The marine-associated lifestyle of ameronothroid mites (Acari, Oribatida) and its evolutionary origin: a review
Pfingstl, Tobias1
1Institute of Zoology, University of Graz, Universitätsplatz 2, 8010 Graz, Austria.
2017 - Volume: 57 Issue: 3 pages: 693-721
https://doi.org/10.24349/acarologia/20174197Keywords
Abstract
The marine intertidal zone constitutes one of the most interesting ecosystems on earth. It represents the border of two completely different worlds, the marine and the terrestrial realms, where environmental parameters are changing constantly, hence challenging the ability of organisms to cope with these extreme conditions. epVery few mite groups have been able to accomplish the land to sea transition, and they are scattered over various taxa. The prostigmatid Halacaridae represent a group that has even managed to cross this line between land and sea completely having radiated in marine waters. They are cosmopolitan, occurring from shallow waters to the bathyal and even the abyssal (e.g. Bartsch 2004) and hence represent true marine organisms. Other mite groups have not crossed this barrier, though they have successfully invaded the littoral zone. arAmong Mesostigmata, some genera of the family Ologamasidae, for example Hydrogamasus and Psammonsella, have adapted to intertidal marine habitats. Members of Halolaelapidae, e.g. Halolaelaps and Halodarcia, are also known to inhabit littoral algae and beach wrack in coastal zones. Representatives of Parasitidae and Laelapidae are likewise common predators in these habitats (e.g. Lindquist et al. 2009). The same applies to representatives of the prostigmatid families Bdellidae, Rhagidiidae and Erythraeidae (Walter et al. 2009). Littoral species that forage on marine algae can also be found in the endeostigmatic genus Nanorchestes, and the whole astigmatine family Hyadesiidae is composed of marine intertidal mites, occurring on virtually all coastlines (e.g. Schuster 1979). Last, several isolated oribatid mite species, for example Hermannia subglabra Berlese, 1910, Acrotritia clavata (Märkel 1964), Passalozetes bidactylus (Coggi, 1900), Punctoribates quadrivertex (Halbert, 1920), Haloribatula tenareae Schuster, 1957 etc. are associated with marine shores; they are thalassophilous or thalassobiont, meaning respectively that they occur in coastal areas or they have completely adapted to marine-associated environments (e.g. Schuster 1957, 1966, 1979, 1989; Luxton 1967, Syamjith and Ramani 2013). Other than Halacaridae, all the above mentioned groups are examples of infrequent marine associations within larger evolutionary clades suggesting that the invasion of coastal habitats happened repeatedly and relatively recently in geological time. arOnly one group of oribatid mites stands out as supposedly representing an ancient, monophyletic taxon that evolved and radiated in the marine littoral environment, the superfamily Ameronothroidea. More than 90% of oribatid mite species living in intertidal habitats belong to this superfamily (Procheş and Marshall 2001) and they are characteristic elements of the marine littoral fauna. The first member of this assemblage was discovered more than 140 years ago (Thorell 1871), and since then three families, 17 genera and approximately 84 species have been described (Subías 2004, update 2016). Despite a large literature concerning diverse aspects of this substantial group, the present state of knowledge still shows many gaps, relationships are largely unclear, and the evolutionary processes that led to the occupation of intertidal habitats are scarcely understood. Most of the literature is highly limited in scope, and only a few comprehensive studies include all members of Ameronothroidea (Wallwork 1964, Weigmann and Schulte 1977, Behan-Pelletier 1997).arThe aim of the present paper is to summarize and synthesize existing literature on ameronothroid mites and to provide a concise and coherent overview of the biological, ecological, zoogeographical and evolutionary aspects of the marine-associated lifestyle of this taxon. Additionally, theories about the evolutionary invasion of littoral habitats by ameronothroid mites are reviewed and evaluated based on recent findings.
