee from India — Coop. Econ. Inst. Rpt., 25(19): 379-381.
Bakiani S.K., Monfared A., Hajiqanbar H., Azhari S. 2016 — A survey on Apoidea bees (Insecta: Hymenoptera) and their associated mites in Fars Province, Iran — J. Ins. Biodivers. Syst., 02(2): 285-299.
Balogh J., Mahunka S. 1962 — Beiträge zur Tarsonemini-Fauna Ungarns III. (Acari, Trombidiformes) — Fol. Entomol. Hung., 15(29): 509-514.
Banks N. 1904 — A treatise on the Acarina, or mites — Proc. U. S. Nat. Mus., 28(1382): 1-114.
Banks N. 1906 — Descriptions of some new mites — Proc. Entomol. Soc. Wash., 7(1905): 133-142.
Banks N. 1914 — New Acarina — J. Entomol. Zool. (Pomona), 6:44-66.
Basolo F., Funk R.C. 1974 — Ectoparasites from Microtus ochrogaster, Peromyscus leucopus and Cryptotis parva in Coles County, Illinois — Trans. Ill. Acad. Sci., 67(2): 211-221.
Beer R.E., Cross E.A. 1960 — A New Genus and Two New Species of Scutacarid Mites (Acarina) Phoretic upon Bees — J. Kans. Entomol. Soc., 33(2): 49-57.
Berghoff S.M., Franks N.R. 2007 — First Record of the Army Ant Cheliomyrmex morosus in Panama and its High Associate Diversity — Biotropica, 39(6): 771-773.
Berghoff S.M., Wurst E., Ebermann E., Sendova-Franks A.B., Rettenmeyer C.W., Franks N. 2009 — Symbionts of societies that fission: mites as guests or parasites of army ants — Ecol. Entomol., 34(6): 684-695. doi:10.1111/j.1365-2311.2009.01125.x ![]()
Berlese A. 1903 — Diagnosi di alcuni nuove specie di Acari italiani, mirmecofili e liberi — Zool. Anz., 27: 12-28.
Biani N.B., Mueller U.G., Wcislo W.T. 2009 — Cleaner mites: sanitary mutualism in the miniature ecosystem of neotropical bee nests — Am. Nat., 173: 841-47. doi:10.1086/598497 ![]()
Binns E.S. 1979 — Scutacarus baculitarsus Mahunka (Acarina: Scutacaridae) phoretic on the mushroom fly Megaselia halterata (Wood) — Acarologia, 21: 91-107.
Brady S.G., Fisher B.L., Schulltz T.R., Ward P.S. 2014 — The rise of army ants and their relatives: diversification of specialized predatory doryline ants — BMC Evol. Biol. 14: 93. doi:10.1186/1471-2148-14-93 ![]()
Campbell K.U., Klompen H., Crist T.O. 2013 — The diversity and host specificity of mites associated with ants: the roles of ecological and life history traits of ant hosts — Insect. Soc., 60: 31-41. doi:10.1007/s00040-012-0262-6 ![]()
Çankaya N.E., Kaftanoglu O. 2006 — An Investigation on Some Diseases and Parasites of Bumblebee Queens (Bombus terrestris L.) in Turkey — Pak. J. Biol. Sci., 9(7): 1281-1286.
Chmielewski W. 1971 — The mites (Acarina) found on bumblebees (Bombus Latr.) and in their nests — Ekol. Pol., 19: 57-71.
Chmielewski W., Baker R.A. 2008 — Mites (Acarina) phoretic on some common bumblebee species (Bombus spp.) from the Puławy area (South-eastern Poland) — J. Apic. Sci., 52(1): 37-47.
Chydyrov P.R. 2007 — New Mites Species of the Cohort Tarsonemina (Tromidiformes, Acarina) of the Fauna of Turkmenistan — Entomol. Rev., 87(6): 767-775. doi:10.1134/S0013873807060127 ![]()
Cockerell T.D.A. 1910 — Descriptions and records of bees — Ann. Mag. Nat. Hist., 8(6): 727-284.
Cross E.A., Bohart G.E. 1969 — Phoretic behavior of four species of Alkali Bee mites as influenced by season and host sex — J. Kans. Entomol. Soc., 42: 195-219.
Cross E.A., Bohart G.E. 1992 — The Biology of Imparipes apicola (Acari: Scutacaridae) and Its Relationships to the Alkali Bee, Nomia melanderi (Hymenoptera: Halictidae), and to Certain Fungi in the Bee Cell Ecosystem — J. Kans. Entomol. Soc., 65(2): 157-173.
Dastych H. 1978 — Imparipes (Imparipes) rafalskii sp. nov. (Acari, Tarsonemina) a New Species of Scutacarid Mite from Poland — Bull. Acad. Pol. Sci., 26(4): 251-255.
Delfinado M.D., Baker E.W. 1976 — New species of Scutacaridae (Acarina) associated with insects — Acarologia, 18(2): 264-301.
Delfinado M.D., Baker E.W. 1978 — Terrestrial Mites of New York-VII. Key to the Species of Scutacaridae and Descriptions of New Species — J. N. Y. Entomol. Soc., 86(2): 87-101.
Delfinado M.D., Baker E.W., Abbatiello M.J. 1976 — Terrestrial Mites of New York-III. The family Scutacaridae — J. N. Y. Entomol. Soc., 84(2): 106-145.
Dobrev D. 1992 — Neue Angaben über die Scutacariden-Fauna Bulgariens (Acari: Heterostigmata) — Fol. Entomol. Hung., 53: 9-15.
Drummond R.O. 1957 — Observations on Fluctuations of Acarine Populations from Nests of Peromyscus leucopus — Ecol. Monogr., 27(2): 137-152. doi:10.2307/1948573 ![]()
Early M., Goff M.L. 1986 — Arthropod succession patterns in exposed carrion on the island of O'Ahu, Hawaiian islands, USA — J. Med. Entomol., 23(5): 520-531. doi:10.1093/jmedent/23.5.520 ![]()
Ebermann E. 1978 — Zoogeographisch bedeutsame Milbenfunde in der Steiermark (Arach., Acari, Scutacaridae) — Mitt. Abt. Zool. Landesmus. Joanneum, 7(3): 185-190.
Ebermann E. 1979 — Ein Beitrag zur Erforschung der Milben-Familie Scutacaridae (Acari-Trombidiformes) in Österreich und angrenzenden Gebieten — Carinthia II, 169: 259-280.
Ebermann E. 1980a — Zur Kenntnis der ostalpinen Milbenfauna (Acari, Fam. Scutacaridae) — Mitt. Naturwiss. Ver. Steiermark, 110: 143-149.
Ebermann E. 1980b — Zur Zoogeographie, Taxonomie und Larvalentwicklung einiger Scutacariden-Arten aus Nordamerika (Acari, Trombidiformes) — Zool. Anz., 205(1/2): 123-135.
Ebermann E. 1981 — Imparipes (Imparipes) brevitarsus n.sp. (Acari, Scutacaridae), eine mit Ameisen vergesellschaftete neue Milbenart aus Österreich — Mitt. Naturwiss. Ver. Steiermark, 111: 183-192.
Ebermann E. 1982 — Zuchtversuche und morphologische Untersuchungen an heimischen Milben (Acari, Scutacaridae) — Mitt. Naturwiss. Ver. Steiermark, 112: 155-165.
Ebermann E. 1988 — Imparipes (Imparipes) pselaphidorum n. sp., a new scutacarid species phoretic upon African beetles (Acari, Scutacaridae; Coleoptera, Pselaphidae) — Acarologia, 29(1): 35-42.
Ebermann E. 1990 — Taxonomic consequences of the polymorphism found in scutacarids (Acari, Fam. Scutacaridae) — Entomol. Mitt. Zool. Mus. Hambg., 10: 29-42.
Ebermann E. 1991a — Das Phänomen Polymorphismus in der Milbenfamilie Scutacaridae (Acari, Heterostigmata, Tarsonemina, Scutacaridae) — Zoologica, 141: 76 pp.
Ebermann 1991b — Records of polymorphism in the mite family Scutacaridae (Acari, Tarsonemina) — Acarologia, 32(2): 119-138.
Ebermann E. 1992 — Scutacarus acarorum (Goeze, 1780); Heterostigmata, Scutacaridae- an example for the interrelationship between phoresy and polymorphism in mites — In: Kropczynska D. et al. (Eds.). The Acari; Proceedings of the 2nd Symposium EURAAC. pp. 193-196.
Ebermann E. 1995 — Indication of jumping ability in the mite family Scutacaridae (Acari, Tarsonemina) — Entomol. Mitt. Zool. Mus. Hambg., 11(152): 205-209.
Ebermann E. 1998 — Imparipes (Sporichneuthes nov. subgen.), a remarkable new taxon in the family Scutacaridae (Acari, Heterostigmata) — In: Ebermann E. (Ed.). Arthropod Biology: Contributions to Morphology, Ecology and Systematics. Biosystematics and Ecology Series, Österreichische Akademie der Wissenschaften, Vienna, 14. pp. 179-214.
Ebermann E. 2004 — Tragewirt-Gemeinschaften (Phoresie) bei Spinnentieren (Arachnida) — Denisia, 12: 93-110.
Ebermann E., Fain A. 2002 — A new subgenus of phoretic mite (Acari: Scutacaridae) Associated with African Halictid Bees (Hymenoptera: Halictidae) — Int. J. Acarol., 28(4): 367-371. doi:10.1080/01647950208684312 ![]()
Ebermann E., Goloboff P.A. 2002 — Association between neotropical burrowing spiders (Araneae: Nemesiidae) and mites (Acari: Heterostigmata, Scutacaridae) — Acarologia, 42(2): 173-184.
Ebermann E., Hall M. 2003 — First record of sporothecae within the Mite Family Scutacaridae (Acari, Tarsonemina) — Zool. Anz., 242: 367-375. doi:10.1078/0044-5231-00110 ![]()
Ebermann E., Hall M. 2004 — A new species of scutacarid mites transferring fungal spores (Acari, Tarsonemina) — Rev. Suisse Zool., 111(4): 941-950. doi:10.5962/bhl.part.80279 ![]()
Ebermann E., Hall M. 2005 — Examination on the distribution and morphology of the mite species Imparipes (I.) apicola (Banks, 1914) (Acari, Scutacaridae) — Mitt. Naturwiss. Ver. Steiermark, 134: 189-197.
Ebermann E., Moser J.C. 2008 — Mites (Acari: Scutacaridae) associated with the red imported fire ant, Solenopsis invicta Buren (Hymenoptera: Formicidae), from Lousiana and Tenessee, U.S.A. — Int. J. Acarol., 34(1): 55-69. doi:10.1080/01647950808683706 ![]()
Ebermann E., Palacios-Vargas J.C. 1988 — Imparipes (Imparipes) tocatlphilus n.sp. (Acari, Tarsonemina, Scutacaridae) from Mexico and Brazil: First record of ricinuleids as phoresy hosts for scutacarid mites — Acarologia, 29: 347-354.
Ebermann E., Krisper G. 2014 — Milben als Mitbewohner in Ameisennestern — In: Wagner H.C. (Ed.). Die Ameisen Kärntens. Verbreitung, Biologie, Ökologie und Gefährdung. Klagenfurt: Naturwissenschaftlicher Verein für Kärnten. pp. 329-342.
Ebermann E., Rodrigues S.R. 2001 — Pygmodispus (Pygmodispus) bicornutus spec. nov., a new phoretic mite species from South America (Acari, Heterostigmata, Scutacaridae) — Stud. Neotrop. Faun. Environ., 36(1): 67-71. doi:10.1076/snfe.36.1.67.8884 ![]()
Ebermann E., Hajiqanbar H., Haddan-Irani-Nejad K. 2003 — New records of phoretic and soil-living mites from Iran (Acari, Heterostigmata, Scutacaridae) — Rev. Suisse Zool., 110(2): 247-253. doi:10.5962/bhl.part.80185 ![]()
Ebermann E., Hall M., Hausl-Hofstätter U., Jagersbacher-Baumann J., Kirschner R., Pfingstl T., Plassnig E. 2013 — A new phoretic mite species with remarks to the phenomenon "Sporothecae" (Acari, Scutacaridae; Hymenoptera, Aculeata) — Zool. Anz., 252 (2): 234-242. doi:10.1016/j.jcz.2012.06.003 ![]()
Ebermann E., Messner M., Jagersbacher-Baumann J. 2016 — First observations on phoresy hosts of Imparipes (Sporichneuthes) dispar Rack, 1964 (Acari: Heterostigmatina: Scutacaridae) — Soil Org., 88(2): 133-138.
Ebermann E., Paill W., Proksch M. 2011 — Laufkäfer als Phoresiewirte für Milbenarten der Gattung Archidispus (Heterostigmata, Scutacaridae) in Kärnten — Carinthia II, 201: 435-444.
Eickwort G.C. 1979 — Mites associated with sweat bees (Halictidae) — In: Rodriguez J.G (Ed.). Recent Advances in Acarology Vol. 1. New York: Academic Press. pp. 575-588. doi:10.1016/B978-0-12-592201-2.50082-8 ![]()
Eickwort G.C. 1990 — Associations of mites with social insects — Ann. Rev. Entomol., 35: 469-488. doi:10.1146/annurev.en.35.010190.002345 ![]()
Eickwort G.C. 1994 — Evolution and life-history patterns of mites associated with bees — In: Houck M.A. (ed.) Mites. Ecological and Evolutionary Analyses of Life-History Patterns. New York, London: Chapman & Hall. pp. 218-251.
Eickwort G.C., Eickwort K.R. 1971 — Aspects of the biology of Costa Rican Halictine bees, II. Dialictus umbripennis (Hymenoptera: Halictidae) and Adaptations of its Caste Structure to Different Climates — J. Kans. Entomol. Soc., 44(3): 343-373.
Elbadry E.A., Yousef A.A., Metwally S.H. 1976 — Life history studies on the soil mite Scutacarus agypticus with description of the immature stages (Acarina: Scutacaridae) — Pedobiologia, 16(3): 167-170.
El-Sharabasy H.M. 2010 — A survey of mite species associated with the red palm weevil, Rhynchophorus ferrugineus (Olivier) in Egypt — Egypt. J. Biol. Pest Control, 20(1): 67-70.
Estébanes-González M.L., Cervantes F.A. 2005 — Mites and ticks associated with some small mammals in Mexico — Int. J. Acarol., 31(1): 23-37. doi:10.1080/01647950508684413 ![]()
Farish D.J., Axtell R.C. 1971 — Phoresy redefined and examined in Macrocheles muscaedomesticae (Acarina: Macrochelidae) — Acarologia, 13(1): 16-29.
Fain A., Baugnée J.-Y. 1996 — Acariens phorétiques ou parasites récoltés sur des insectes du sud de la Belgique. Deuxième note — Bull. Ann. Soc. R. Belge Entomol., 132: 19-33.
Fain A., Miessen G. 1997 — Acariens (Acari) récoltés sur divers Coléoptères (Insecta), principalement des espèces du genre Onthophagus (Scarabaeidae) en Belgique — Bull. Ann. Soc. R. Belge Entomol., 133: 357-362.
Fain A., Baugnée J.-Y., Hidvegi F. 1992 — Acariens phorétiques ou parasites récoltés sur des Hyménoptères et un Homoptère dans la région de Treignes, en Belgique — Bull. Ann. Soc. R. Belge Entomol., 128: 335-338.
Fain A., Noti M.I., Dufrêne M. 1995 — Observations on the mites (Acari) associated with Carabidae (Coleoptera) in Belgium. I. Annotated list of species — Int. J. Acarol., 21(2): 107-122. doi:10.1080/01647959508684051 ![]()
Ferragut F. — Orden Prostigmata [Internet] — [17.08.2015] Revista IDE@-SEA 14:1-8. Available from: http://www.sea-entomologia.org/IDE@/revista_14.pdf ![]()
Friedl A. 2000 — Heimische Ameisenarten als Wirte/Phoresiewirte von bodenbewohnenden Milben der Familie Scutacaridae (Acari, Heterostigmata, Tarsonemina) [Master thesis]—Graz: Institute of Zoology, University of Graz. pp. 60.
Goeze J.A.E. 1780 — Herrn Pastor Gözens neuentdeckte Theile an einigen Insekten — Naturforscher, 14: 93-102.
Gotwald W.H. 1996 — Mites l Live with army ants: A natural history of some myrmecophilous Hitch-hikers, Browsers, and Parasites — . Kans. Entomol. Soc., 69(4) suppl: 232-237.
Greenslade P., Clift A. 2004 — Review of pest arthropods recorded from commercial mushroom farms in Australia — Australas. Mycol., 23(3): 77-93.
Guerra T.J., Romero G.Q., Costa J.C., Lofego A.C., Benson W.W. 2012 — Phoretic dispersal on bumblebees by bromeliad flower mites (Mesostigmata, Melicharidae) — Insect. Soc., 59: 11-16. doi:10.1007/s00040-010-0091-4 ![]()
Hall M., Ebermann E. 2005 — Zoogeographical aspects of some scutacarid mites and their phoresy hosts (Acari, Heterostigmata; Hymenoptera, Aculeata) — Rev. Suisse Zool., 112(1): 215-224. doi:10.5962/bhl.part.80296 ![]()
Hajiqanbar H., Khaustov A. 2014 — First record of the genus Archidispus (Acari: Scutacaridae) from Iran with description of a new species — Pers. J. Acarol., 3(1): 1-8.
Hoey-Chamberlain R., Rust M.K., Klotz J.H. 2013 — A review of the biology, ecology and behaviour of velvety tree ants of North America — Sociobiology, 60(1): 1-10.
Hörnschemeyer T. 2005 — Archostemata Kolbe, 1908 — In: Beutel R.G., Leschen R.A.B. (Eds.) Coleoptera, Vol. I. Morphology and Systematics (Archostemata, Adephaga, Myxophaga, Polyphaga partim). Handbook of Zoology Vol. IV, Arthropoda: Insecta. Berlin: De Gruyter. pp. 157-182.