Data presented in this paper are based on published literature, but some unpublished photographs, electron microscopic images and distribution maps are added to illustrate various topics. Light photographs were made with an Olympus E5 digital SLR camera attached to either an Olympus BH-2 microscope (for micrographs) or to an Olympus SZX 12 stereo microscope (for normal photographs). Electron micrographs were taken at the Research Institute for Electron Microscopy and Fine Structure Research, Graz, University of Technology (FELMI) using a Leitz AMR microscope and a Zeiss Gemini Ultra 55. For this purpose specimens stored in 70% ethanol were dehydrated in ascending ethanol concentrations, mounted on aluminium stubs with double-sided sticky tape, and then sputter coated with gold–palladium.
Brachypyline classification used in this text follows the broad outlines of Norton and Behan-Pelletier (2009). The term “Ameronothridae sensu stricto (s. str.)” refers to the northern hemispheric ameronothrid taxa (in the sense of Vitzthum 1942); the more inclusive term “Ameronothridae sensu lato (s. l.)” refers to this northern hemispheric component plus all southern hemispheric Podacaridae (in the sense of Wallwork 1964). These terms are only used in the Taxonomy and Systematics Section, in all other sections these groups are given as Ameronothridae and Podacaridae. A general overview of oribatid morphology is given by Norton and Behan-Pelletier (2009).arSpecies authors of all marine-associated ameronothroid mites are given in Table 2 and therefore they are not cited when they are mentioned for the first time in text. However, species authors for non-ameronothroid taxa are given in the text.
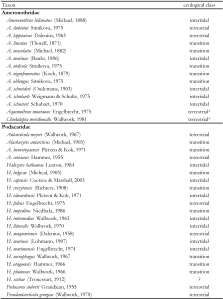
At present, different classifications of the superfamily Ameronothroidea can be found in the literature (Table 1), and several taxonomic issues are still unresolved. However, all of these classifications (e.g. Schulte and Weigmann 1977, Subías 2004, Norton and Behan-Pelletier 2009 etc.) acknowledge the Ameronothridae, Fortuyniidae and Selenoribatidae as ameronothroid families. The marine-associated lifestyle of most members of this group was surely one of the main reasons for their unification, but this motivation resulted in some ambiguous or conflicting taxonomic arrangements that are best understood in a historical context.



The first marine-associated oribatid mites were discovered in the late nineteenth century, but were mostly classified in genera that have very different concepts today. Thorell (1871) described the first ameronothroid mite, Ameronothrus lineatus (in Eremaeus), from the polar Island Spitsbergen. In subsequent years, further littoral species of mainly polar and cold temperate coasts were found and described by the most renowned acarologists of that time (e.g. L. Koch 1879, Michael 1882, Oudemans 1903). Berlese proposed the first modern ameronothroid genera, Ameronothrus (Berlese 1896) and Halozetes (Berlese 1916) but included the former in Carabodidae. Years later Willmann (1931) recognized that Ameronothrus represents an independent taxon; accordingly Vizthum (1942) cited Willmann, 1931 as the family author. However, Willmann did not mention the family name in his work and, following the rules of the code of nomenclature, only the author giving the name for the first time is to be seen as the taxon author. Consequently, Vitzthum (1942) is the author of Ameronothridae. In his essay on the classification of oribatid mites, Grandjean (1954) included Halozetes in Ameronothridae and confirmed the distinct status of this family. Only one year later, Grandjean (1955) described Podacarus auberti, another littoral species from an Antarctic island. He noted its systematic relationship to Ameronothridae, but proposed the family Podacaridae based on divergent morphological features of the immatures of this species. Van der Hammen (1960) was the first to find an intertidal species in tropical regions and included this new taxon, Fortuynia marina, in Podacaridae. When van der Hammen (1963) proposed Fortuynia yunkeri, the second species of this genus, he also described its juvenile stages; their morphology diverged conspicuously from juveniles of Podacaridae, and accordingly he excluded Fortuynia from this family and proposed the family Fortuyniidae within Ameronothroidea. Shortly afterwards, Wallwork (1964) considered the proposal of Fortuyniidae justified, and included only Podacarus, Alaskozetes and Halozetes in his revision of Podacaridae. Nevertheless, Balogh (1965) ignored these changes and assigned the genus Fortuynia again to Podacaridae, regarding the obvious differences as only of generic significance. Luxton (1967) disagreed with Balogh’s