Jagersbacher-Baumann J. 2007 — Untersuchungen zur Ultrastruktur der mechanosensitiven Ausstattung von Imparipes (Sporichneuthes) dispar Rack, 1964 (Acari, Tarsonemina) [Master thesis] — Graz: Institute of Zoology, University of Graz. pp. 79.
Jagersbacher-Baumann J. 2014 — Species differentiation of scutacarid mites (Heterostigmatina) using multivariate morphometric methods — Exp. Appl. Acarol., 62: 279-292. doi:10.1007/s10493-013-9747-x ![]()
Jagersbacher-Baumann J. 2015 — Traditional and geometric morphometric analyses reveal homogeneity in European Scutacarus acarorum Goeze, 1780 populations (Acari, Scutacaridae, Heterostigmatina) — J. Nat. Hist., 49(19-20): 1173-1190. doi:10.1080/00222933.2014.974705 ![]()
Jagersbacher-Baumann J., Ebermann E. 2012a — Fungal spore transfer and intraspecific variability of a newly described African soil mite (Heterostigmata, Scutacaridae, Heterodispus) — Zool. Anz., 251(2): 101-114. doi:10.1016/j.jcz.2011.05.008 ![]()
Jagersbacher-Baumann J., Ebermann E. 2012b — Heterodispus cordidiscus n. sp., a new soil mite from Egypt, and remarks on the nominal species H. elongatus Trägårdh, 1905 (Acari: Heterostigmatina: Scutacaridae) — Zootaxa, 3520: 56-70.
Jagersbacher-Baumann J., Ebermann E. 2013 — Methods for rearing scutacarid mites (Acari, Heterostigmatina) and the influence of laboratory cultures on morphometric variables — Exp. Appl. Acarol., 59(4): 447-462. doi:10.1007/s10493-012-9621-2 ![]()
Kalúz S. 2001 — First records of mites (Acari, Scutacaridae, Rhagidiidae) from Slovakia — Biologia, 56(5): 524-544.
Kamali K., Ostovan H., Atamehr A. 2001 — A catalog of mites & ticks (Acari) of Iran — Tehran: Scientific Publishing Center of Azad University. pp 192.
Karafiat H. 1959 — Systematik und Ökologie der Scutacariden — In: Stammer H.J. (Ed.) Beiträge zur Systematik und Ökologie mitteleuropäischer Acarina. Band I, Tyroglyphidae und Tarsonemini. Leipzig: Akad. Verlagsgesellschaft Geest & Portig K.-G. pp. 627-712.
Katlav A., Hajiqanbar H., Talebi A.A. 2015 — A contribution to the knowledge of heterostigmatic mites (Acari: Prostigmata) in western Mazandaran Province, Northern Iran — Acarologia, 55(3): 311-320. doi:10.1051/acarologia/20152175 ![]()
Katlav A., Hajiqanbar H., Talebi A.A. 2016 — Two remarkable new species of the Superfamily Pygmephoroidea (Acari: Heterostigmata) associated with beetles (Coleoptera: Carabidae, Staphylinidae) — Ann. Entomol. Soc. Am., 109: 136-144. doi:10.1093/aesa/sav099 ![]()
Khaustov A.A. 2002 — Two new species of mites of the genus Archidispus (Acarina: Heterostigmata: Scutacaridae) from Byelorussia and Crimea — Acarina, 10(2): 155-159.
Khaustov A.A. 2003 — Archidispusdubinini sp. nov., a new species of mites of the family Scutacaridae (Acarina: Heterostigmata) from Ukraine — Acarina, 11(1): 57-60.
Khaustov A.A. 2006 — Two new species of mites of the genus Scutacarus (Acari, Heterostigmata, Scutacaridae) from Ukraine — Vestnik zoologii, 49(2): 161-164.
Khaustov A.A. 2008 — Mites of the family Scutacaridae of Eastern Palaearctic —Akademperiodyka. pp. 290.
Khaustov A.A. 2015a — New records of the genus Scutacarus (Acari: Heterostigmatina: Scutacaridae) from ant nests in Western Siberia, Russia — Acarina, 23(1): 85-91.
Khaustov A.A. 2015b — Mites of the family Scutacaridae (Acari: Pygmephoroidea) associated with Formica fusca L. (Hymenoptera: Formicidae) from Western Siberia, Russia — Acarologia, 55(4): 377-386. oibib10.1051/acarologia/20152177
Khaustov A.A. 2015c — Three new species of myrmecophilous scutacarid mites (Acari: Scutacaridae) from Western Siberia, Russia — Zootaxa, 4013(2): 265-279.
Khaustov A.A. 2015d — Myrmecophilous pygmephoroid mites (Acari: Pygmephoroidea) associated with Lasius flavus (Hymenoptera: Formicidae) in Russia — Zootaxa, 4044(3): 345-370.
Khaustov A.A. 2016a — Myrmecophilous pygmephoroid mites (Acari: Pygmephoroidea) associated with Lasius fuliginosus (Hymenoptera: Formicidae) in Western Siberia, Russia — Int. J. Acarol., 42(2): 92-105. doi:10.1080/01647954.2015.1124921 ![]()
Khaustov A.A. 2016b — Three new species of the family Scutacaridae (Acari: Pygmephoroidea) associated with Lasius umbratus (Hymenoptera: Formicidae) from Western Siberia, Russia — Int. J. Acarol., 42(8): 382-390. doi:10.1080/01647954.2016.1203994 ![]()
Khaustov A.A. 2017a — Two new species of pygmephoroid mites (Acari: Pygmephoroidea: Neopygmephoridae, Scutacaridae) associated with Lasius flavus (Hymenoptera: Formicidae) from Far East of Russia — Int. J. Acarol., 43(3): 232-238. doi:10.1080/01647954.2016.1265585 ![]()
Khaustov A.A. 2017b — Two new species of myrmecophilous scutacarid mites (Acari: Scutacaridae) from Chile — Syst. Appl. Acarol., 22(1): 115-124. doi:10.11158/saa.22.1.12 ![]()
Khaustov A.A., Chydyrov P.R. 2004 — New species of mites of the family Scutacaridae (Acari: Heterostigmata) associated with ants (Hymenoptera, Formicidae) from Turkmenistan — Acarina, 12(2): 87-103.
Khaustov A.A., Husband R.W. 2004 — Three new species of Archidispus Karafiat (Acari: Scutacaridae) phoretic on beetles of the genus Stenolophus (Coleoptera: Carabidae) from the U.S.A. — Int. J. Acarol., 30(3): 239-249. doi:10.1080/01647950408684390 ![]()
Khaustov A.A., Hugo-Coetzee E.A., Ermilov S.G. 2017 — A new genus of the mite family Scutacaridae (Acari: Heterostigmata) associated with Trinervitermes trinervoides (Isoptera: Termitidae) from South Africa. — Zootaxa 4258(5): 462-476. doi:10.11646/zootaxa.4258.5.4 ![]()
Khaustov A.A., Lee, J.-H., Lee S.-J., Park J.-S. 2016 — A new species of the genus Coronipes (Acari: Heterostigmata: Scutacaridae) associated with Reticultitermes speratus kyushuensis (Isoptera: Rhinotermitidae) from Korea — Acarina, 24(1): 17-25.
Knapp J. 2008 — Phoretische Milben auf xylophagen Käfern [Master thesis]— Graz: Institute of Zoology, University of Graz. pp. 90.
Knee W., Beaulieu F., Skevington J.H., Kelso S., Forbes M.R. 2012 — Cryptic species of mites (Uropodoidea: Uroobovella spp.) associated with burying beetles (Silphidae: Nicrophorus): The collapse of a host generalist revealed by molecular and morphological analyses — Mol. Phylogen. Evol., 65: 276-286. doi:10.1016/j.ympev.2012.06.013 ![]()
Kontschán J. 2015 — First record of three mite species (Acari) in Greece collected on commercial bumblebee (Hymenoptera: Apidae: Bombus terrestris Linnaeus, 1758) — Ecol. Mont., 2(2): 158-161.
Kontschán J., Jeon M.J., Hwang J.M., Seo H.Y. 2016 — First record of four bee (Hymenoptera: Apidae) associated mite specis (Acari) from Democratic People's Republic of Korea — J. Species Res., 5(1): 27-30. doi:10.12651/JSR.2016.5.1.027 ![]()
Krantz G.W., Walter D.E. 2009 — A manual of Acarology — Lubbock (TX): Texas Tech University Press. pp 807.
Krivolutsky D.A., Lebedeva N.V., Gavrilo MV. 2004 — Soil microarthropods in the feathers of Antarctic Birds — Dok. Biol. Sciences, 397: 342-345.
Kuhlmann M. 1998 — Nachweise mit Bienen und Wespen (Hymenoptera Aculeata) assoziierter Milben (Acari) und Fächerflügler (Strepsiptera) — Linz. Biol. Beitr., 30(1): 69-80.
Kurosa K. 1970 — The scutacarid mites of Japan.I. Imparipes (Archidispus) amarae sp. nov. — Bull. Nat. Sci. Mus., 13(2): 121-127.
Kurosa K. 1972a — The scutacarid mites of Japan.II. Lophodispus latus gen. et sp. nov. — Bull. Nat. Sci. Mus., 15(1): 29-35.
Kurosa K. 1972b — The scutacarid mites of Japan. III. Three new species of the subgenus Archidispus — Bull. Nat. Sci. Mus., 15(4): 623-636
Kurosa K. 1974 — The scutacarid mites of Japan. IV. Three New Species of Imparipes (Archidispus) associated with Stenolophine ground beetles — Annot. Zool. Jpn., 47(3): 160-169.
Kurosa K. 1976 — The Scutacarid mites of Japan. V. Three new Archidispus associated with Stenolophine ground beetles — Kontyû, 44(2): 172-183.
Kurosa K. 1977 — The Scutacarid mites associated with ground beetles in Japan — In: Satsusa M, Aoki J (Eds.). Contributions to Acarology in Japan. Tokyo: Zukan-no-Hokuryukan Co. Ltd. pp. 371-404.
Kurosa K. 1978 — The Scutacarid Mites of Japan. VI. Four more Archidispus associated with Stenolophine ground beetles — Annot. Zool. Jpn., 51(2): 111-124.
Kurosa K. 1980 — Caraboacaridae, Pygmephoridae, Scutacaridae. — In: Ehara S. (Ed.): Illustrations of the mites and ticks of Japan. Tokyo: Zenkoku Nôson Kyôiku Kyôkai Inc. pp. 214-241.
Kurosa K. 1983 – The Scutacarid mites of Japan. VII. Revised definition of the genera Archidispus and Imparipes, with descriptions of five new Archidispus — Annot. Zool. Jpn., 56(4): 315-331.
Kurosa K. 1984 — The Scutacarid Mites of Japan. VIII. Further Archidispus associated with Stenolophine ground beetles — Kontyû, 52(2): 243-252.
Kurosa K. 1989 — The Scutacarid Mites of Japan. IX. Five new Archidispus associated with ground beetles — Jpn. J. Entomol., 57(1): 3-24.
Kurosa K. 1990 — The Scutacarid Mites of Japan. X. Three new Archidispus associated with Stenolophus (Coleoptera, Carabidae) — Jpn. J. Entomol., 58(1): 1-10.
Kurosa K. 1991 — The Scutacarid Mites of Japan. XI. Four new Archidispus associated with ground beetles of the tribes Nebriini, Bembidiini and Patrobini — Jpn. J. Entomol., 59(3): 539-554.
Kurosa K. 2003 — The Scutacarid Mites of Japan. XII. Two new Archidispus associated with Stenolophine carabid beetles — Spec. Bull. Jpn. Soc. Coleopterol., 6: 41-49.
Kurosa K. 2005 — The Scutacarid mites of Japan. XIII. Two new Archidispus phoretic on carabid beetles mainly belonging to the Pterostichini and Harpalini — Elytra, 33(1): 3-12.
Kurosa K. 2009 — The Scutacarid Mites of Japan. XIV. A new species of Archidispus phoretic on Stenolophine carabid beetles — J. Acarol. Soc. Jpn., 18(2): 85-90. doi:10.2300/acari.18.85 ![]()
Lang J.D., Mahunka S. 1977 — Archidispus sphecis sp. n. (Acari: Tarsonemida), a new mite species on digger wasps in Vietnam — Parasitol. Hung., 10: 125-127.
Larsson J.I.R. 2007 – Cytological variation and pathogenicity of the bumble bee parasite Nosema bombi (Microspora, Nosematidae) — J. Invertebr. Pathol., 94: 1-11. doi:10.1016/j.jip.2006.07.006 ![]()
Lenoir A., Aron S., Cerdá X., Hefetz A. 2009 — Cataglyphis desert ants: a good model for evolutionary biology in Darwin's anniversary year- A review — Israel J. Entomol., 39: 1-32.
Lindquist E. 1975 — Associations between mites and other arthropods in forest floor habitats — Can. Entomol., 107: 425-437. doi:10.4039/Ent107425-4 ![]()
Loghmani A., Hajiqanbar H., Talebi A.A. 2014 — New records of mites of the superfamily Pygmephoroidea (Acari: Heterostigmatina) associated with insects from northeastern Iran and new host records — Syst. Appl. Acarol., 19(2): 154-159. doi:10.11158/saa.19.2.5 ![]()
Lombardini G. 1960 — Acari nuovi. XLI — Redia, 45: 255-261.
Maggi M., Lucia M., Abrahamovich A.H. 2011 — Study of the acarofauna of native bumblebee species (Bombus) from Argentina — Apidologie, 42:280-292.
Mahunka S. 1963 — Beiträge zur Kenntnis der Milbenfauna (Acari) von Säugetiernestern — Acta Zool. Acad. Sci. Hung., 9(3-4): 355-373.
Mahunka S. 1964a — Neue Scutacariden aus Angola/ Acari: Tarsonemini/ — Publ. Cult. Co. Diaman. Angola, 68: 115-137.
Mahunka S. 1964b — Beiträge zur Kenntnis der in Ställen und Stallmist lebenden Milben (Acari) — Opusc. Zool. Budapest, 5(1): 99-105.
Mahunka S. 1965a — Ergebnisse der zoologischen Forschungen von Dr. Z. Kaszab in der Mongolei; 30. Acari: Pyemotidae and Scutacaridae — Ann. Hist.-Nat. Mus. Nat. Hung., 57: 435-441.
Mahunka S. 1965b — Die Tarsonemini (Acari-)Fauna Ungarischer Dauerwiesen und Hutweiden — Acta Zool. Acad. Sci. Hung., 11(1-2): 137-151.
Mahunka S. 1966 — Zwei Neue Arten aus der Gruppe Tarsonemini aus China (Acari) — Vestn. Cesk. Spol. Zool., 30(4): 315-318.
Mahunka S. 1967a — 83. Acari: Pyemotidae and Scutacaridae; Ergebnisse der zoologischen Forschungen von Dr. Z. Kaszab in der Mongolei — Reichenbachia, 9(1): 1-13.
Mahunka S.1967b — Beiträge zur Kenntnis der tschechoslowakischen Tarsonemini-Fauna — Vestn. Cesk. Spol. Zool., 31(3): 240-244.
Mahunka S. 1967c — A survey of the Scutacarid (Acari: Tarsonemini) fauna of Australia — Aust. J. Zool., 15: 1299-1323. doi:10.1071/ZO9671299 ![]()
Mahunka S. 1968a — The scientific results of the Hungarian Soil Zoological Expedition to South America. 4 Acari: Scutacaridae I. A Survey of the Scutacarid Fauna of Chile — Acta Zool. Acad. Sci. Hung., 14(1-2): 139-166.
Mahunka S. 1968b — Beiträge zur Tarsonemini-Fauna Ungarns V. (Acari, Trombidiformes) — Folia Entomol. Hung., 21(8): 127-135.
Mahunka S. 1969a — Pyemotidae und Scutacaridae IV. Ergebnisse der zoologischen Forschungen von Dr. Z. Kaszab in der Mongolei (Acari) — Reichenbachia, 12(10): 83-112.
Mahunka S. 1969b — Imparipes (I.) eickworti sp. n., a new scutacarid mite (Acari, Tarsonemina) from Dialictus umbripennis Ellis (Hym.) — Parasitol. Hung., 2: 153-158.
Mahunka S. 1969c — Imparipes (Archidispus) nickelli sp. n., a new mite species (Acari, Tarsonemina) from a carabid species in the United States of America — Parasitol. Hung., 2: 149-152.
Mahunka S. 1970a — Zwei neue Heterodispus-Arten und einige interessante, in Ameisenhaufen lebende Milben aus Ungarn (Acari: Tarsonemina) — Folia Entomol. Hung., 23(16): 313.331.
Mahunka S. 1970b — Beiträge zur Kenntnis der Milbenfauna der Ötztaler-Alpen 1. Tarsoneminen-Arten aus der Umgebung von Obergurgl — Opusc. Zool. Budapest, 10(2): 271-289.
Mahunka S. 1970c — Two new Scutacarid Mites (Acari, Tarsonemina) from Pogonomyrmex occidentalis (Hymenoptera) in the United States of America — Parasitol. Hung., 3: 87-96.
Mahunka S. 1970d — Dudichiana foveolata gen. nov., sp. nov., sowie einige andere neue und interessante Milbenarten aus Ameisenhaufen — Opusc. Zool. Budapest, 10(1): 143-149.
Mahunka S. 1971 — Tarsonemina (Acari) species from Korea. Zoological Collectings of the Hungarian Natural History Museum in Korea (Nr.3.) — Acta Zool. Hung., 17(3-4): 271-294.
Mahunka S. 1972a — Neue und interessante Milben aus dem Genfer Museum. X. Milben aus Kleinsäugernestern Nordtirols (Österreich) — Ber. Naturwiss.-Med. Ver. Innsbruck, 59: 57-62.
Mahunka S. 1972b — Tetüatkák- Tarsonemina. Fauna Hungariae 110. XVIII. kötet, 16. füzet (Arachnoidea). Budapest: Akademiai Kiado. pp 215.
Mahunka S. 1973a — Auf Insekten lebende Milben (Acari: Acarida, Tarsonemida) aus Afrika. I. — Acta Zool. Acad. Sci. Hung., 19(1-2): 75-123.
Mahunka S. 1973b — Auf Insekten lebende Milben (Acari: Acarida, Tarsonemida) aus Afrika. II. — Acta Zool. Acad. Sci. Hung., 19(3-4): 289-337.
Mahunka S. 1973c — Neue Tarsonemiden (Acari) aus der Mongolei — Ann. Hist.-Nat. Mus. Nat. Hung., 65:309-315.
Mahunka S. 1974a — Auf Insekten lebende Milben (Acari: Acarida, Tarsonemida) aus Afrika. III. — Acta Zool. Acad. Sci. Hung., 20(1-2): 137-154.
Mahunka S. 1974b — Auf Insekten lebende Milben (Acari: Acarida, Tarsonemida) aus Afrika. IV. — Acta Zool. Acad. Sci. Hung., 20(3-4): 367-402.
Mahunka S. 1974c — Beiträge zur Kenntnis der an Hymenopteren lebenden Milben (Acari), I. — Ann. Hist.-Nat. Mus. Nat. Hung., 66: 389-394.
Mahunka S. 1974d — Some New Data to the Scutacarid Fauna of Australia (Acari) — Folia Entomol. Hung., 27(2): 97-102.
Mahunka S. 1975a — Auf Insekten lebende Milben (Acari: Acarida, Tarsonemida) aus Afrika. V. — Acta Zool. Acad. Sci. Hung., 21(1-2): 39-72.
Mahunka S. 1975b — Beiträge zur Kenntnis der Tarsonemiden (Acari) von Kleinsäugernestern aus der Umgebung von Ljubljana (Jugoslawien) — Parasitol. Hung., 8: 75-83.
Mahunka S. 1975c — Neue und auf Insekten lebende Milben aus Australien und Neu-Guinea (Acari: Acarida, Tarsonemida) — Ann. Hist.-Nat. Mus. Nat. Hung., 67: 317-325.
Mahunka S. 1977a — The examination of myrmecophilous tarsonemid mites based on the investigations of Dr. C. W. Rettenmeyer (Acari). I. — Acta Zool. Acad. Sci. Hung., 23(1-2): 99-132.
Mahunka S. 1977b — The examination of myrmecophilous tarsonemid mites based on the investigations of Dr. C. W. Rettenmeyer (Acari). II. — Acta Zool. Acad. Sci. Hung., 23(3-4): 341-370.
Mahunka S. 1977c — Neue und Interessante Milben aus dem Genfer Museum XIX. Einige Angaben zur Kenntnis der Milbenfauna der Ameisen-Nester (Acari: Acarida, Tarsonemida) — Arch. Sci. Genève, 30(1): 91-106.
Mahunka S. 1977d — Three new scutacarid species (Acari: Tarsonemida) from the nests of small mammals in the United States of America — Parasitol. Hung., 10: 129-134.
Mahunka S. 1979 — Auf Insekten lebende Milben aus Afrika. VI. (Acari: Acarida, Tarsonemida) — Acta Zool. Acad. Sci. Hung., 25(1-2): 127-157.
Mahunka S. 1981 — The Pygmephoroid fauna of the Hortobágy National Park (Acari: Tarsonemida) — Fauna Horto. Nat. Park, 1: 343-370.
Mahunka S. 1982 — Zwei neue Scutacariden-Arten aus Mexico (Acari: Tarsonemina) — Misc. Zool. Hung., 1: 133-138.
Mahunka S. 1986 — Tarsonemids of the Kiskunság National Park (Acari) — In: Mahunka S. (Ed.). The Fauna of the Kiskunság National Park I, Series: Natural History of the National Parks of Hungary 4. Budapest: Akadémiai Kiadó. pp. 435-455.
Mahunka S., Mahunka-Papp L. 1980 — Beiträge zur Kenntnis der europäischen myrmecophilen Tarsoneminen (Acari). I. — Folia Entomol. Hung., 41(33): 283-292.
Mahunka S., Moser J.C. 1980 — Scutacarus scolyti sp. n. a new scutacarid species (Acari: Tarsonemina) from Germany — Parasitol. Hung., 13: 99-102.
Mahunka S., Philips J.R. 1978 — Tarsonemid mites associated with birds in the USA (Acarina: Tarsonemida) — Folia Entomol. Hung., 31(2): 191-200.
Mahunka S., Zaki A.M. 1992 — Phoretic Scutacarus mites (Acari: Heterostigmata) from Egypt — Folia Entomol. Hung., 52: 59-61.
Messner M.J. 2001 — Über Morphologie, Entwicklung, Ernährung und Phoresie bei Imparipes (Sporichneuthes) dispar Rack, 1964 (Acari, Tarsonemina) [Master thesis]— Graz: Institute of Zoology, University of Graz. pp. 82.
Metwali S.H. 1981 — Studies on Tarsonemini (Acarina) associated with ants in forests of Poland — Entomol. Mitt. Zool. Mus. Hamburg, 7(112): 87-100.
Metwali S.H. 1984 — Survey on the Family Scutacaridae (Acari) in Egypt II — Acarologia, 25(3): 242-248.
Metwali S.H, Ahmed A.E.E. 1987 — New Scutacarid Mites Associated with Ants in Egypt (Acari: Scutacaridae) — Bull. Entomol. Soc. Egypt, 67: 23-34.
Michael A.D. 1884 — The Hypopus Question, or the Life-History of certain Acarina — J. Linn. Soc. Lond. (Zool.), 17: 371-394. doi:10.1111/j.1096-3642.1884.tb02247.x ![]()
Modak A. 1991 — Taxonomic studies of house dust mite fauna of West Bengal with reference to the biology and ecology of Dermatophagoides pteronyssus [PhD thesis]— Burdwan: University of Burdwan. pp 306. (Available from: http://hdl.handle.net/10603/68548 [accessed 24.10.2016] ![]() ).
).
Moser J.C., Blomquist S.R. 2011 — Phoretic arthropods of the Red Imported Fire Ant in Central Louisiana — Ann. Entomol. Soc. Am., 104(5): 886-894. doi:10.1603/AN11010 ![]()
Moser J.C., Eidmann H.H., Regnander J.R. 1989 — The mites associated with Ips typographus in Sweden — Ann. Entomol. Fenn., 55: 23-27.
Nemestóthy K.K., Mahunka S. 1981 — Tarsoneminen (Acari) aus dem Naturschutzgebiet Wacholders "Borókás" bei Barcs, Ungarn — Dun. Dolg., 2: 157-166.
Norton R.A. 1973 — Phoretic Mites Associated with the Hermit Flower Beetle, Osmoderma eremicola Knoch (Coleoptera: Scarabaeidae) — Am. Midl. Nat., 90(2): 447-449. doi:10.2307/2424466 ![]()
Norton R.A. 1977 — An example of phoretomorphs in the mite family Scutacaridae — J. GA. Entomol. Soc., 12(3): 185-186.
Norton R.A., Ide G.S. 1974 — Scutacarus baculitarsus agaricus n. subsp. (Acarina: Scutacaridae) from Commercial Mushroom Houses, with Notes on Phoretic Behavior — J. Kans. Entomol. Soc., 47(4): 527-534.
Okabe K. 2013 — Ecological characteristics of insects that affect symbiontic relationships with mites — Entomol. Sci., 16: 363-378.
Ordway E. 1964 — Sphecodes pimpinellae and other enemies of Augochlorella (Hymenoptera: Halictidae) — J. Kans. Entomol. Soc., 37: 139-152.
Oudemans A.C. 1913 — Acarologisches aus Maulwurfsnestern — Arch. Naturg., 79(9):68-136.
Paik Y.H., Takaoka M., Matsuoka H., Ishii A. 1992 — Mite fauna and mite antigen in house dust from houses in Seoul, Korea — Jpn. J. Sanit. Zool., 43(1): 29-35. doi:10.7601/mez.43.29 ![]()
Paoli G. 1911 — Monografia dei Tarsonemidi — Redia, 7: 215-281.
Parmentier T., Dekoninck W., Wenseleers T. 2015 — Metapopulation processes affecting diversity and distribution of myrmecophiles associated with red wood ants — Basic Appl. Ecol., 16(6): 553-562. doi:10.1016/j.baae.2015.04.008 ![]()
Philips J.R., Poole A., Holt D. 1988 — Nest mites of ospreys and short-eared owls in Massachusetts salt marshes, USA — Progress in Acarology, 2: 305-307.
Rack G. 1964 — Scutacaridae von Hamburg (Acarina, Trombidiformes) — Mitt. Hamburg Zool. Mus. Inst., Kosswig-Festschrift: 185-194.
Rack G. 1966 — Scutacaridae von Hamburg. II. (Acarina, Trombidiformes) — Abh. Verh. Naturwiss. Ver. Hamburg, 10: 97-112.
Rack G. 1973 — Archidispus pterostichi sp. n. und die Gattung Archidispus KARAFIAT, 1959 (Acarina, Trombidiformes, Scutacaridae) — Entomol. Mitt. Zool. Mus. Hamburg, 80(4): 319-326.
Rettenmeyer C.W. 1961a — Behaviour, abundance and host specificity of mites found on neotropical army ants (Acarina; Formicidae: Dorylinae) — XI. Internationaler Kongress für Entomologie Wien, Sonderdruck aus den Verhandlungen. Band 1: 610-612.
Rettenmeyer C.W. 1961b — Arthropods associated with Neotropical army ants with a review of the behaviour of these ants (Arthropoda: Formicidae: Dorylinae) [PhD thesis] — Kansas: University of Kansas. pp. 604.
Rettenmeyer C.W., Rettenmeyer M.E., Joseph J., Berghoff S.M. 2011 — The largest animal association centered on one species: the army ant Eciton burchellii and its more than 300 associates — Insect. Soc., 58: 281-292. doi:10.1007/s00040-010-0128-8 ![]()
Revainera P., Lucia M., Abrahamovich A.H., Maggi M. 2014 — Spatial aggregation of phoretic mites on Bombus atratus and Bombus opifex (Hymenoptera: Apidae) in Argentina — Apidologie, 45: 579-589.
Rodrigues S.R., Marchini L.C., Carbonari J.J. 2001 — Ácaros das famílias Scutacaridae e Pygmephoridae (Acari: Heterostigmata) associados a besouros coprófagos (Coleoptera: Scarabaeidae) no Brasil — Neotrop. Entomol., 30(3): 387-390. doi:10.1590/S1519-566X2001000300008 ![]()
Roy S., Gupta S.K., Debnath N. 2016 — Some new reports of insect associated mites from west Bengal — Glob. J. Res. Anal., 5(6): 402-403.
Samšiňák K. 1989 — Mites on flies of the family Sphaeroceridae II — Acarologia, 30(2): 85-105.
Schlagbauer J. 1997 — Untersuchungen zur Taxonomie und Lebensweise an Milben der Gattung Lophodispus Kurosa, 1972 (Acari, Heterostigmata, Scutacaridae): L. irregularis (Mahunka, 1971) und L. bulgaricus Dobrev, 1992 [Master thesis] — Graz: Institute of Zoology, University of Graz. pp. 160.
Schousboe C. 1986. — On the biology of Scutacarus acarorum Goeze (Acari: Trombidiformes) — Acarologia, 27(2): 151-158.
Schwarz H.H., Huck K. 1997 — Phoretic mites use flowers to transfer between foraging bumblebees — Insect. Soc., 44: 303-310.
Seastedt T.R., Reichmann O.J., Todd T.C. 1986 — Microarthropods and Nematodes in Kangaroo Rat Burrows — Southw. Natural., 31(1): 114-116.
Seifert B. 1996 — Ameisen: beobachten, bestimmen. Augsburg: Weltbild Verlag. pp. 352.
Sevastianov V.D. 1965 — On the number of mites carried and exterminated by the ant Lasius fuliginosus Latr. during a season — Zool. Zh., 44(11): 1651-1660.
Sevastianov V.D. 1983 — New species of the genus Scutacarus (Trombidiformes, Scutacaridae) — Zool. Zh., 62(10): 1487-1496.
Sevastyanov V.D., Uzhevskaya S.F. 2003 — Mites of the genus Tarsonemina (Heterostigmata, Trombidiformes) dwelling in mammalian nests and burrows — Problems of Modern Parasitology II. Proceedings of the International Conference and III Congress of Parasitological Society at RAS: 103-104.
SiNAViMo, Sistema Nacional Argentino de Vigilancia y Monitoreo de plagas. — Bothynus striatellus [Internet] — [29.06.2015]. (Available from: http://www.sinavimo.gov.ar/plaga/bothynus-striatellus ![]() ).
).
Sklyar V.E., Sevastianov V.D. 1997 — Mites of the cohort Tarsonemina in the rodent nest microhabitats of the Donetsk Azov Sea area — Vestn. Zool., 31: 45-49.
Skoracka A., Magalhães S., Rector B.G., Kuczyński L. 2015 — Cryptic speciation in the Acari: a function of species lifestyles or our ability to separate species? — Exp. Appl. Acarol., 67: 165-182. doi:10.1007/s10493-015-9954-8 ![]()
Sobhi M., Hajiqanbar H., Mortazavi A. 2017 — First record of the rare genus Lophodispus (Acari: Heterostigmata: Scutacaridae) from Iran with description of a new species associated with ants (Hymenoptera: Formicidae) — Int. J. Acarol., 43(3): 239-244. doi:10.1080/01647954.2016.1256910 ![]()
Sosnina E.F., Sevastianov V.D. 1975 — Mites of cohort Tarsonemina (Trombidiformes) from the nests of small mammals in the forest zone of the Crimean Mountains — Rev. Entomol. URSS, 1: 202-214.
Spicka E.J. 1981 — Ectoparasites and other arthropod associates on two subspecies of plains pocket gophers: Geomys bursarius illinoensis and Geomys bursarius missourensis — Can. J. .Zool., 59(10): 1903-1908. doi:10.1139/z81-259 ![]()
Štorkán J. 1936 — Einige Scutacaridae aus Bulgarien — Mitt. Königl. Naturwissensch. Inst. Sofia, 9: 28-32.
Štorkán J. 1940 — Myrmekofilní Acari z Čech — Vest. Cesk. Zool. Spol., 8: 166-172.
Suski Z.W. 1973 — A revision of Siteroptes cerealium (Kirchner) complex (Acarina, Heterostigmata, Pyemotidae) — Ann. Zool., 30(17): 509-535.
Szymkowiak P., Górski G., Bajerlein D. 2007 — Passive dispersal in arachnids — Biol. Lett., 44(2): 75-101.
Trägårdh I. 1905 — Acariden aus Ägypten und dem Sudan. Zugleich ein Beitrag zur Kenntnis der Entwicklungsgeschichte der Gattungen Phytoptipalpus, Pimeliaphilus, Pterygosoma und Podapolipus — Results of the Swedish Zoological Expedition to Egypt and the White Nile 1901 under the direction of L.A. Jägerskiöld, Part II, (20): 1-124.
Travis B.V. 1941 — Notes on the Biology of the Fire Ant Solenopsis geminata (F.) in Florida and Georgia — Fla. Entomol., 24(1): 15-22. doi:10.2307/3492286 ![]()
Truck A. 2012 — Zur Verbreitung von Dung und Kompost bewohnenden Milbenarten im Süden Österreichs (Acari, Heterostigmata, Scutacaridae) [Master thesis] — Graz: Institute of Zoology, University of Graz. pp. 45.
Vega F.E., Blackwell M. 2005 — Insect-fungal associations: Ecology and Evolution — Oxford: University Press. pp. 352.
Vitzthum H. 1919 — Neue myrmecophile Milben — Zool. Anz., 24(8): 185-207.
Vitzthum H. 1927 — Eine Notiz über Parasitus vesparum Oudemans 1905 und Fannia scalaris (Fabricius 1794) — Zeitschr. Ins.-Biol., 22: 46-48.
Vitzthum H. 1928 — Acarologische Beobachtungen (13. Reihe) — Zool. Anz., 75: 281-295.
Walter D.E., Proctor H.C. 1999 — Mites: Ecology, evolution, and behavior — Wallingford, Oxon, UK: CAB International. pp. 322.
Wagner H.C. (ed.). 2014 — Die Ameisen Kärntens. Verbreitung, Biologie, Ökologie und Gefährdung — Naturwissenschaftlicher Verein für Kärnten, Klagenfurt. pp. 464.
Wang C., Powell J.E., O'Connor B.M. 2002 — Mites and nematodes associated with three subterranean termite species (Isoptera: Rhinotermitidae) — Fla. Entomol., 85(3): 499-506. doi:10.1653/0015-4040(2002)085[0499:MANAWT]2.0.CO;2 ![]()
Witt R. 1998 — Wespen beobachten, bestimmen — Augsburg: Weltbild Verlag. pp. 360.
Womersley H. 1955 — The acarina fauna of mutton birds' nests on a Bass Strait Island — Aust. J. Zool., 3(3): 412-438. doi:10.1071/ZO9550412 ![]()
Yousef A.E.A., Metawally S.H. 1973 — A New Species of the Genus Scutacarus (Acarina: Scutacaridae) in Egypt — Acarologia, 15(3): 457-460.
Zahradník J. 1985 — Käfer Mittel- und Nordwesteuropas. Ein Bestimmungsbuch für Biologen und Naturfreunde — Hamburg, Berlin: Paul Parey. pp. 498.
Zaki A.M., Osman A.A., Darwish E.T.E. 1987 — Scutacarus longitarsus Berl. (Acarina: Scutacaridae) phoretic on the sphaerocerid fly, Leptocera nigra Olivier (Diptera: Sphaeroceridae), on fig trees in Egypt — Fol. Entomol. Hung., 48: 245-246.
Zhang Z.-Q., Pesic V., Smit H., Bochkov A.V., Khaustov A.A., Baker A., Wohltmann A., Wen T.-H., Amrine J.W., Beron P., Lin J.-Z., Gabrys G., Husband R. 2011 — Order Trombidiformes Reuter, 1909. In: Zhang Z.-Q. (Ed.). Animal biodiversity: an outline of higher-level classification and survey of taxonomic richness — Zootaxa, 3148: 129-138.