opinion and confirmed again the validity of Fortuyniidae bringing this debate for now to an end; since then, Fortuyniidae are commonly accepted as a distinct taxon of Ameronothroidea.arHowever, the discovery of further ameronothroid species in subsequent years led to a controversy about the systematic status of Podacaridae and Ameronothridae. Based on detailed morphological investigations, Schubart (1975) stated that these two families are likely to be two subfamilies of a single family. Weigmann and Schulte (1977) supported Schubart’s hypothesis and included all former podacarid genera in Ameronothridae arguing that there is no decided gap among the taxa and that their groupings into separate monophyletic families are no longer justified. Some authors (Subías 2004, Norton and Behan-Pelletier 2009) accepted this taxonomic reorganization, others (Luxton 1990b, Woas 2002) still recognized Podacaridae and so both systematic groupings still are found in the literature.arThe ameronothroid family Selenoribatidae, on the other hand, has never been seriously challenged. Strenzke (1961) described the first selenoribatid species, Selenoribates foveiventris, from coasts of the Red Sea, then Schuster (1963) proposed two years later Selenoribatidae, when he discovered Thalassozetes riparius, another intertidal species from Mediterranean shores. Grandjean (1966) was the first to give a detailed diagnosis for this family and later (Grandjean 1968) provided further important taxonomic and systematic information strengthening the concept of Selenoribatidae. Since then the number of selenoribatid taxa quintupled and though there have been some problems at the generic or species level (e.g. synonymies, unjustified transfers; for details see Pfingstl and Schuster 2012a), the family remained a well delimited and established monophyletic group. arThe last and probably the most overlooked ameronothroid family are the Tegeocranellidae which differ in their ecology from nearly all other members in being exclusively associated with freshwater habitats. The distinctness of this monogeneric taxon was never questioned but the position within the system of higher oribatids and especially its inclusion within the Ameronothroidea is still controversial. Balogh and Balogh (1992) included this taxon initially in the Polypterozetoidea, a superfamily characterized as eupheredermous Oribatida, which means the nymphs retain the exuvial scalp of the preceding stage after the moult. But nymphs of Tegeocranellidae do not retain scalps and are thus classified as apheredermous. Behan-Pelletier (1997) noticed this fundamental mistake and moreover demonstrated plausibly that the family Tegeocranellidae is indeed a member of the Ameronothroidea, closely related to Fortuyniidae and Selenoribatidae. Although Behan-Pelletier’s reasoning was based on clear synapomorphic characters and thus conclusive, several authors seem to have simply neglected her excellent work. For example, in his annually updated checklist of oribatid mites, Subías (2004) includes the Tegeocranellidae in the superfamily Tectocepheoidea without any explanation and therefore causes persisting confusion about the systematic relationship of this family. arIn summary, a universally accepted systematic concept of Ameronothroidea is lacking and certain groupings, e.g. Podacaridae included in Ameronothridae (Weigmann and Schulte 1977) or Tegeocranellidae included in Tectocepheoidea (Subías 2004), remain questionable.
Marine-associated Ameronothroidea can be found in all oceans, and their distribution extends from coasts of the Arctic to shores of the Antarctic (Figure 1). The occurrence of the families is strongly correlated with different climate zones, i.e. the Ameronothridae and Podacaridae are predominantly contained within cold-temperate regions around both poles (e.g. Procheş and Marshall 2001), and only a small fraction of northern hemispheric Ameronothridae can be found beyond the limits of these areas. Ameronothrus bilineatus, A. marinus, A. schneideri and A. schusteri may occur towards the transition-zone to the tropics (Schulte 1975) though only A. maculatus clearly stands out from the other cold-adapted taxa with records from coasts of Tunisia (Schulte 1975) and the tropical Caribbean Island Curaçao (Willmann 1936).
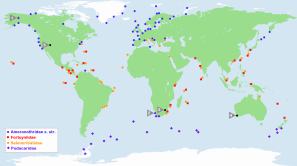


In contrast to the northern hemispheric Ameronothridae, the southern hemispheric Podacaridae include exclusively cold-adapted taxa and are largely confined to Antarctic and Sub-Antarctic regions (Procheş and Marshall 2001). The only occurrence outside these polar areas is that of Halozetes capensis, a possible endemic species living on temperate shores of South Africa (Coetzee and Marshall 2003).