ANNEX: LIST OF SCUTACARID SPECIES ASSOCIATED WITH DIFFERENT HOSTS
I. Scutacarid species associated with Arachnida, Entognatha and Ectognatha (excl. Coleoptera and Aculeata)
II. Scutacarid species associated with Carabidae (Hexapoda, Ectognatha, Coleoptera)
III. Scutacarid species associated with Coleoptera excl. Carabidae (Hexapoda, Ectognatha)
IV. Scutacarid species associated with Apoidea and Vespoidea excl. Formicidae (Hexapoda, Ectognatha, Aculeata)
V. Scutacarid species associated with Formicidae excl. Formicinae and Myrmicinae (Hexapoda, Ectognatha, Aculeata, Vespoidea)
VI. Scutacarid species associated with Formicinae (Hexapoda, Ectognatha, Aculeata, Formicidae)
VII. Scutacarid species associated with Myrmicinae (Hexapoda, Ectognatha, Aculeata, Formicidae)
VIII. Scutacarid species associated with Aves and Mammalia
The following information will be given for each host in telegraphic style: associated scutacarid species; distribution area; type of association (i = inquiline, ip = inquiline and phoretic, nk = not known, p = phoretic); references and remarks (if available)
I. SCUTACARID SPECIES ASSOCIATED WITH ARACHNIDA, ENTOGNATHA AND ECTOGNATHA (EXCL. COLEOPTERA AND ACULEATA)
Arachnida
Acari
Haemogamasidae: Haemogamasus ambulans
Scutacarus talpae (Oudemans, 1913); Netherlands; p; Oudemans 1913; in nest of Talpa europea
Parasitidae: Parasitellus fucorum
Scutacarus acarorum (Goeze, 1780); Holarctic; p; Rack 1964, Chmielewski 1971, Schousboue 1986, Schwarz & Huck 1997, Khaustov 2008, Baumann unpubl.; on Bombus spp. (→ hyperphoresy!) or in nests of Bombus spp.
Scutacarus deserticolus Mahunka, 1969; Austria; p; Ebermann 1991a; on Bombus spp. (→ hyperphoresy!) or in nests of Bombus spp.
Aranaea
Nemesiidae: Stenoterommata iguazu, S. platense, S. uruguai
Scutacarus adgregatus Ebermann & Goloboff, 2002; Argentina; p; Ebermann & Goloboff 2002
Scutacarus araneophilus Ebermann & Goloboff, 2002; Argentina; p; Ebermann & Goloboff 2002
Ricinulei
Ricinoididae: Cryptocellus boneti
Imparipes (Imparipes) tocatlphilus Ebermann & Palacios-Vargas, 1988; Brazil, Mexico; p; Ebermann & Palacios-Vargas 1988; also in soil, guano from caves
Hexapoda: Entognatha
Diplura
Campodeidae: Campodea sp.
Imparipes (Imparipes) intentatus Khaustov, 2008; Ukraine; p; Khaustov 2008
Hexapoda: Ectognatha
Dermaptera
Forficulidae
Imparipes (Imparipes) histricinus Berlese, 1903; Italy, Sudan; p; Trägårdh 1905, Paoli 1911; also in soil, moss; is Imparipes forficulae sensu Trägårdh
Diptera
Anisopodidae, Ceratopogonidae, Chironomidae, Conopidae, Dolichopodidae, Mycetophilidae, Psychodidae, Scatopsidae
Imparipes (Sporichneuthes) dispar Rack, 1964; Austria; p; Ebermann et al. 2016
Muscidae: Musca domestica
Scutacarus sp.; India; p; Roy et al. 2016
Phoridae: Aphiochaeta enderleini
Rettenmeyerella petropolitana (Vitzthum, 1928); Brazil; p; Vitzthum 1928
Phoridae: Megaselia dakotensis
Scutacaropsis baculitarsus agaricus (Norton & Ide, 1974); USA (Delaware, Pennsylvania); p; Norton & Ide 1974; also in mushroom-bedding compost
Phoridae: Megaselia halterata
Scutacaropsis baculitarsus (Mahunka, 1968); Chile, Europe; p; Binns 1979; also in debris
Phoridae
Rettenmeyerella petropolitana solenifera Mahunka, 1977; Costa Rica; p; Mahunka 1977a; Phoridae associated with Eciton hamatum
Scutacarus australiensis Mahunka, 1967; Australia; p; Greenslade & Clift 2004; also in compost
Sciaridae
Imparipes (Sporichneuthes) dispar Rack, 1964; Austria; p; Ebermann et al. 2016
Scutacarus australiensis Mahunka, 1967; Australia; p; Greenslade & Clift 2004; also in compost
Sphaeroceridae: Leptocera nigra
Scutacarus longitarsus (Berlese, 1905); cosmopolitic; p; Zaki et al. 1987, Khaustov 2008; also in banks of rivers and lakes
Sphaeroceridae: Leptocera fontinalis
Scutacarus longitarsus (Berlese, 1905); USA (Arizona); p; Samšiňák 1989
Sphaeroceridae: Copromyza costalis, Copromyza acutangula, Coproica pseudolugubris
Imparipes (Imparipes) penicillatus Mahunka, 1967; Mongolia; p; Mahunka 1973c; also in soil
Heteroptera
Reduviidae: Pasira mediterranea
Imparipes (Imparipes) nikitensis Khaustov, 2005; Crimea; p; Khaustov 2008
Hymenoptera: Terebrantia/Parasitica
Eucoilidae, Proctotrupoidea, Pteromalidae
Imparipes (Sporichneuthes) dispar Rack, 1964; Austria; p; Ebermann et al. 2016
Isoptera
Rhinotermitidae: Coptotermes formosanus
Coronipes samsinaki (Mahunka, 1966); China; i; Mahunka 1966
“Scutacaridae"; China; ip; Wang et al. 2002
Rhinotermitidae: Reticulitermes flavipes
“Scutacaridae"; China; ip; Wang et al. 2002
Rhinotermitidae: Reticulitermes speratus kyushuensis
Coronipes sperati Khaustov et al. 2016; South Korea; p; Khaustov et al. 2016
Rhinotermitidae: Reticulitermes virginicus
Imparipes (Imparipes) adleri Delfinado & Baker, 1976; USA; i; Delfinado & Baker 1976
Rhinotermitidae: Reticulitermes sp.
Imparipes (Imparipes) cf. adleri Delfinado & Baker, 1976; Spain; ip; Baumann & Ferragut unpubl.
Termitidae: Odontotermes nolaensis
Archidispus brevisetus (Mahunka, 1964); Angola; i; Mahunka 1964a
Termitidae: Pericapritermes appellans
Imparipes (Imparipes) termitophilus Silvestri, 1918; Western Africa; n.k.; Khaustov et al. 2016
Termitidae: Pseudacanthotermes spiniger
Imparipes (Imparipes) angolensis Mahunka, 1964; Angola; p; Mahunka 1964a
Scutacarus mirabilis Mahunka, 1964; Angola; p; Mahunka 1964a
Termitidae: Trinervitermes trinervoides
Gerdalbertia elongata Khaustov, Hugo-Coetzee & Ermilov, 2017; South Africa; ip; Khaustov et al. 2017
Orthoptera
Caelifera
Imparipes (Imparipes) rectangulatus Mahunka, 1977; Costa Rica, Ecuador, USA (Kansas); p; Mahunka 1977a
Gryllidae: Brachytrupes membranaceus
Heterodispus pilosus Mahunka, 1964; Angola; p; Mahunka 1964a
Gryllotalpidae: Gryllotalpa gryllotalpa
Heterodispus severus Khaustov, 2008; Ukraine; p; Khaustov 2008
“Insecta" (not further classified in literature)
Archidispus certus Mahunka, 1974; Ghana; p; Mahunka 1974b
Archidispus magyari Mahunka, 1974; Ghana; p; Mahunka 1974b
Archidispus telekii Mahunka, 1974; Ghana; p; Mahunka 1974b
Diversipes pygmodispoides Mahunka, 1973; Tanganyika; p?¸ Mahunka 1973a; found in soil traps together with insects
Heterodispus (Teherodispus) konrathi Mahunka, 1974; Ghana; p; Mahunka 1974b
Pygmodispus (Allodispus) angulosus Mahunka, 1979; South Africa; p; Mahunka 1979
Pygmodispus (Allodispus) brachiosus Paoli, 1911; Tanganyika; p?¸ Mahunka 1973a; found in soil traps together with insects
Pygmodispus (Allodispus) pseudocoprophilus Mahunka, 1979; Ghana; p; Mahunka 1979
Reductacarus africanus Mahunka, 1975; Ghana; p; Mahunka 1975a
Scutacarus appropinquatus Mahunka, 1979; Tanzania; p; Mahunka 1979
Scutacarus endroedii Mahunka, 1979; South Africa; p; Mahunka 1979
Scutacarus ghanensis Mahunka, 1974; Ghana; p; Mahunka 1974a
Scutacarus parapelvus Mahunka, 1974; Ghana; p; Mahunka 1974a
Scutacarus perlatus Mahunka, 1974; Ghana; p; Mahunka 1974a
Scutacarus soosi Mahunka, 1974; Nigeria; p; Mahunka 1974a
Scutacarus undulomarginatus Mahunka, 1979; Tanzania; p; Mahunka 1979
Thaumatopelvis gracilis Mahunka, 1974; Ghana; p; Mahunka 1974a
II. SCUTACARID SPECIES ASSOCIATED WITH CARABIDAE (HEXAPODA, ECTOGNATHA, COLEOPTERA)
Brachyninae
Brachinus stenoderus
Archidispus riparius Kurosa, 1989; Japan; p; Kurosa 1989
Broscinae
Broscus cephalotes
Archidispus carabidophilus Sevastianov, 1974; Crimea, Russia, Ukraine; p; Khaustov 2008
Craspedonotus tibialis
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Carabinae
Carabus opaculus
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Elaphrinae
Elaphrus spp.
Archidispus bembidii (Karafiat, 1959); Belgium, Germany, Japan; p; Karafiat 1959, Kurosa 1980, Fain et al. 1995
Harpalinae
Acupalpus spp.
Archidispus acupalpi Kurosa, 2009; Japan; p; Kurosa 2009
Archidispus bembidii (Karafiat, 1959); Germany, Iran, Japan; p; Karafiat 1959, Kurosa 1980, Katlav et al. 2015
Archidispus conspicuus Kurosa, 1978; Japan; p; Kurosa 1978, 1980
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus foliatus (Kurosa, 1978); Japan; p, Kurosa 1978, 1980
Archidispus insolitus (Kurosa, 1974); Japan; p; Kurosa 1974, 1977, 1980
Archidispus longicaudatus (Kurosa, 1978); Japan; p; Kurosa 1978, 1980
Archidispus masatakai Kurosa, 2003; Japan; p; Kurosa 2003
Archidispus minor (Karafiat, 1959); Germany; p; Karafiat 1959
Archidispus ohtanii Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus rostratus Kurosa, 1976; Japan; p, Kurosa 1976, 1977, 1980
Archidispus triangularis (Kurosa, 1974); Japan; p; Kurosa 1974, 1977, 1980
Archidispus yanoi Kurosa, 1984; Japan; p, Kurosa 1984
Anisodactylus spp.
Archidispus amarae (Kurosa, 1970); Austria, Japan, Moldavia, Ukraine; p; Kurosa 1970, 1972b, 1977, 1980, Khaustov 2008
Archidispus bembidii (Karafiat, 1959); Germany, Japan; p; Karafiat 1959, Kurosa 1980
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus minor (Karafiat, 1959); Germany; p; Karafiat 1959
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Harpalus spp.
Archidispus amarae (Kurosa, 1970); Austria, Japan, Moldavia, Ukraine; p; Khaustov 2008
Archidispus carabidophilus Sevastianov, 1974; Russia, Ukraine; p; Khaustov 2008
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus curtonoti (Kurosa, 1972); Japan; p; Kurosa 1980, 1977
Archidispus longicaudatus (Kurosa, 1978); Japan; p; Kurosa 1980
Archidispus minor (Karafiat, 1959); Germany, Hungary; p; Karafiat 1959, Mahunka 1972b
Archidispus ophoni (Kurosa, 1972); Japan; p; Kurosa 1980, 1977
Archidispus sellnicki (Mahunka, 1964); Hungary, Moldavia, Russia, Ukraine; p; Khaustov 2008
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Archidispus tauricus Khaustov, 2002; Crimea; p; Khaustov 2002, 2008
Loxoncus spp. (syn. Anoplogenius)
Archidispus acupalpi Kurosa, 2009; Japan; p; Kurosa 2009
Archidispus anoplogenii (Kurosa, 1974); Japan; p; Kurosa 1974, 1977, 1980
Archidispus bembidii (Karafiat, 1959); Japan; p; Kurosa 1980
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus conspicuus Kurosa, 1978; Japan; p; Kurosa 1978, 1980
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus foliatus (Kurosa, 1978); Japan; p; Kurosa 1978, 1980
Archidispus fusiformis Kurosa, 1976; Japan; p, Kurosa 1976, 1977
Archidispus insolitus (Kurosa, 1974); Japan; p; Kurosa 1970, 1974, 1980
Archidispus longicaudatus (Kurosa, 1978); Japan; p; Kurosa 1978, 1980
Archidispus masatakai Kurosa, 2003; Japan; p; Kurosa 2003
Archidispus ohtanii Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus okumurai Kurosa, 1983; Japan; p, Kurosa 1983
Archidispus omega Kurosa, 1976; Japan; p; Kurosa 1976, 1977, 1980
Archidispus papillosus Kurosa, 1978; Japan; p; Kurosa 1978
Archidispus proximus Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus pseudomega Kurosa, 1984; Japan; p; Kurosa 1984
Archidispus rostratus Kurosa, 1976; Japan; p; Kurosa 1976, 1977, 1980
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Archidispus triangularis (Kurosa, 1974); Japan; p, Kurosa 1974, 1977, 1980
Ophonus spp.
Archidispus ophoni (Kurosa, 1972); Japan; p; Kurosa 1972b
Oxycentrus argutoroides
Archidispus similis Kurosa, 1989; Japan; p, Kurosa 1989
Stenolophus spp.
Archidispus acupalpi Kurosa, 2009; Japan; p; Kurosa 2009
Archidispus americanus Khaustov & Husband, 2004; USA (Arkansas); p, Khaustov & Husband 2004
Archidispus anoplogenii (Kurosa, 1974); Japan; p; Kurosa 1974
Archidispus arakawanus Kurosa, 1990; Japan; p; Kurosa 1990
Archidispus bembidii (Karafiat, 1959); Germany, Hungary; p; Karafiat 1959, Mahunka 1972b, Kurosa 1980
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus conspicuus Kurosa, 1978; Japan; p; Kurosa 1978, 1980
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus difficilis Kurosa, 1983; Japan; p, Kurosa 1983
Archidispus dubinini Khaustov, 2003; Ukraine; p; Khaustov 2003, 2008
Archidispus foliatus (Kurosa, 1978); Japan; p; Kurosa 1978, 1980
Archidispus fukudai Kurosa, 1984; Japan; p; Kurosa 1984
Archidispus fusiformis Kurosa, 1976; Japan; p, Kurosa 1976, 1977, 1980
Archidispus insolitus (Kurosa, 1974); Japan; p; Kurosa 1974, 1977, 1980
Archidispus iriomotensis Kurosa, 1990; Japan; p, Kurosa 1990
Archidispus kazuyoshikurosai Khaustov, 2004; Moldavia, Ukraine, p; Khaustov 2008
Archidispus longicaudatus (Kurosa, 1978); Japan; p; Kurosa 1978
Archidispus longisolenidiatus Khaustov, 2008; Ukraine; p, Khaustov 2008
Archidispus masatakai Kurosa, 2003; Japan; p, Kurosa 2003
Archidispus minor (Karafiat, 1959); Germany, Hungary; p; Karafiat 1959
Archidispus missouriensis Khaustov & Husband, 2004; USA (Missouri); p; Khaustov & Husband 2004
Archidispus nickelli (Mahunka, 1969); USA (Kansas); p, Mahunka 1969c
Archidispus ocellatus Khaustov & Husband, 2004; USA (Missouri); p; Khaustov & Husband 2004
Archidispus omega Kurosa, 1976; Japan; p, Kurosa 1976, 1977, 1980
Archidispus papillosus Kurosa, 1978; Japan; p; Kurosa 1978, 1980
Archidispus pseudomega Kurosa, 1984; Japan; p; Kurosa 1984
Archidispus rostratus Kurosa, 1976; Japan; p; Kurosa 1976, 1977, 1980
Archidispus satoui (Kurosa, 1983); Japan; p, Kurosa 1983
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus stenolophi Kurosa, 1990; Japan; p, Kurosa 1990
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Archidispus triangularis (Kurosa, 1974); Japan; p, Kurosa 1974, 1977, 1980
Archidispus ukrainicus Khaustov, 2004; Ukraine; p; Khaustov 2008
Archidispus undulatus Kurosa, 2003; Japan; p; Kurosa 2003
Trichotichnus spp.
Archidispus jezoensis Kurosa, 1983; Japan; p; Kurosa 1983
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Licininae
Chlaenius spp.
Archidispus conciliatus Khaustov, 2008; Crimea; p; Khaustov 2008
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus ohtanii Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus proximus Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Lachnocrepis spp.
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus proximus Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus tonegawanus Kurosa, 2005; Japan; p; Kurosa 2005
Oodes spp.
Archidispus proximus Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Nebriinae
Nebria spp.
Archidispus diploi Kurosa, 1991; Japan, Russia (far east); p; Kurosa 1991, Khaustov 2008
Omophroninae
Omophron aequalis aequalis
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Platyninae
Agonum spp.
Archidispus bembidii (Karafiat, 1959); Belgium, Germany, Hungary, Japan; p, Karafiat 1959, Mahunka 1972b, Kurosa 1980, Fain et al. 1995
Archidispus magnificus (Karafiat, 1959); Austria, France, Germany, Hungary, Russia, Ukraine; p; Karafiat 1959, Mahunka 1972b, Ebermann 1991b, Khaustov 2008, also in soil
Archidispus minor (Karafiat, 1959); Germany, Hungary; p; Karafiat 1959, Mahunka 1972b
Calathus sp.
Archidispus minor (Karafiat, 1959); Germany; p; Karafiat 1959
Colpodes japonicus, Platynus spp.
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Synuchus arcuaticollis
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Pterostichinae
Amara spp.
Archidispus amarae (Kurosa, 1970), Austria, Japan, Moldavia, Ukraine, p; Kurosa 1970, 1972b, 1977, Khaustov 2008
Archidispus carabidophilus Sevastianov, 1974; Russia, Ukraine, p; Khaustov 2008
Archidispus celiae (Kurosa, 1972); Japan; p; Kurosa 1972b, 1977, 1980
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus curtonoti (Kurosa, 1972); Japan; p; Kurosa 1972b, 1977, 1980
Archidispus minor (Karafiat, 1959); Germany, Hungary, Iran; p; Karafiat 1959, Mahunka 1972b, Loghmani et al. 2014
Archidispus ophoni (Kurosa, 1972); Japan, Moldavia; p, Khaustov 2008
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus sellnicki (Mahunka, 1964); Hungary, Moldavia, Russia, Ukraine; p; Khaustov 2008
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Archidispus tauricus Khaustov, 2002; Ukraine; p; Khaustov 2002, 2008
Chlaeminus annamensis
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Lesticus magnus
Archidispus ohtanii Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Poecilus spp.
Archidispus carabidophilus Sevastianov, 1974; Russia, Ukraine, p; Khaustov 2008
Archidispus minor (Karafiat, 1959); Germany; p; Karafiat 1959
Archidispus tauricus Khaustov, 2002; Ukraine; p; Khaustov 2002, 2008
Pterostichus spp.
Archidispus belorussicus Khaustov, 2002; Belarus; p; Khaustov 2002, 2008
Archidispus bembidii (Karafiat, 1959); Iran; p; Katlav et al. 2015
Archidispus carabidophilus Sevastianov, 1974; Russia, Ukraine, p; Khaustov 2008
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus jezoensis Kurosa, 1983; Japan; p, Kurosa 1983
Archidispus minor (Karafiat, 1959), Germany, Hungary; p; Karafiat 1959, Mahunka 1972b
Archidispus ohtanii Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus pseudocurtonoti Khaustov, 2008; Belarus; p; Khaustov 2008
Archidispus pterostichi Rack, 1973; Hungary, Netherlands, Ukraine; p, Rack 1973, Khaustov 2008
Archidispus quietus Khaustov, 2008; Belarus; p; Khaustov 2008
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus subintermissus Kurosa, 1983; Japan; p; Kurosa 1983
Archidispus tonegawanus Kurosa, 2005; Japan; p; Kurosa 2005
Trechinae
Bembidion spp.
Archidispus bembidii (Karafiat, 1959); Asia, Africa, Europe, North America; p; Karafiat 1959, Mahunka 1972b, Kurosa 1980, Ebermann 1991a, Fain et al. 1995, Fain & Miessen 1997
Archidispus chujoi Kurosa, 2005; Japan; p; Kurosa 2005
Archidispus cnemidoti Kurosa, 1991; Japan; p, Kurosa 1991
Archidispus crassiradix Kurosa, 1989; Japan; p; Kurosa 1989
Archidispus magnificus (Karafiat, 1959); Austria; Ebermann et al. 2011
Archidispus nobukoae Kurosa, 1991; Japan; p; Kurosa 1991
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus sugiyamai Kurosa, 1991; Austria, Japan, Ukraine; p, Kurosa 1991, Khaustov 2008, Ebermann et al. 2011
Diplous caligatus
Archidispus diploi Kurosa, 1991; Japan, Russia (far east); p; Kurosa 1991, Khaustov 2008
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Archidispus sugiyamai Kurosa, 1991; Japan, Ukraine; p, Kurosa 1991, Khaustov 2008
Diplous depressus
Archidispus riparius Kurosa, 1989; Japan; p, Kurosa 1989
Elaphropus diabrachys
Archidispus esfarayenicus Hajiqanbar & Khaustov, 2014; Iran; p, Hajiqanbar & Khaustov 2014
Patrobus flavipes
Archidispus similis Kurosa, 1989; Japan; p; Kurosa 1989
Pogonistes rufoaeneus
Archidispus szaboi (Mahunka, 1977); Hungary, Ukraine, p; Khaustov 2008
“Carabidae" (not further classified in literature)
Archidispus amarae (Kurosa, 1970); Austria; p; Ebermann 1991a, also in soil
Archidispus armatus (Karafiat, 1959); Hungary; p; Mahunka 1981; also in soil
Archidispus bembidii (Karafiat, 1959); Germany; p, Karafiat 1959, Khaustov 2008; up to 100 specimens/beetle; near water
Archidispus conspicuus Kurosa, 1978; Iran; p, Katlav et al. 2015
Archidispus minor (Karafiat, 1959); Asia, Europe; p; Karafiat 1959, Mahunka 1981, Ebermann 1991b, Khaustov 2008, also in soil
Archidispus ohtanii Kurosa, 1989; Japan, Moldavia, Ukraine; p, Khaustov 2008
Archidispus truncatus (Delfinado & Baker, 1976); USA (Florida); p; Delfinado & Baker 1976
III. SCUTACARID SPECIES ASSOCIATED WITH COLEOPTERA EXCL. CARABIDAE (HEXAPODA, ECTOGNATHA)
Cupepidae
Cupes concolor
Imparipes (Imparipes) cupes Delfinado & Baker, 1978; USA; p; Delfinado & Baker 1978
Curculionidae
Hylurgops palliatus
Scutacarus scolyti Mahunka & Moser, 1980; Germany; p; Mahunka & Moser 1980
Ips typographus
Scutacarus cf. culmusophilus Sevastianov, 1975; Austria; p; Knapp 2008
Scutacarus scolyti Mahunka & Moser, 1980; Germany, Russia, Sweden, ip; Mahunka & Moser 1980, Moser et al. 1989, Khaustov 2008
Pityogenes chalcographus
Scutacarus cf. culmusophilus Sevastianov, 1975; Austria; p; Knapp 2008
Scutacarus scolyti Mahunka & Moser, 1980; Austria, Russia; ip; Khaustov 2008, Schäffer & Baumann unpubl.
Rhynchophorus ferrugineus
Scutacarus sp.; Egypt; n.k.; El-Sharabasy 2010
Trypodendron lineatum
Scutacarus scolyti Mahunka & Moser, 1980; Germany; p; Mahunka & Moser 1980
Heteroceridae
Augyles hispidulus
Scutacarus sphaeroideus Karafiat, 1959; Russia; p, Khaustov & Sazhnev 2016
Heterocerus fenestratus
Scutacarus longitarsus (Berlese, 1905); Germany, Italy; p, Karafiat 1959, also in moss
Scutacarus sphaeroideus Karafiat, 1959; Russia; p, Khaustov & Sazhnev 2016
Heterocerus flexuosus
Scutacarus sphaeroideus Karafiat, 1959; Russia; p, Khaustov & Sazhnev 2016
Heterocerus marginatus
Scutacarus longitarsus (Berlese, 1905); Germany, Italy; p, Karafiat 1959, also in moss
Heterocerus sp.
Archidispus bembidii (Karafiat, 1959); Holarctic; p; Khaustov 2008
Scutacarus sphaeroideus Karafiat, 1959; Japan; p; Kurosa 1980
Hydrophilidae
Cercyon lateralis
Imparipes (Sporichneuthes) dispar Rack, 1964; Austria; p; Ebermann et al. 2016
Cercyon ustulatus
Pygmodispus equestris Paoli, 1911; Austria, Germany, Hungary, Italy, Ukraine; p; Karafiat 1959, Khaustov 2008; also in moss, forest litter
Coelostoma sp.
Archidispus bembidii (Karafiat, 1959); Holarctic; p; Khaustov 2008
Pselaphidae
Centrophthalmus sp.
Imparipes (Imparipes) pselaphidorum Ebermann, 1988; Africa; p; Ebermann 1988
Ptiliidae
Scutacarus emadi Mahunka & Zaki, 1992; Egypt; p; Mahunka & Zaki 1992
Scutacarus longitarsus (Berlese, 1905); Egypt; p; Mahunka & Zaki 1992
Scutacarus pugillator (Paoli, 1911); Austria, Egypt; p; Mahunka & Zaki 1992, Truck 2012; also in manure, soil
Scarabaeidae
Aphodius lividus
Pygmodispus bicornutus Ebermann & Rodrigues, 2001; Brazil; p; Ebermann & Rodrigues 2001; also in soil
Scutacarus longitarsus (Berlese, 1905); Brazil; p; Rodrigues et al. 2001
Ataenius spp.
Pygmodispus bicornutus Ebermann & Rodrigues, 2001; Brazil; p; Ebermann & Rodrigues 2001, Rodrigues et al. 2001; also in soil
Scutacarus sp.; Brazil; p; Rodrigues et al. 2001
Bothynus striatellus
Archidispus sacculiger (Mahunka, 1968); chile; p; Mahunka 1968a
Copris sp.
Pygmodispus (Allodispus) brachiosus Paoli, 1911; Java; p; Paoli 1911
Digitonthophagus gazella
Pygmodispus bicornutus Ebermann & Rodrigues, 2001; Brazil; p; Rodrigues et al. 2001; also in soil
Osmoderma eremicola
Heterodispus near elongatus (Trägardh, 1904); USA (New York); p; Norton 1973
Pleurophorus anatolicus, Rhyssemodes orientalis
Scutacarus iranicus Ebermann et al., 2003; Iran; p, Ebermann et al. 2003, Loghmani et al. 2014
Staphylinidae
Archidispus armatus (Karafiat, 1959); Iran; p; Katlav et al. 2015
Lamnacarus ornatus Balogh & Mahunka, 1963; Austria; p; Truck 2012
Atheta malleus
Lamnacarus ornatus Balogh & Mahunka, 1963; Austria, Hungary, Lithuania, Mongolia, Russia; p; Ebermann 1991a, Khaustov 2008, also in dung
Bledius sp.
Archidispus irregularis Katlav & Hajiqanbar, 2016; Iran; p; Katlav et al. 2016
Boopinus fuliginosus
Scutacarus sphaeroideus Karafiat, 1959; Germany; p; Karafiat 1959; also in moss
Lithocharis nigriceps
Imparipes (Sporichneuthes) dispar Rack, 1964; Austria; p; Ebermann et al. 2016
Philonthus fulvipes, P. micans
Archidispus armatus (Karafiat, 1959); Germany, Hungary; p; Karafiat 1959, Mahunka 1972b
Stenus spp.
Scutacarus longipes Rack, 1975; Austria, USA; p; Ebermann 1978; also on Fragaria plants
Scutacarus sphaeroideus Karafiat, 1959; cosmopolitic; p; Karafiat 1959, Khaustov 2008; on shores of rivers and lakes; also in moss
Trogophloeus corticinus
Scutacarus longitarsus (Berlese, 1905); Austria; p; Ebermann 1991a; also in manure
Tychus mutinensis
Imparipes (Imparipes) histricinus Berlese, 1903; Italy; p; Paoli 1911; also in soil, moss
Tenebrionidae
Anomalipus elephas
Heterodispus foveatus Jagersbacher-Baumann & Ebermann 2012; Botswana; p; Jagersbacher-Baumann & Ebermann 2012a
Blaps halophila
Heterodispus temperatus Khaustov, 2008; Ukraine; p; Khaustov 2008
“Coleoptera" (not further classified in literature)
Archidispus cornutus (Mahunka, 1973); Ghana; p; Mahunka 1973a
Diversipes horridolatus Mahunka, 1975; Australia; p; Mahunka 1975c
Lamnacarus expansus Mahunka, 1973; Ghana; p; Mahunka 1973b
Scutacarus elacatus Mahunka, 1973; Ghana; p; Mahunka 1973b
Scutacarus rhabdiformis Mahunka, 1973; Ghana; p; Mahunka 1973b
Symbolocrasis acutimera Mahunka, 1973; Ghana; p; Mahunka 1973b
Symbolocrasis hypostigma Mahunka, 1973; Ghana; p; Mahunka 1973b
Symbolocrasis synmixta Mahunka, 1973; Ghana; p; Mahunka 1973b
Thaumatopelvis gracilis Mahunka, 1974; Ghana; p; Ebermann 1980b
Thaumatopelvis sellata Mahunka, 1973; Ghana; p; Mahunka 1973b, Ebermann 1980b
IV. SCUTACARID SPECIES ASSOCIATED WITH APOIDEA AND VESPOIDEA EXCL. FORMICIDAE (HEXAPODA, ECTOGNATHA, ACULEATA)
Apoidea: Apidae
Apis mellifera
Imparipes (Imparipes) apicola (Banks, 1914); Canada; n.k.; Banks 1914
Scutacarus acarorum (Goeze, 1780); n.k.; i; Schousboe 1986
Bombus spp.
Imparipes (Imparipes) degenerans Berlese, 1904; Florida, Germany, Italy, Russia; p; Karafiat 1959; also in moss; only 1 finding on B. terrestris!
Parascutacarus indicus Baker & Delfinado, 1975; India; p; Baker & Delfinado 1975
Scutacarus acarorum (Goeze, 1780); Austria, Belgium, Czechoslovakia, Denmark, Germany, Great Britain, Greece, Hungary, Japan, Korea, Mongolia, New York, North America, Poland, Russia, Sweden, Switzerland, Turkey; ip; Michael 1884, Paoli 1911, Karafiat 1959, Mahunka 1965a, 1967a, 1967b, 1972b, Cross & Bohart 1969, Chmielewski 1971, Delfinado et al. 1976, Kurosa 1980, Schousboe 1986, Ebermann 1992, Fain et al. 1992, Fain & Baugnée 1996, Schwarz & Huck 1997, Çankaya & Kaftanoglu 2006, Larsson 2007, Chmielewski & Baker 2008, Khaustov 2008, Jagersbacher-Baumann 2014a, Kontschán 2015, Kontschán et al. 2016; also in moss, soil
Scutacarus acarorum (Goeze, 1780) (?); Argentina; p; Maggi et al. 2011, Revainera et al. 2014; could be S. mendax (pers. comm. Revainera)
Scutacarus cf. mendax Karafiat, 1959; Brazil, p; Guerra et al. 2012; det. Baumann (unpubl.)
Scutacarus deserticolus Mahunka, 1969; Austria; ip; Ebermann 1991a, Baumann unpubl.
Scutacarus mendax Karafiat, 1959; Austria; i; Jagersbacher-Baumann 2014b
Scutacarus occultatus Sevastianov, 1975; Austria, Hungary, Ukraine; ip; Khaustov 2008, Jagersbacher-Baumann 2014b
Scutacarus pygmephoroides Mahunka, 1967; Hungary, Mongolia; i; Mahunka 1967a, 1972b
Scutacarus spinosus Storkan, 1936; Mongolia; i; Mahunka 1967a, 1968a
Dasypoda plumipes
Imparipes (Imparipes) rafalskii Dastych, 1978; Poland; p; Dastych 1978
“Bees" (not further classified in literature)
Diversipes zwoelferi Karafiat, 1959; Germany, Poland; i; Karafiat 1959, also in meadow soil
Scutacarus acarorum (Goeze, 1780); Germany; p; Karafiat 1959; in detritus of nest of “wild bees", attached to wings
Scutacarus unicus Delfinado & Baker, 1976; USA (New York); n.k.; Delfinado & Baker 1976; also in leaf litter
Apoidea: Halictidae
Augochlorella spp.
Imparipes spp.; USA (Kansas); i; Ordway 1964
Halictus spp.
Imparipes spp; USA; n.k.; Banks 1904
Imparipes (Apidacarus) paulyi Ebermann & Fain, 2002; Algeria, Namibia, Uganda, Iran; p; Ebermann & Fain 2002, Loghmani et al. 2014
Imparipes (Imparipes) apicola (Banks, 1914); Austria, Belgium, Germany, Mexico, Poland, USA; n.k.; Cross & Bohart 1969, 1992, Delfinado & Baker 1976, Kuhlmann 1998, Ebermann & Hall 2005
Imparipes (Imparipes) apidophilus Mahunka, 1974, Hungary; p; Mahunka 1974c
Imparipes (Imparipes) americanus (Banks, 1904); Canada (British Columbia); n.k.; Banks 1906
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
Imparipes (Imparipes) floridensis Delfinado & Baker, 1976; USA (Florida); i; Delfinado & Baker 1976
Scutacarus acarorum (Goeze, 1780); Belgium; p; Fain & Baugnée 1996
Lasioglossum spp.
Imparipes (Imparipes) apicola (Banks, 1914); Austria, Belgium, Germany, Mexico, Poland, USA; ip; Cross & Bohart 1969, 1992, Delfinado & Baker 1976, Eickwort 1979, Kuhlmann 1998, Ebermann & Hall 2005
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Iran, Poland, Slovakia; p; Ebermann et al. 2013, Bakiani et al. 2016
Imparipes (Imparipes) eickworti Mahunka, 1969; Costa Rica; ip; Mahunka 1969b, Eickwort & Eickwort 1971
Imparipes (Imparipes) ithacensis Delfinado & Baker, 1976; USA (New York); n.k.; Delfinado & Baker 1976
Imparipes (Imparipes) mexicanus Delfinado & Baker, 1976; Mexico; p; Delfinado & Baker 1976
Imparipes (Imparipes) neotropicus Delfinado & Baker, 1976; Chile; p; Delfinado & Baker 1976
Imparipes (Imparipes) vulgaris Delfinado & Baker, 1976; USA (California); ip; Delfinado & Baker 1976
Scutacarus eickworti Delfinado & Baker, 1976; USA (New York); p; Delfinado & Baker 1976
Nomia spp.