The families of Fortuyniidae and Selenoribatidae, on the other hand, are limited to shorelines of the subtropics and tropics (e.g. Schuster 1989) and hence they appear to be warm-adapted mites. Most members can be found in the tropical Eastern Pacific, the tropical Western Atlantic and the Indo-Pacific (Procheş and Marshall 2001, Pfingstl and Schuster 2014) while the least inhabited area is the Mediterranean-Atlantic region.
Besides the aerial climate, a few authors (Schuster 1966, Pfingstl and Schuster 2014, Pfingstl 2016) suggested that the temperature of large ocean currents may also shape distributional patterns. For example, warm-adapted selenoribatid mites occur abundantly on the South American Atlantic coast where the warm Brazil Current flows southwards, whereas these mites are absent from the Pacific shore at the same latitudes where the cold Humboldt Current prevails (Pfingstl and Schuster 2014). A similar situation is found at the southern tip of Africa: cold-adapted ameronothrid species occur on the west coast where the cold Benguela current is present, and warm-adapted fortuyniid and selenoribatid taxa inhabit the east coast where the warm Agulhas Current flows (Pfingstl 2016). arHowever, aerial and oceanic climate are closely linked, and we do not know the relative importance of air or water temperature in the distribution of these taxa. But given the fact that stenotopic intertidal species are spending half of their lives in the seawater, it is likely that water temperature influences their biogeography. arAlthough the worldwide distribution of Ameronothroidea shows a relatively well-defined pattern, two exceptional records have seemed enigmatic. The first is the discovery of Alaskozetes coriaceus (Hammer 1955) from an arctic hill in Alaska. Marie Hammer described this species and proposed the genus Alaskozetes based on a single specimen that was given to her from that location. Surprisingly, this was the only Alaskozetes specimen ever found in the northern hemisphere, all others have been collected later in the southern hemisphere from Sub-Antarctic and Antarctic areas, where this genus is abundant. Such a disjunctive distribution on opposite Polar Regions, with more than 10 000 km in between, is more than extraordinary. It was suggested that the finding in Alaska may have been caused by migratory birds flying from Pole to Pole with the mites attached to their feathers (Schuster 1966). The second biogeographic puzzle was the finding of Ameronothrus bilineatus specimens from the littoral zone of South Africa (Weigmann 1975b). Before that, this species was known to occur only in the northern hemisphere, namely on European coasts with a latitudinal distribution range from Scandinavia to Portugal. Although Weigmann (1975b) mentioned the possibility of birds having transported specimens from Europe to South Africa, he favored the idea of a continuous distribution from Southern Europe along the African west coast. Procheş and Marshall (2001), on the other hand, clearly classified the South African A. bilineatus population as introduced, but offered no potential explanation, so this biogeographic mystery also remains unsolved.
Vast coastlines of each continent are still largely uncharted in terms of intertidal oribatid mite occurrences and thus known biogeographic patterns may change considerably in the future.
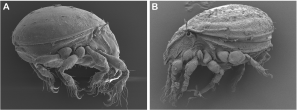
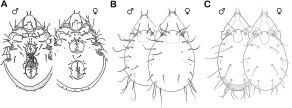
Luxton (1990b) was correct in noting that marine-associated oribatid mites appear at first sight to show no modifications for life on the sea shore. Being air-breathing animals that have not completely transgressed the ecological barrier between marine and terrestrial environments, they strongly resemble typical terrestrial oribatid mites (Figure 2). Nevertheless, evolutionary adaptations to the intertidal lifestyle can be found in all biological aspects of these mites.


Daily tidal submergence is surely one of the main selective factors shaping the morphology of intertidal mites. Schubart (1971, 1975) noticed a causal correlation between frequent inundation and reduction of sensilli in the Ameronothridae. Species living predominantly in supralittoral zones (the uppermost area of the littoral zone), like Ameronothrus lineatus or A. maculatus, in general have small sensilli while species dwelling in the intertidal zone, like A. bilineatus and A. marinus, have lost their sensilli and bothridia completely. A reduction of these sensory organs may prevent overstimulation when the mite is submerged and subject to strong tidal wave action and constantly moving water. Indeed, the majority of ameronothroid mites show relatively simple and short sensilli. Members of the genus Fortuynia, which live in the lower eulittoral area (e.g. Pfingstl 2013b), exhibit especially short and compact sensilli.