Imparipes (Imparipes) apicola (Banks, 1914); USA (Idaho, Nevada, Utah, Wyoming); ip; Cross & Bohart 1969, 1992; in Cross & Bohart 1969 handled as Imparipes americanus
Imparipes (Imparipes) texanus (Cockerell, 1910); USA; n.k.; Cockerell 1910
Nasutiscutacarus ampliatus Beer & Cross, 1960; Philippines, p; Beer and Cross 1960
Nasutiscutacarus anthrenae Beer & Cross, 1960; Indonesia; p; Beer and Cross 1960
Nomiapis sp.
Imparipes (Apidacarus) paulyi Ebermann & Fain, 2002; Iran; p; Loghmani et al. 2014
Sphecodes spp.
Imparipes (Imparipes) apicola (Banks, 1914); Austria, Belgium, Germany, Mexico, Poland, USA; n.k.; Cross & Bohart 1969, 1992, Delfinado & Baker 1976, Kuhlmann 1998, Ebermann & Hall 2005
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
“Halictidae" (not further classified in literature)
Imparipes (Imparipes) apicola (Banks, 1914); Belgium; p; Fain et al. 1992; given as Imparipes hystricinus dispar; later determined as Imparipes apicola by Ebermann (pers. comm.)
Apoidea: Sphecidae
Cerceris spp., Crossocerus spp., Lestica spp.
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
Diodontus spp., Gorytes spp.
Imparipes (Imparipes) apicola (Banks, 1914); Austria, Belgium, Germany, Mexico, Poland, USA; n.k.; Cross & Bohart 1969, 1992, Delfinado & Baker 1976, Kuhlmann 1998, Ebermann & Hall 2005
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Ectemnius spp.
Imparipes (Imparipes) apicola (Banks, 1914); Austria, Belgium, Germany, Mexico, Poland, USA; n.k.; Cross & Bohart 1969, 1992, Delfinado & Baker 1976, Kuhlmann 1998, Ebermann & Hall 2005
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
Larra spp., Sphex spp.
Archidispus sphecis Mahunka, 1977; Vietnam; p; Lang & Mahunka 1977
Pemphredon spp., Stigmus solskyi
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
“Sphecidae" (not further classified in literature
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Ukraine; n.k.; Khaustov 2008
Apoidea: Andrenidae
Andrena spp.
Imparipes (Imparipes) apicola (Banks, 1914); Austria, Belgium, Germany, Mexico, Poland, USA; n.k.; Cross & Bohart 1969, 1992, Delfinado & Baker 1976, Ebermann & Hall 2005
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
“Andrenidae" (not further classified in literature)
Imparipes (Imparipes) apicola (Banks, 1914); USA; ip; Eickwort 1979
Imparipes (Imparipes) cf. parapicola Delfinado, Baker & Abbatiello, 1976; Iran; p; Kamali et al. 2001
Apoidea: Colletidae
Hylaeus spp.
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
Apoidea: Megachilidae
Megachile lapponica
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
Osmia papaveris
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Apoidea: Crabronidae
Bembix tarsata, Mimesa equestris, Psenulus concolor, Trypoxylon minus
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
Lindenius spp.
Imparipes (Imparipes) breganti Ebermann & Hall, 2004; Austria, Belgium, Germany; p; Ebermann & Hall 2004
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
Mimumesa dahlbomi
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
Vespoidea: Vespidae
Symmorphus bifasciatus
Imparipes (Imparipes) haeseleri Ebermann & Hall, 2003; Austria, Belgium, Germany, India, Italy, Poland, Ukraine; p; Ebermann & Hall 2003, Hall & Ebermann 2005
Vespa sp.
Scutacarus acarorum (Goeze, 1780); Germany; p; Vitzthum 1927; extremely rare
Vespoidea: Mutillidae
Myrmilla calva
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
Tricholabiodes sp.
Scutacarus subquadratus Khaustov & Chydyrov, 2004; Iran; p; Loghmani et al. 2014
Vespoidea: Pompilidae
Anoplius infuscatus, Caliadurgus fasciatellus, Evagetes proximus
Imparipes (Imparipes) burgeri Ebermann & Jagersbacher-Baumann, 2013; Austria, Belgium, Germany, Poland, Slovakia; p; Ebermann et al. 2013
V. SCUTACARID SPECIES ASSOCIATED WITH FORMICIDAE EXCL. FORMICINAE AND MYRMICINAE (HEXAPODA, ECTOGNATHA, ACULEATA, VESPOIDEA)
Dolichoderinae
Tapinoma erraticum
Imparipes (Imparipes) comatus Mahunka, 1970; France, Hungary, Ukraine; i; Mahunka 1977c, Khaustov 2008 Imparipes (Imparipes) pennatus Karafiat, 1959; France; n.k.; Mahunka 1977c
Tapinoma simrothi
Scutacarus sabinaesimilis Khaustov & Chydyrov, 2004; Turkmenistan; i, Khaustov 2008
Tapinoma sp.
Lophodispus tapinoma Sobhi & Hajiqanbar 2017; Iran; p; Sobhi et al. 2017
Liometopum apiculatum
Imparipes (Imparipes) liometopi Mahunka, 1982; Mexico; p; Mahunka 1982; nom. nov. (Mahunka & Rack 1984), I. ( I.) mexicanus
Scutacarus athiashenriotae Mahunka, 1982; Mexico; p; Mahunka 1982
Dorylinae
Cheliomyrmex morosus
Imparipes (Imparipes) lapillatus Mahunka, 1977; Panama; p; Berghoff & Franks 2007; also in soil and litter
Eciton spp.
Imparipes (Imparipes) crudelatus Mahunka, 1977; Ecuador, Panama; ip; Mahunka 1977b, Rettenmeyer et al. 2011
Imparipes (Imparipes) cf. crudelatus Mahunka, 1977; Panama; p; Berghoff et al. 2009
Imparipes (Imparipes) egisetus Mahunka, 1977; Costa Rica; i; Mahunka 1977b
Imparipes (Imparipes) lapillatus Mahunka, 1977; Ecuador, Panama; ip; Mahunka 1977b, Berghoff et al. 2009
Imparipes (Imparipes) marianae Mahunka, 1977; Costa Rica, Ecuador; p; Mahunka 1977a, Rettenmeyer et al. 2011
Imparipes (Imparipes) officius Mahunka, 1977; Costa Rica, i; Mahunka 1977b
Imparipes (Imparipes) recisus Mahunka, 1977; Panama; i; Mahunka 1977b
Imparipes (Imparipes) rectangulatus Mahunka, 1977; Costa Rica, Ecuador, Panama, USA (Kansas); p; Mahunka 1977a, Berghoff et al. 2009, Rettenmeyer et al. 2011
Imparipes (Imparipes) suboletus Mahunka, 1977; Panama; ip; Mahunka 1977b, Berghoff et al. 2009, Rettenmeyer et al. 2011
Imparipes sp.; Panama; p; Berghoff et al. 2009
Imparipes (Telodispus) opusculus Mahunka, 1977; Brazil, Costa Rica, Mexico, ip; Mahunka 1977b, Rettenmeyer et al. 2011
Imparipes (Telodispus) rationis Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Pygmodispus (Pygmodispus) dorylini Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Rettenmeyerella petropolitana solenifera Mahunka, 1977; Costa Rica, Ecuador, i; Mahunka 1977a, also from refuse deposit with Phoridae, Staphylinidae
Scutacaropsis problematicus Mahunka, 1977; Costa Rica, Ecuador, p; Mahunka 1977a, Rettenmeyer et al. 2011
Scutacaropsis scutacaroides Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Scutacarus andrassyi Mahunka, 1968; Panama; p; Berghoff et al. 2009
Scutacarus omittatus Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Scutacarus pseudocomus Mahunka, 1977; Panama; ip; Mahunka 1977b, Rettenmeyer et al. 2011
Scutacarus setarus Mahunka, 1977; Costa Rica; i; Mahunka 1977b
“Scutacaridae"; Central America; i; Rettenmeyer 1961b
Labidus spp.
Archidispus calcarifer (Mahunka, 1977); Costa Rica, Panama, i; Mahunka 1977b
Archidispus repus Mahunka, 1977; Costa Rica, Panama; i; Mahunka 1977b
Imparipes (Imparipes) compensatus Mahunka, 1977; Costa Rica, Ecuador; i; Mahunka 1977b
Imparipes (Imparipes) convexus Mahunka, 1977; Costa Rica, Ecuador; n.k.; Mahunka 1977a
Imparipes (Imparipes) rettenmeyeri Mahunka, 1977; Costa Rica, Ecuador; n.k.; Mahunka 1977a
Imparipes (Telodispus) nabilatus Mahunka, 1977; Costa Rica; i, Mahunka 1977b
Imparipes (Telodispus) opusculus Mahunka, 1977; Brazil, Costa Rica, Mexico, i; Mahunka 1977b
Scutacaropsis problematicus Mahunka, 1977; Costa Rica, Ecuador; n.k.; Mahunka 1977a
Thaumatopelvis minutissimus Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a, Ebermann 1980b
Thaumatopelvis rugosus Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a, Ebermann 1980b
Neivamyrmex spp.
Archidispus statitus (Mahunka, 1977); Brazil; i; Mahunka 1977b
Archidispus willmanni Mahunka, 1977; Costa Rica; n.k.; Mahunka 1977a
Imparipes (Imparipes) cassovaricus Mahunka, 1970; Costa Rica; n.k.; Mahunka 1977a
Imparipes (Imparipes) compensatus Mahunka, 1977; Costa Rica, Ecuador; i; Mahunka 1977b
Imparipes (Imparipes) dimidiatus Mahunka, 1977; Costa Rica; n.k.; Mahunka 1977a
Imparipes (Imparipes) egisetus Mahunka, 1977; Costa Rica; i; Mahunka 1977b
Imparipes (Imparipes) rectangulatus Mahunka, 1977; Costa Rica, Ecuador, USA (Kansas); n.k.; Mahunka 1977a
Imparipes (Imparipes) saevus Mahunka, 1977; Costa Rica; i; Mahunka 1977b
Imparipes (Telodispus) elzingai Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Imparipes (Telodispus) moralesi Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Imparipes (Telodispus) moricus Mahunka, 1977; USA; i; Mahunka 1977b
Imparipes (Telodispus) retrosus Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
Thaumatopelvis minutissimus Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a, Ebermann 1980b
Nomamyrmex esenbeckii
Archidispus repus Mahunka, 1977; Costa Rica, Panama; i; Mahunka 1977b
Scutacarus chadabi Mahunka, 1977; Ecuador; n.k.; Mahunka 1977a
“Dorylinae" (not further classified in literature)
Scutacaridae; Brazil, Panama; p; Rettenmeyer 1961a; “20 different species"
Ponerinae
Ponera coarctata
Imparipes (Imparipes) malus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
“Ants" (not further classified in literature)
Archidispus haarloevi (Karafiat, 1959); Denmark, Hungary; i; Karafiat 1959, Mahunka 1981; also in meadow soil, oak litter
Genus Imparipes; USA; n.k.; Banks 1904
Imparipes (Imparipes) degenerans Berlese, 1904; Germany, Italy, Russia, USA (Florida); ip; Karafiat 1959; also in moss
Imparipes (Imparipes) hortobagyensis Mahunka, 1981; Hungary; i; Mahunka 1981
Imparipes (Imparipes) histricinus Berlese, 1903; Germany, Hungary, Italy, Russia, Sudan; ip; Karafiat 1959, Mahunka 1981; also in forest litter
Imparipes (Imparipes) imaginatus Mahunka, 1981; Hungary; i; Mahunka 1981
Imparipes (Imparipes) intermissus Karafiat, 1959; Hungary; i; Mahunka 1965b
Imparipes (Imparipes) longisetosus Willmann, 1951; Germany, Hungary; i; Mahunka 1981; also in saline turf
Imparipes (Imparipes) myrmecophilus Mahunka, 1972; Hungary; i; Mahunka 1981; also in forest litter, rotten debris of oak wood
Imparipes (Imparipes) obsoletus Rack, 1966; Europe; i; Mahunka 1981; also in soil
Imparipes (Imparipes) pennatus Karafiat, 1959; Germany, Hungary; i; Mahunka 1970b
Imparipes (Imparipes) robustus Karafiat, 1959; Bulgaria, Germany; ip; Karafiat 1959, Dobrev 1992; also in moss
Imparipes (Imparipes) steinmanni Mahunka, 1971; Korea; i; Mahunka 1971
Imparipes (Imparipes) tenuis Mahunka, 1981; Hungary; i; Mahunka 1981
Imparipes (Imparipes) vasarhelyii Mahunka & Mahunka-Papp, 1980; Romania; i; Mahunka & Mahunka-Papp 1980
Pygmodispus calcaratus Paoli, 1911; Hungary; i; Mahunka 1986; also in horse manure
Scutacarus bugacensis Mahunka, 1986; Hungary; i; Mahunka 1986
Scutacarus bursula (Berlese, 1903); Germany, Italy; n.k.; Karafiat 1959
Scutacarus calcaratus Storkan, 1936; Bulgaria; n.k.; Štorkán 1936
Scutacarus carsticus Mahunka & Mahunka-Papp, 1980; Hungary; i; Mahunka & Mahunka-Papp 1980
Scutacarus concinnus Mahunka, 1964; Hungary; i; Mahunka 1970b
Scutacarus echidna (Berlese, 1905); Palaearctic; i; Mahunka 1981; also in turf, soil
Scutacarus ellipticus Karafiat, 1959; Europe; i; Mahunka 1981, 1986; also in soil, moss
Scutacarus exspectatus Karafiat, 1959; Hungary; i; Mahunka 1981
Scutacarus gratus hortobagyensis Mahunka, 1981; Hungary; i; Mahunka 1981, 1986; also in litter
Scutacarus hauseri Mahunka, 1977; Hungary, Switzerland; ip; Mahunka 1981, 1986, Dobrev 1992
Scutacarus kassaii Mahunka, 1965; Hungary; i; Mahunka 1986
Scutacarus laetificus Rack, 1966; Bulgaria; i; Dobrev 1992
Scutacarus latifrons Mahunka, 1964; Hungary, Italy; i; Mahunka 1970b
Scutacarus longisetus (Berlese, 1904); Germany, Hungary; i; Karafiat 1959, Mahunka 1981, 1986; also in oak litter, earth from under carcasses, grassy samples
Scutacarus longisetus bucephalos Balogh & Mahunka, 1963; Hungary; i; Mahunka 1986
Scutacarus molnari Mahunka, 1981; Hungary; i; Mahunka 1981; in saline habitat
Scutacarus ovoideus Karafiat, 1959; Germany, Hungary; i; Karafiat 1959, Mahunka 1981; also in moss
Scutacarus peractus Karafiat, 1959; Hungary; i; Mahunka 1981; also in oak litter, rotten debris
Scutacarus pleurotricha Mahunka, 1970; Hungary; i; Mahunka 1970d, 1981
Scutacarus pygmephoroides Mahunka, 1967; Hungary; i; Mahunka 1986; also in litter of Populus
Scutacarus quadrangularis (Paoli, 1911); Hungary; i; Balogh & Mahunka 1962; also in litter, meadow soil, moss; “Variatipes"- group: no claw on leg I
Scutacarus rakonczayi Mahunka, 1981; Bulgaria, Hungary; i; Mahunka 1981, Dobrev 1992
Scutacarus rotundatus [sic]; Bulgaria; n.k.; Štorkán 1936; should be S. rotundus (Berlese, 1903)
Scutacarus silvestri (Berlese, 1903); Hungary; i; Mahunka 1986; also in soil, moss; in text “S. silvestris"
Scutacarus sphaeroideus Karafiat, 1959; Hungary; i; Mahunka 1981
Scutacarus spinitarsus Mahunka, 1986; Hungary; i; Mahunka 1986; also in litter, manure
Scutacarus spinosus Storkan, 1936; Hungary; i; Mahunka 1963, 1981; also in meadow soil, mossy turf
Scutacarus stammeri Karafiat, 1959; Hungary; i; Mahunka 1981, 1986
Scutacarus striatomarginatus Mahunka, 1986; Hungary; i; Mahunka 1986
Scutacarus subcomosus Mahunka, 1970; Hungary; i; Mahunka 1970d
Scutacarus subellipticus Delfinado & Baker, 1976; USA (New York); i; Delfinado & Baker 1976
Scutacarus subterraneus spinosus Storkan, 1936; Bulgaria; n.k.; Štorkán 1936
Scutacarus tackei Willmann, 1942; Europe; i; Mahunka 1981; also in rotten straw
Scutacarus tackei suborbiculatus Rack, 1964; Europe; i; Mahunka 1981; also in mossy turf, meadow litter
Scutacarus terrenus Delfinado & Baker, 1976; USA (New York); i; Delfinado & Baker 1976
Scutacarus transfusionis Mahunka & Mahunka-Papp, 1980; Hungary; i; Mahunka & Mahunka-Papp 1980
VI. SCUTACARID SPECIES ASSOCIATED WITH FORMICINAE (HEXAPODA, ECTOGNATHA; ACULEATA, FORMICIDAE)
Camponotus spp.
Imparipes (Imparipes) robustus Karafiat, 1959; Austria, Bulgaria, France, Germany, Hungary, Japan, Mongolia, Russia, Ukraine (Palaearctic?); n.k.; Khaustov 2008; also in soil, litter
Scutacarus circularis (Berlese, 1903); Italy; p; Berlese 1903, Paoli 1911
Scutacarus echidna (Berlese, 1905); Germany; i; Rack 1966
Scutacarus kassaii Mahunka, 1965; Austria, Czechoslovakia, Hungary, Russia; p; Mahunka 1967b,1968a, Ebermann 1978
Scutacarus novellus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Scutacarus rarissimus Sevastianov, 1983; Ukraine; p; Sevastianov 1983
Scutacarus rotundulus Khaustov & Chydyrov, 2004; Turkmenistan; i; Khaustov & Chydyrov 2004, Khaustov 2008
Scutacarus tyrrhenicus Ebermann, 1986; Italy, Ukraine; n.k.; Khaustov 2008
Cataglyphis aenescens
Scutacarus berdyevi Chydyrov 2007; Turkmenistan; p; Chydyrov 2007
Cataglyphis bicolor
Scutacarus agypticus Yousef & Metawally, 1973; Egypt; p; Yousef & Metawally 1973, Elbadry et al. 1976
Scutacarus unicosimilis Metwali, 1984; Egypt; i; Metwali 1984
Scutacarus wisniewskii Metwali, 1984; Egypt; i; Metwali 1984
Cataglyphis emery
Imparipes (Imparipes) kataglyphi Khaustov & Chydyrov, 2004; Turkmenistan, i; Khaustov & Chydyrov 2004, Khaustov 2008
Cataglyphis cf. nodus
Imparipes (Imparipes) histricinus Berlese, 1903; Iran; p; Loghmani et al. 2014
Scutacarus subquadratus Khaustov & Chydyrov, 2004; Iran; p; Loghmani et al. 2014
Lasius spp.