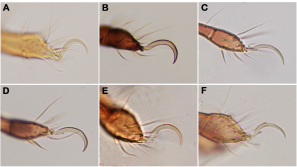
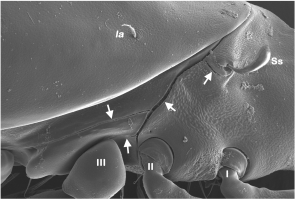
Another morphological trait affected by tidal wave action is the tarsal claws of the legs. Karasawa and Hijii (2004b) demonstrated that monodactylous species dominate in littoral environments and consequently they stated that a reduction in the number of claws may be an evolutionary adaptation to regular tidal flooding. All members of Fortuyniidae and Selenoribatidae, which are stenotopic inhabitants of the intertidal zone, show only a single claw on their legs, whereas Ameronothridae and Podacaridae, which have both intertidal and non-intertidal representatives, possess either one or three claws. Concerning the latter, Schubart (1975) noted that Ameronothrus species living on hard substrates (e.g. rocks, shore fortifications) are tridactylous, while species dwelling on soft substrates (e.g. salt meadows, silted rocks) are monodactylous, accordingly he postulated a correlation between type of substratum and claw number. Apart from the claw number, Pugh et al. (1987b) demonstrated that claws of intertidal Ameronothrus species are proportionally longer than those of congeneric supralittoral species. Indeed, the majority of intertidal oribatid species possesses elongated claws (Figure 3) allowing them to grip the substrate more tightly during tidal flooding (Karasawa and Hijii 2004b); these claws may be equipped with spine-like structures (Figures 3D–F), as in most selenoribatid species (e.g. Strenzke 1961, Grandjean 1968, Pfingstl 2013c, 2015b).


Intertidal ameronothroid mites are air-breathing animals respiring with a tracheal system, therefore they had to evolve special adaptations in order to survive daily tidal flooding. Ameronothroid mites are known to use plastron respiration when flooded. A plastron is an alternative respiration system absorbing oxygen from the surrounding water via a thin layer of air trapped by hydrophobic hairs or other cuticular projections (definition after Fielden et al. 2011) and is known to occur in many aquatic and semi-aquatic arthropod taxa, like insects and spiders. Plastrons of intertidal ameronothroid mites (Figure 4) are associated with cuticular and/or cerotegumental structures (e.g. Pugh et al. 1987a, 1990; Messner et al. 1992, Pfingstl and Krisper 2014). It was demonstrated by Pugh et al. (1987a) that the cerotegument of Ameronothrus marinus contains approximately 21 % airspace and that of A. maculatus even 60%. Although the air-retaining surface structures are more or less similar in all investigated taxa, the specific mode of plastron respiration differs considerably among them. For example, in flooded fortuyniid and selenoribatid adults the retained layer of air always communicates with the tracheal system (Pfingstl and Krisper 2014), whereas in submerged Ameronothrus individuals no such connection was found, and diffusion of oxygen from the plastron is assumed to occur across the weakly sclerotized cuticle instead via the tracheae (Pugh et al. 1987b, 1990). Similarly, in juvenile Fortuyniidae and Selenoribatidae air is retained in certain large lateral and ventral folds of the soft integument, and these folds are equipped with series of pores leading into tracheal structures supplying the internal organs with oxygen (Pfingstl and Krisper 2014). In Ameronothrus nymphs, on the other hand, the folding pattern is conspicuously different, tracheal structures are absent and gas transfer is assumed to take place across the whole soft integument (Messner et al. 1992).



Adults of Fortuyniidae have furthermore evolved a unique complex system of lateral cuticular taenidia, also called van der Hammen’s organ (e.g. Hammen 1963, Travé 1986, Pugh et al. 1990) (Figure 5). This system of taenidia connects the dorsal and ventral plastron area and is thought to facilitate rapid equilibrium of pressure changes between the respiratory system and the surrounding environment caused by rough wave movements (Pugh et al. 1990). Although present in all fortuyniid members, the specific configuration of taenidia slightly varies among the genera (e.g. Pfingstl and Schuster 2012b, Pfingstl and Krisper 2014).


Several authors investigated the efficiency of plastron respiration in certain ameronothroid taxa and demonstrated that these mites are able to survive astonishingly long periods of time under flooded conditions (Table 3). Alismobates inexpectatus, Fortuynia atlantica and Carinozetes bermudensis specimens survived an average of one month under water (Pfingstl 2013a), A. lineatus and A. marinus specimens more than two months (Schuster 1979, Pugh et al. 1987a) and A. maculatus tolerated inundation for more than one month (Schuster 1966). Maximum survival times ranged from nearly two to eight months in these species (Schuster 1966, 1979; Pfingstl 2013b).
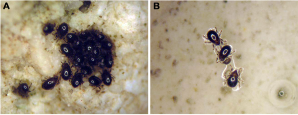
Intertidal mites are confined to a relatively few microhabitats, such as rocky shore lichens, kelp holdfasts, etc., which provide protection from direct and indirect stresses of severe wave action (Pugh and Mercer 2001), but behavioural adaptations further minimize these stresses. Several marine-associated oribatid mites are relatively quick and active climbers (Pfingstl 2013a); this allows them to escape rising tide and retreat into crevices or other protected spots. There may be a relation among the speed of this locomotion, the substrate and the mite’s vertical occurrence in the littoral area (Pfingstl 2013a). Species dwelling on rocky exposed substrate in the lower eulittoral, such as F. atlantica, are very fast climbers (Pfingstl 2013a), while species living in the upper and more vegetated eulittoral area, e.g. Halozetes otagoensis, Ameronothrus schneideri and Alaskozetes antarcticus, are moderate or even sluggish runners (Luxton 1966, 1990a, Strong 1967, Goddard 1979). The more exposed and closer the animals are to the water’s edge, the more quickly they must react and move to avoid being washed away. arAnother behavioural strategy to prevent dislodgement by waves is aggregation. This has been observed often and in many intertidal taxa, e.g. Fortuyniidae (Krisper and Schuster 2008, Pfingstl 2013a), Ameronothridae (Schulte et al. 1975, Schulte 1976b, Bücking et al. 1998, Søvik 2004) and Podacaridae, (Peckham 1967, Goddard 1979, Convey 1994b) where hundreds of individuals of different age are crouched next to and on top of each other (Figure 6A). Protection against wave action is a primary function of this behaviour, but in some instances aggregations may aid mate location (Block and Convey 1995) and synchronization of moults (Søvik 2004).


Certain fortuyniid and selenoribatid species show thigmotactic behaviour (Pfingstl 2013a) by entering narrow crevices and small depressions when these are present. This behaviour is advantageous in a periodically flooded environment because it limits the time the animals spend in open areas, exposed to the tidal surge. Intertidal oribatid mites usually are either inactive (e.g. Schulte 1973, Behan-Pelletier and Eamer 2007) or show low activity when flooded (Pfingstl 2013a). Pugh and King (1986) demonstrated that intertidal mites have activity cycles synchronized with diurnal and circatidal patterns, so that they are most active during periods of low tide and bright daylight. Individuals of A. marinus even perform rhythmic daily vertical migrations to avoid tidal flooding. In this way, they reduce the time of submergence and extend the time of possible food intake (Schulte 1973).arThe temperate species A. lineatus shows another interesting strategy to reduce or avoid the mechanical stress of wave movement. In summer, when reproduction reaches its peak, adults migrate into the uppermost eulittoral where they lay eggs; offspring develop there for the next two months, then deutonymphs move back to the littoral zone where development is completed (Bücking et al. 1998). By these seasonal vertical migrations the dangers of the tidal cycle are avoided for at least the most vulnerable stages: eggs, larvae and protonymphs.