Archidispus intermissus (Karafiat, 1959); Austria, Belarus, Germany, Hungary, Japan, Kazakhstan, Poland, Ukraine; p; Karafiat 1959, Mahunka 1972b, Khaustov 2008
Imparipes (Imparipes) bisetus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) brevibasis (Sevastianov, 1983); Russia, Ukraine; i; Sevastianov 1983, Khaustov 2016a
Imparipes (Imparipes) brevitarsus Ebermann, 1981; Austria, Russia; ip; Ebermann 1981, Friedl 2000, Ebermann & Krisper 2014, Khaustov 2015d
Imparipes (Imparipes) cf. histricinus Berlese, 1903; Austria; p; Ebermann 1978
Imparipes (Imparipes) comatus Mahunka, 1970; France, Hungary, Ukraine; i; Khaustov 2008
Imparipes (Imparipes) extremiorientalis Khaustov, 2017; Russia; p; Khaustov 2017
Imparipes (Imparipes) fuliginosophilus Khaustov, 2016; Russia; p; Khaustov 2016a
Imparipes (Imparipes) histricinus Berlese, 1903; Austria, Czech Republic; i; Štorkán 1940, Ebermann 1980a, 1982; also in moss
Imparipes (Imparipes) histricinus var. vimariensis Vitzthum, 1919; Germany; p; Vitzthum 1919
Imparipes (Imparipes) imaginatus Mahunka, 1981; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) lasii Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) mordax Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) obsoletus Rack, 1966; holarctic; ip; (Paoli 1911), Mahunka 1977c, Ebermann 1979, Friedl 2000, Khaustov 2008, Ebermann & Krisper 2014, Khaustov 2016a, 2016c; also in decaying material; Paoli (1911) published a species identified as I. degenerans- however, the drawings and description strongly indicate that the species was I. obsoletus (Ebermann, pers. comm.)
Imparipes (Imparipes) robustus Karafiat, 1959; Austria, Bulgaria, France, Germany, Hungary, Japan, Mongolia, Russia, Ukraine (Palaearctic?); n.k.; Mahunka 1972b, Khaustov 2008
Imparipes (Imparipes) sevastianovi Khaustov, 2008; Russia, Ukraine; n.k.; Khaustov 2008, 2016a
Imparipes (Imparipes) sklyari Khaustov, 2008; Russia, Ukraine; ip; Khaustov 2008, 2016c
Imparipes (Imparipes) tomentosus Khaustov, 2016; Russia; p; Khaustov 2016b
Imparipes (Imparipes) sp. A (sensu Friedl 2000); Austria; p; Friedl, Ebermann & Krisper 2014
Imparipes (Imparipes) sp.B (sensu Friedl 2000); Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Sporichneuthes) dispar Rack, 1964; Austria; p; Messner 2001
Lophodispus bulgaricus Dobrev, 1992; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Lophodispus irregularis (Mahunka, 1971); Austria, Hungary, Japan, Korea, Ukraine, USA; ip; Kurosa 1972a, Ebermann 1978, 1979, 1980a, 1980b, 1982, Friedl 2000, Khaustov 2008, Ebermann & Krisper 2014; also in soil, rotten wood
Scutacarus acarorum (Goeze, 1780); Belgium; p; Fain & Baugnée 1996
Scutacarus aequalis Khaustov, 2016; Russia; p; Khaustov 2016b
Scutacarus bucephalus Balogh & Mahunka, 1963; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Scutacarus carsticus Mahunka & Mahunka-Papp, 1980; Hungary, Ukraine, ip; Khaustov 2008
Scutacarus crinitus Khaustov, 2015; Russia; p; Khaustov 2015c
Scutacarus ellipticus Karafiat, 1959; Austria, Germany, Hungary, Russia, Switzerland, Ukraine, former Yugoslavia; p; Mahunka 1977c, Friedl 2000, Khaustov 2008, Ebermann & Krisper 2014; also in soil
Scutacarus exspectatus Karafiat, 1959; Austria, Germany, Hungary, Japan, Ukraine; ip; Karafiat 1959, Friedl 2000, Khaustov 2008, Ebermann & Krisper 2014
Scutacarus flexisetosimilis Khaustov, 2016; Russia; p; Khaustov 2016b
Scutacarus flexisetus Karafiat, 1959; Austria, Germany, Hungary, Japan, Russia, Ukraine; ip; Karafiat 1959, Sevastianov 1965, Ebermann 1980a, Mahunka 1972b, Khaustov 2008, 2016a, Ebermann & Krisper 2014
Scutacarus gratus Karafiat, 1959; Germany, Hungary, Ukraine; n.k.; Karafiat 1959, Sevastianov 1965, Mahunka 1972b
Scutacarus gratus hortobagyensis Mahunka, 1981; Austria; p; Ebermann & Krisper 2014
Scutacarus hauseri Mahunka, 1977; Austria, Switzerland; p; Ebermann 1979, Friedl 2000, Ebermann & Krisper 2014
Scutacarus heterotrichus Khaustov, 2015, Russia; ip; Khaustov 2015d
Scutacarus hystrichocentrus Sevastianov, 1993; Ukraine; n.k.; Sevastianov 1983, Khaustov 2008
Scutacarus insolitus Khaustov, 2015; Russia; i; Khaustov 2015d
Scutacarus kassaii Mahunka, 1965; Russia; i; Khaustov 2015a
Scutacarus lasiophilus Khaustov, 2015; Russia; p; Khaustov 2015c
Scutacarus longisetus (Berlese, 1904); Austria, Bulgaria, Crimea, Hungary, Italy, Poland, Russia, Switzerland, Ukraine, former Yugoslavia; ip; Paoli 1911, Ebermann 1978, 1979, Mahunka 1977c, 1986, Friedl 2000, Khaustov 2008, 2016a, 2016c, Ebermann & Krisper 2014
Scutacarus molnari Mahunka, 1981; Russia; ip; Khaustov 2015d
Scutacarus moseri Khaustov, 2015; Russia; ip; Khaustov 2015d
Scutacarus nudus (Berlese, 1886); Germany, Italy, Netherlands; i; Paoli 1911, Parmentier et al. 2015; “Variatipes"- group: no claw on leg I
Scutacarus nudus bisetus Karafiat, 1959; USA (Maryland); i; Ebermann 1980b; “Variatipes"- group: no claw on leg I
Scutacarus ovoideus Karafiat, 1959; Austria, Germany, Greece, Hungary, Poland, Switzerland, Ukraine; i; Mahunka 1977c, Khaustov 2008
Scutacarus ponticulus Mahunka, 1981; Austria, Hungary, Russia, Ukraine; ip; Friedl 2000, Khaustov 2008, 2016c, Ebermann & Krisper 2014
Scutacarus pseudospinosus Khaustov, 2008; Russia, Ukraine; ip; Khaustov 2008, 2016c, on alates
Scutacarus rotundatus [sic]; Czech Republic; n.k.; Štorkán 1940; should be S. rotundus (Berlese, 1903)
Scutacarus rotundus (Berlese, 1903); Germany, Italy; n.k.; Karafiat 1959
Scutacarus rusticus Sevastianov, 1983; Ukraine; p; Sevastianov 1983
Scutacarus sibiriensis Khaustov, 2015; Russia; ip; Khaustov 2015d
Scutacarus spinosus Storkan, 1936; cosmopolitic; ip; Khaustov 2008, 2016c; common in soil
Scutacarus suavis Khaustov, 2008; Crimea; n.k.; Khaustov 2008
Scutacarus subcomosus Mahunka, 1970; Switzerland; n.k.; Mahunka 1977c
Scutacarus subterraneus (Oudemans, 1913); Carpathians, Germany, Hungary, Lithuania, Netherlands, Poland, Russia, Ukraine; ip; Vitzthum 1919, Štorkán 1940, Sevastianov 1965, Sosnina & Sevastianov 1975, Ebermann 1979, Khaustov 2008, 2016a, Ebermann & Krisper 2014
Scutacarus subtilis Rack, 1966; Austria; i; Friedl 2000, Ebermann & Krisper 2014; “Variatipes"- group: no claw on leg I
Scutacarus tackei Willmann, 1942; Austria, Czechoslovakia, France, Germany, Hungary, India, Switzerland, former Yugoslavia; p; Ebermann 1978
Scutacarus tutus Khaustov, 2008; Russia, Ukraine, i; Khaustov 2008, 2016c
Scutacarus velutinosus Sevastianov, 1983, Ukraine; p; Sevastianov 1983
Thaumatopelvis reticulatus Ebermann, 1980; USA (Maryland); p; Ebermann 1980b
Formica spp.
Imparipes (Imparipes) cf. histricinus Berlese, 1903; Austria; i; Mahunka 1970b
Imparipes (Imparipes) cf. histricinus Berlese, 1903; Czechoslovakia; n.k.; Mahunka 1967b
Imparipes (Imparipes) circinnatus Mahunka, 1980; Austria; p; Ebermann & Krisper 2014
Imparipes (Imparipes) comatus Mahunka, 1970; Hungary; i; Mahunka 1970b, 1980
Imparipes (Imparipes) cunicularius Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) gagati Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) nescius Khaustov, 2008; Russia, Ukraine; ip; Khaustov 2008, 2015b
Imparipes (Imparipes) nugax Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) pennatus Karafiat, 1959; Germany, Hungary; n.k.; Karafiat 1959, Mahunka 1972b
Imparipes (Imparipes) robustus Karafiat, 1959; Austria, Bulgaria, France, Germany, Hungary, Japan, Mongolia, Russia, Ukraine (Palaearctic?); p; Mahunka 1967b, 1972b, Friedl 2000, Khaustov 2008, 2015b, Ebermann & Krisper 2014, also in soil, litter
Imparipes (Imparipes) sp.B (sensu Friedl 2000); Austria; p; Friedl 2000; only 1 specimen
Imparipes (Imparipes) tenuis Mahunka, 1981; Austria; i; Friedl 2000
Scutacarus atypicus (Karafiat, 1959); Austria, Germany, Hungary, Russia; p; Karafiat 1959, Mahunka 1972b, Friedl 2000, Ebermann & Krisper 2014, Khaustov 2015b
Scutacarus avarus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Scutacarus carsticus Mahunka & Mahunka-Papp, 1980; Hungary, Ukraine; ip; Khaustov 2008
Scutacarus concinnus Mahunka, 1964; Hungary; i; Nemestóthy & Mahunka 1981
Scutacarus hungaricus Mahunka, 1965; Hungary; i; Nemestóthy & Mahunka 1981
Scutacarus karafiati Khaustov, 2015; Russia; p; Khaustov 2015b
Scutacarus kassaii Mahunka, 1965; Austria, Hungary, Russia; p; Ebermann 1978
Scutacarus nudus (Berlese, 1886); Germany; n.k.; Parmentier et al. 2015; “Variatipes"- group: no claw on leg I
Scutacarus palustris Rack, 1966, Hungary; i; Mahunka 1968b
Scutacarus pilatus Khaustov, 2008; Russia, Ukraine, ip; Khaustov 2008, 2015b
Scutacarus ponticulus Mahunka, 1981; Hungary; i; Nemestóthy & Mahunka 1981
Scutacarus pygmephoroides Mahunka, 1967; Russia; i; Khaustov 2015a
Scutacarus rotundatus [sic]; Czech Republic; n.k.; Štorkán 1940; should be S. rotundus (Berlese, 1903)
Scutacarus rotundus (Berlese, 1903); Austria, Germany, Hungary, Italy, Russia; ip; Berlese 1903, Paoli 1911, Karafiat 1959, Mahunka 1970a, 1970b, 1972b, Khaustov 2015b
Scutacarus silvestri (Berlese, 1903); Austria; p; Friedl 2000, Ebermann & Krisper 2014
Scutacarus stammeri Karafiat, 1959; Germany, Hungary, Italy, Ukraine; i; Karafiat 1959, Mahunka 1972b, Khaustov 2008
Scutacarus tackei Willmann, 1942; Austria; p; Ebermann & Krisper 2014
Paratrechina jaegerskioeldi
Scutacarus gouheri Metwali & Ahmed, 1987; Egypt; p; Metwali & Ahmed 1987
Scutacarus notabilosimillis Metwali & Ahmed, 1987; Egypt; p; Metwali & Ahmed 1987
VII. SCUTACARID SPECIES ASSOCIATED WITH MYRMICINAE (HEXAPODA, ECTOGNATHA; ACULEATA, FORMICIDAE)
Acromyrmex lundi
Imparipes (Telodispus) formicarum Lombardini, 1960; Argentina; n.k.; Lombardini 1960
Aphenogaster spp.
Imparipes (Imparipes) obstinatus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) ursus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Scutacarus amoenus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Scutacarus ellipticus Karafiat, 1959; Austria; p; Ebermann & Krisper 2014
Scutacarus ovoideus Karafiat, 1959; Austria; p; Ebermann & Krisper 2014
Scutacarus sp.; Austria; p; Ebermann & Krisper 2014
Scutacarus tacitus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Atta cephalotes
Imparipes (Imparipes) atta Delfinado & Baker, 1976; Panama; n.k.; Delfinado & Baker 1976
Crematogaster spp.
Lophodispus irregularis (Mahunka, 1971); USA (Maryland); i; Ebermann 1980b
Scutacarus nudus bisetus Karafiat, 1959; USA (Maryland); i; Ebermann 1980b; “Variatipes"- group: no claw on leg I
Scutacarus rapoporti Mahunka, 1968; USA (Alabama); i; Ebermann 1980b
Messor spp.
Imparipes (Imparipes) histricinus Berlese, 1903; Angola, Australia, Austria, Brazil, Hungary, Italy, Mongolia, Tunisia, Ukraine, former Yugoslavia; i; also in soil
Imparipes (Imparipes) ignotus Khaustov & Chydyrov, 2004; Turkmenistan; i; Khaustov & Chydyrov 2004, Khaustov 2008
Imparipes (Imparipes) longicaudus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) messori Metwali & Ahmed, 1987; Egypt; p; Metwali & Ahmed 1987
Imparipes (Imparipes) moderatus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) morosus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) obsoletus Rack, 1966; holarctic; n.k.; Khaustov 2008; also in soil
Imparipes (Imparipes) placidus Khaustov & Chydyrov, 2004; Turkmenistan; i; Khaustov & Chydyrov 2004, Khaustov 2008
Scutacarus fimbrillatosimilis Metwali & Ahmed 1987; Egypt; p; Metwali & Ahmed 1987
Scutacarus quaesitus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Monomorium salomonis
Imparipes (Imparipes) messori Metwali & Ahmed, 1987; Egypt; p; Metwali & Ahmed 1987
Myrmica spp.
Imparipes (Imparipes) charkoviensis Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) circinnatus Mahunka, 1980; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) comatosimilis Metwali, 1981; Austria, Poland; p; Metwali 1981, Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) comatus Mahunka, 1970; France, Hungary,Ukraine; i; Khaustov 2008
Imparipes (Imparipes) histricinus Berlese, 1903; Austria; i; Ebermann 1980a; also in moss
Imparipes (Imparipes) quaesitus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) sp.B (sensu Friedl 2000); Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) sp. C (sensu Friedl 2000); Austria; p; Friedl 2000, Ebermann & Krisper 2014
Scutacarus angustus Mahunka, 1970; Austria, Hungary, Russia, Ukraine; i; Mahunka 1970b, 1972b; “Variatipes"- group: no claw on leg I
Scutacarus atypicus (Karafiat, 1959); Austria, Germany; i; Ebermann 1980a
Scutacarus crassisetus (Paoli, 1911); Austria; i; Mahunka 1970b; also in moss
Scutacarus ellipticus Karafiat, 1959; Austria, Germany, Hungary, Russia, Switzerland, Ukraine, former Yugoslavia; ip; Karafiat 1959, Mahunka 1972b, 1977c, Friedl 2000, Khaustov 2008, Ebermann & Krisper 2014; also in soil
Scutacarus eucomus (Berlese, 1908); Austria; i; Mahunka 1970b; also in soil; “Variatipes"- group: no claw on leg I
Scutacarus hauseri Mahunka, 1977; France; n.k.; Mahunka 1977c
Scutacarus kassaii Mahunka, 1965; Russia; i; Khaustov 2015a
Scutacarus latus Karafiat, 1959; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Scutacarus longisetus (Berlese, 1904); Austria, Bulgaria, Hungary, Italy, Poland, Switzerland, Ukraine, former Yugoslavia; i; Mahunka 1977c, Ebermann 1978, Khaustov 2008
Scutacarus myrmecophilus Metwali, 1981; Poland, Ukraine; i; Metwali 1981, Khaustov 2008
Scutacarus myrmicinus Khaustov, 2015; Russia; i; Khaustov 2015c
Scutacarus ovoideus Karafiat, 1959; Austria, Germany, Greece, Hungary, Poland, Switzerland, Ukraine; ip; Mahunka 1977c, Ebermann 1979, Friedl 2000, Khaustov 2008, Ebermann & Krisper 2014
Scutacarus pegazzanoae Ebermann, 1986; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Scutacarus plumosus (Paoli, 1911); Austria; p; Friedl 2000, Ebermann & Krisper 2014
Scutacarus quadrangularis (Paoli, 1911); Austria; i; Mahunka 1970b; also in moss, litter, soil; “Variatipes"- group: no claw on leg I
Scutacarus subtilis Rack, 1966; Austria; i; Mahunka 1970b; also in moss; “Variatipes"- group: no claw on leg I
Scutacarus tackei Willmann, 1942; Austria, Czechoslovakia, France, Germany, Hungary, India, Switzerland, former Yugoslavia; p; Ebermann 1978, Ebermann & Krisper 2014; also in soil
Scutacarus yuliae Khaustov, 2006; Ukraine; p; Khaustov 2006, 2008
Pheidole pallidula
Imparipes (Imparipes) kugitangensis Khaustov & Chydyrov, 2004; Turkmenistan, i; Khaustov 2008
Scutacarus magyarosimilis Metwali & Ahmed, 1987; Egypt; p; Metwali & Ahmed 1987
Pogonomyrmex occidentalis
Imparipes (Imparipes) latispinus Mahunka, 1970; USA (Nebraska); n.k.; Mahunka 1970c
Scutacarus wranoskyi Mahunka, 1970; USA (Nebraska); n.k.; Mahunka 1970c
Solenopsis spp.
Imparipes (Imparipes) histricinus Berlese, 1903; Italy; p; Berlese 1903
Imparipes (Imparipes) cf. parapicolosimilis Metwali, 1981; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) louisianae Ebermann & Moser, 2008; USA (Louisiana); p, Ebermann & Moser 2008, Moser & Blomquist 2011; on alates
Scutacarus andrassyi Mahunka, 1968; USA (Louisiana); n.k.; Ebermann & Moser 2008, Moser & Blomquist 2011
Scutacarus n.sp. near deserticolus Mahunka, 1969; USA (Louisiana); n.k.; Ebermann & Moser 2008, Moser & Blomquist 2011
Scutacarus nanus Ebermann & Moser, 2008; USA (Louisiana); n.k.; Ebermann & Moser 2008, Moser & Blomquist 2011
Scutacarus tertius Ebermann & Moser, 2008; USA (Louisiana); n.k.; Ebermann & Moser 2008, Moser & Blomquist 2011
Temnothorax spp.
Imparipes (Imparipes) histricinus Berlese, 1903; Iran; p; Loghmani et al. 2014
Scutacarus claviger (Paoli, 1911); Iran; p; Loghmani et al. 2014
Tetramorium spp.
Imparipes (Imparipes) cf. imaginatus Mahunka, 1981; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) cf. tenuis Mahunka, 1981; Austria; p; Friedl 2000, Ebermann & Krisper 2014
Imparipes (Imparipes) comatus Mahunka, 1970; France, Hungary, Ukraine; i; Khaustov 2008
Imparipes (Imparipes) hortobagyensis Mahunka, 1981; Hungary, Ukraine; i; Khaustov 2008
Imparipes (Imparipes) histricinus Berlese, 1903; Austria, Czech Republic, Italy; ip; Berlese 1903, Štorkán 1940, Ebermann 1980a; also in moss
Imparipes (Imparipes) lentus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) parapicolosimilis Metwali, 1981; Poland; i; Metwali 1981
Imparipes (Imparipes) paucus Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Imparipes (Imparipes) robustus Karafiat, 1959; Austria, Bulgaria, France, Germany, Hungary, Japan, Mongolia, Russia, Ukraine (Palaearctic?); n.k.; Khaustov 2008; also in soil, litter
Imparipes (Imparipes) sklyari Khaustov, 2008; Ukraine; n.k.; Khaustov 2008
Scutacarus bursula (Berlese, 1903); Germany, Italy, Ukraine; ip; Paoli 1911, Khaustov 2008
Scutacarus cf. paolii Mahunka, 1965; Austria; p; Ebermann & Krisper 2014
Scutacarus ellipticus Karafiat, 1959; Austria; p; Ebermann & Krisper 2014
Scutacarus longisetus (Berlese, 1904); Austria, Bulgaria, Hungary, Italy, Poland, Switzerland, Ukraine, former Yugoslavia; i; Khaustov 2008
Scutacarus paolii Mahunka, 1965; Austria, Hungary; i; Ebermann 1980a; also in soil
Scutacarus rakonczayi Mahunka, 1981; Austria; p; Ebermann & Krisper 2014
Scutacarus subquadratus Khaustov & Chydyrov, 2004; Turkmenistan; n.k.; Khaustov 2008
Stenamma debile
Imparipes (Imparipes) cf. imaginatus Mahunka, 1981; Austria; p; Ebermann & Krisper 2014
Imparipes (Imparipes) cf. parapicolosimilis Metwali, 1981; Austria; p; Ebermann & Krisper 2014
“Myrmicinae" (not further classified in literature)
Imparipes (Imparipes) rectangulatus Mahunka, 1977; USA (Kansas); n.k.; Mahunka 1977a
Scutacarus rettenmeyeri Mahunka, 1977; USA (Kansas); n.k.; Mahunka 1977a
VIII. SCUTACARID SPECIES ASSOCIATED WITH AVES AND MAMMALIA
Aves: Accipitridae
Scutacarus ameropannonicus Mahunka & Philips, 1978; USA (New York); i; Mahunka & Philips 1978; in fecal pellet
Aves: “Antarctic birds"
Scutacaridae; Antarctica; p; Krivolutsky et al. 2004
Aves: Procellariidae
Puffinus tenuirostris
Heterodispus longisetosus (Womersley, 1955); Australia; i; Womersley 1955
Aves: Strigiformes
Pygmodispus (Pygmodispus) abestus Mahunka & Philips, 1978; USA (New York); i; Mahunka & Philips 1978; in fecal pellet
Scutacarus subsphaeroideus Mahunka & Philips, 1978; USA (New York); i; Mahunka & Philips 1978; in fecal pellet
Scutacaridae; USA (Massachusetts); i; Philips et al. 1988
Aves: “birds" (not further classified in literature)
Imparipes (Imparipes) obsoletus Rack, 1966; USA (New York); i; Delfinado et al. 1976
Scutacarus imitans Delfinado & Baker, 1976; USA (New York); i; Delfinado& Baker 1976
Scutacarus meansi Delfinado & Baker, 1978; USA (New York); i; Delfinado & Baker 1978; also in dog food (sic!)
Scutacarus subaffinis Delfinado & Baker, 1978; USA (New York); i; Delfinado & Baker 1978
Scutacarus unicus indefinitus Delfinado & Baker, 1978; USA (New York); i; Delfinado & Baker 1978
Scutacarus uniformis Delfinado et al., 1976; USA (New York); i; Delfinado et al. 1976
Mammalia: Eulipotyphla
Soricidae: Blarina brevicauda
Scutacarus impar Delfinado & Baker, 1976; USA (New York); i; Delfinado & Baker 1976; in wet habitats
Scutacarus subspinosus Delfinado & Baker, 1976; USA (New York); i; Delfinado & Baker 1976
Soricidae: Crocidura suaveolens
Reductacarus singularis Mahunka, 1963; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus eucomus (Berlese, 1908); Ukraine; i; Sevastyanov & Uzhevskaya 2003; “Variatipes"- group: no claw on leg I
Soricidae: Sorex sp.
Scutacarus apodemi Mahunka, 1963; Hungary; i; Mahunka 1963
Scutacarus ormayi Mahunka, 1963; Hungary; i; Mahunka 1972b
Scutacarus spinosus Storkan, 1936; Hungary; i; Mahunka 1963; in meadow soil
“Soricidae" (not further classified in literature)
Scutacarus imitans Delfinado & Baker, 1976; USA (New York); i; Delfinado & Baker 1976
Scutacarus subspinosus Delfinado & Baker, 1976; USA (New York); i; Delfinado & Baker 1976
Talpidae: Talpa europea
Scutacarus acarorum (Goeze, 1780); Europe; n.k.; Oudemans 1913
Scutacarus mahnerti Mahunka, 1972; Austria; i; Mahunka 1972a
Scutacarus mendax Karafiat, 1959; Slovakia; i; Kalúz 2001
Scutacarus talpae (Oudemans, 1913); Netherlands; i; Oudemans 1913; phoretic on gamasids in nest!
“Talpidae" (not further classified in literature)
Scutacarus subterraneus (Oudemans, 1913); Balkans, Germany, i; Karafiat 1959; also in meadow soil
Mammalia: Rodentia
Cricetidae: Clethrionomys glareolus
Scutacarus subterraneus (Oudemans, 1913); Ukraine; i; Sevastyanov & Uzhevskaya 2003
Cricetidae: Cricetulus migratorius
Heterodispus elongatus (Paoli, 1911) (?); Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) comatus Mahunka, 1970; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) obsoletus Rack, 1966; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Lophodispus irregularis (Mahunka, 1971); Ukraine; i; Sevastyanov & Uzhevskaya 2003; syn. Imparipes (I.) taurensis Sevastianov, 1975
Pygmodispus (Pygmodispus) zicsii Mahunka, 1964; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus eucomus (Berlese, 1908); Ukraine; i; Sevastyanov & Uzhevskaya 2003; “Variatipes"- group: no claw on leg I
Scutacarus kassaii Mahunka, 1965; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus quadrangularis (Paoli, 1911); Ukraine; i; Sevastyanov & Uzhevskaya 2003; “Variatipes"- group: no claw on leg I
Scutacarus spinosus Storkan, 1936; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus tacensis Mahunka, 1964; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Cricetidae: Microtus arvalis
Heterodispus elongatus (Paoli, 1911); Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) carabidophilus Sevastianov, 1974; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) cavernophilus Sevastianov, 1974; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) crassimerus Mahunka, 1968; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) degenerans Berlese, 1904; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) hungaricus Balogh & Mahunka, 1962; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) kossensis Sevastianov, 1975; Ukraine; i; Sosnina & Sevastianov 1975
Imparipes (Sporichneuthes) kaszabi Mahunka, 1967; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Lophodispus irregularis (Mahunka, 1971); Ukraine; i; Sosnina & Sevastianov 1975, Sevastyanov & Uzhevskaya 2003; syn. Imparipes (I.) taurensis Sevastianov, 1975
Scutacarus culmusophilus Sevastianov, 1975; Ukraine; i; Sosnina & Sevastianov 1975
Scutacarus occultatus Sevastianov, 1975; Ukraine; i; Sosnina & Sevastianov 1975
Cricetidae: Microtus brandti
Imparipes (Imparipes) mongolicus Mahunka, 1967; Hungary, Mongolia; i; Mahunka 1967a, 1972b
Imparipes (Sporichneuthes) kaszabi Mahunka, 1967; Mongolia; i; Mahunka 1967a
Reductacarus singularis Mahunka, 1963; Mongolia; i; Mahunka 1967a
Scutacarus sphaeroideus Karafiat, 1959; Mongolia; i; Mahunka 1967a
Scutacarus tacensis Mahunka, 1964; Hungary, Mongolia; i; Mahunka 1967a, 1972b
Cricetidae: Microtus pennsylvanicus
Scutacarus spinosus Storkan, 1936; USA (New York); i; Delfinado & Baker 1976; also in leaf litter
Cricetidae: Microtus sp.
Reductacarus singularis Mahunka, 1963; Hungary; n.k.; Mahunka 1972b
Scutacarus deserticolus Mahunka, 1969; Hungary, Mongolia; i; Mahunka 1969a, 1972b
Cricetidae: Peromyscus leucopus
Scutacaridae; USA (Illinois); p; Drummond 1957, Basolo & Funk 1974
Geomyidae: Geomys bursarius
Imparipes (Imparipes) spickai Mahunka, 1977; USA (Illinois); p; Mahunka 1977d
Scutacarus geomyi Mahunka, 1977; USA (Illinois); p; Mahunka 1977d
Scutacarus missouriensis Mahunka, 1977; USA (Missouri); p, Mahunka 1977d
Geomyidae: Geomys bursarius illinoensis
Imparipes (Imparipes) spickai Mahunka, 1977; USA (Illinois); p; Spicka 1981
Scutacarus geomyi Mahunka, 1977; USA (Illinois); p; Spicka 1981
Geomyidae: Geomys bursarius missouriensis
Scutacarus missouriensis Mahunka, 1977; USA (Missouri); p; Spicka 1981
Heteromyidae: Chaetodipus spinatus lambi
Imparipes (Imparipes) cupes Delfinado & Baker, 1978; Mexico; p; Estébanes-González & Cervantes 2005, skin of dorsal region; in small cavities that appear as scabs
Heteromyidae: Dipodomys spectabilis
Scutacaridae; Australia; i; Seastedt et al. 1986
Muridae: Apodemus agrarius
Heterodispus elongatus (Paoli, 1911) (?); Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) obsoletus Rack, 1966; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus eucomus (Berlese, 1908); Ukraine; i; Sevastyanov & Uzhevskaya 2003; “Variatipes"- group: no claw on leg I
Muridae: Apodemus flavicollis
Imparipes (Imparipes) degenerans Berlese, 1904; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) histricinus Berlese, 1903; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Lophodispus irregularis (Mahunka, 1971); Ukraine; i; Sosnina & Sevastianov 1975, Sevastyanov & Uzhevskaya 2003; syn. Imparipes (I.) taurensis Sevastianov, 1975
Scutacarus culmusophilus Sevastianov, 1975; Ukraine; i; Sosnina & Sevastianov 1975, Sevastyanov & Uzhevskaya 2003
Scutacarus indistinctus Sevastianov, 1975; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus palustris Rack, 1966; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus tener Sevastianov, 1975; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Muridae: Apodemus sylvaticus
Heterodispus citelli Mahunka, 1970; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Heterodispus elongatus (Paoli, 1911); Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Apidacarus) platycephalus Sevastianov, 1974; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) carabidophilus Sevastianov, 1974; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) cavernophilus Sevastianov, 1974; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) comatus Mahunka, 1970; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) crassimerus Mahunka, 1968; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) histricinus Berlese, 1903; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Imparipes) penicillatus Mahunka, 1967; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Imparipes (Sporichneuthes) kaszabi Mahunka, 1967; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Reductacarus singularis Mahunka, 1963; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus apodemi Mahunka, 1963; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus culmusophilus Sevastianov, 1975; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus eucomus (Berlese, 1908); Ukraine; i; Sevastyanov & Uzhevskaya 2003; “Variatipes"- group: no claw on leg I
Scutacarus kassaii Mahunka, 1965; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus quadrangularis (Paoli, 1911); Ukraine; i; Sevastyanov & Uzhevskaya 2003; “Variatipes"- group: no claw on leg I
Scutacarus sphaeroideus Karafiat, 1959; Ukraine; i; Sevastyanov & Uzhevskaya 2003
Scutacarus spinosus Storkan, 1936; Hungary, Ukraine; i; Sevastyanov & Uzhevskaya 2003, Mahunka 1963
Muridae: Apodemus sp.
Scutacarus apodemi Mahunka, 1963; Hungary, Ukraine; i; Mahunka 1963, Sosnina & Sevastianov 1975
Scutacarus ormayi Mahunka, 1963; Hungary; i; Mahunka 1972b
Scutacarus spinosus Storkan, 1936; Hungary; i; Mahunka 1963; also in soil
Muridae: Desmodillus auricularis, Dipodillus harwoodi, Gerbilliscus (= Tatera) leucogaster, Rhabdomys pumilio
Heterodispus foveatus Jagersbacher-Baumann & Ebermann, 2012; Kenya, Namibia, Rwanda, South Africa; ip; Jagersbacher-Baumann & Ebermann 2012a
“Muridae" (not further classified in literature)
Diversipes exhamulatus (Michael, 1886); Germany; i; Rack 1966; also in soil, decaying material
Imparipes (Imparipes) obsoletus Rack, 1966; Germany; i; Rack 1966
Scutacarus peractus Karafiat, 1959; Germany; i; Rack 1966; also in soil, decaying material
Sciuridae: Citellus citellus
Heterodispus citelli Mahunka, 1970; Hungary; i; Mahunka 1970a
Heterodispus elongatus (Paoli, 1911); Hungary; i; Balogh & Mahunka 1962
Heterodispus pannonicus Mahunka, 1963; Hungary; i; Mahunka 1963, Jagersbacher-Baumann & Ebermann 2012b
Reductacarus singularis Mahunka, 1963; Hungary; i; Mahunka 1963
Scutacarus spinosus Storkan, 1936; Hungary; i; Mahunka 1963; also in meadow soil
Sciuridae: Sciurus carolinensis
Scutacarus imitans Delfinado & Baker, 1976; USA (New York); p; Delfinado & Baker 1976
Sciuridae: Xerus inauris
Heterodispus foveatus Jagersbacher-Baumann & Ebermann, 2012; South Africa; i; Jagersbacher-Baumann & Ebermann 2012a
Mammalia: “rodents" (not further classified in literature)
Heterodispus tragardhi (Delfinado & Baker, 1976); USA (New York); i; Delfinado & Baker 1976
Scutacarus kassaii Mahunka, 1965; Ukraine; i; Sklyar & Sevastianov 1997
Mammalia: “small mammals" (not further classified in literature)
Scutacarus longisetus (Berlese, 1904); Slovenia; i; Mahunka 1975b; also in ant nests
Scutacarus major (Paoli, 1911); Slovenia; i; Mahunka 1975b
Scutacarus plumosus (Paoli, 1911); Slovenia; i; Mahunka 1975b
Scutacarus quadrangularis (Paoli, 1911); Slovenia; i; Mahunka 1975b; “Variatipes"- group: no claw on leg I
Scutacarus sphaeroideus Karafiat, 1959; Slovenia; i; Mahunka 1975b
Scutacarus spinosus Storkan, 1936; Slovenia; i; Mahunka 1975b
Mammalia: carrion of Felis silvestris catus
Scutacaridae; USA (Hawaii); n.k.; Early & Goff 1986
acarologia_4238_Supplementary-Material.pdf



2017-05-04
Date accepted:
2017-09-04
Date published:
2018-02-07
Edited by:
Bertrand, Michel

This work is licensed under a Creative Commons Attribution 4.0 International License
2018 Baumann, Julia
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)