Despite these strategies, intertidal mites surely are surprised often by waves and washed away, and for such an event the tropical F. atlantica has evolved an interesting behaviour. When exposed to a sudden flood, specimens immediately splay the legs, so that they float like a raft on the surface of the water until they again reach firm substrate (Pfingstl 2013a) (Figure 6B). By this so-called “floating behaviour” the mites increase their chances of survival in the open water and successful dispersal by ocean currents (Pfingstl 2013a).
As noted, aggregation may facilitate finding mates (Block and Convey 1995) or deposition of eggs in areas far above tidal range (Bücking et al. 1998), but there are several other reproductive traits that have evolved as adaptations to the marine environment. For example, most Ameronothridae are ovoviviparous (for a detailed discussion of this phenomenon see Norton 1994), with the larva having developed within the mother’s body and hatching from the egg immediately (larviparity) or within a few hours after deposition (Luxton 1966, Weigmann 1975a, Bücking et al. 1998, Søvik 2003, Søvik et al. 2003). This way, the vulnerable egg stage is extremely shortened and the mobile larva can promptly escape tidal waves. The tropical Fortuyniidae and Selenoribatidae, on the other hand, are oviparous; their egg stage takes approximately 15–35 days, similar to those of typical terrestrial oribatid mites (Pfingstl 2013a). However, they protect their eggs from tidal movement by either gluing them into tiny crevices of the substrate, as for example in F. atlantica, or they push them deep into the intertidal algae so that the algal mass completely encloses and protects the eggs, e.g. as in A. inexpectatus (Pfingstl 2013a).arSpermatophores of males appear even more fragile structures that are easily destroyed by the mechanical effects of the tides, but no special littoral adaptations are known, which Alberti (1974) already demonstrated for Trombidiformes. Typical stalked spermatophores were found in Ameronothridae (Luxton 1966, Schuster 1979, Søvik 2004), Fortuyniidae and Selenoribatidae (Pfingstl 2013a). Pfingstl (2013a) suggested that spermatophore deposition, detection and utilization by females may happen quickly during the short time frame of low tide. In this way spermatophores would not come in contact with water and hence would be safe. arDistinct sexual dimorphism is present in a small percentage of ameronothroid species, where it is expressed to different degrees and in various regions of the body (Pfingstl 2015a, Behan-Pelletier and Eamer 2010, Behan-Pelletier 2015) (Figure 7). In A. lineatus and A. nigrofemoratus, males exhibit conspicuously longer legs than females (e.g. Schubart 1975) whereas in the podacarid species Podacarus auberti, Halozetes belgicae and Alaskozetes antarcticus males show a remarkable aggenital neotrichy which is absent in females (e.g. Wallwork 1964). Not a single case of sexual dimorphism is known in selenoribatid mites, whereas a handful of fortuyniid species have elaborate sexually dimorphic characters. Fortuynia yunkeri males show only slight modifications of the fourth leg (Hammen 1963) but males of F. atlantica possess certain lanceolate notogastral setae, enlarged notogastral porose areas and a pair of obvious lateral protuberances on the notogaster (Krisper and Schuster 2008); in males of F. dimorpha the posterior part of the gastronotic region is completely covered with a porose area and several notogastral setae are conspicuously elongated (Pfingstl 2015a). The function of these modified characters is unknown but if they are linked to dermal glands like the octotaxic system of brachypyline mites (Alberti et al. 1997, Norton and Alberti 1997, Norton et al. 1997) and porose areas of mixonomatid Collohmanniidae (Norton and Sidorchuk 2014), they probably play a role in semiochemical communication linked with associative mating (Pfingstl 2015a, Behan-Pelletier 2015). Although never observed, direct sperm transfer in the littoral environment would be clearly advantageous because it reduces the stress for the sperm package in this intermittently dry and wet habitat.


However, at least two fortuyniid species have managed to completely avoid the stress of tidal inundation on spermatophores by simply not producing them. In Fortuynia hawaiiensis and Fortuynia maledivensis males are completely absent and females reproduce via thelytoky (Pfingstl and Jagersbacher-Baumann 2016). This reproductive mode entails a few advantages because it reduces costs of producing males, mate finding and sperm transfer in the extreme conditions of intertidal environments and furthermore it facilitates the colonization of new coastlines.
As far as known, marine-associated oribatid mites are generalized feeders with various degrees of specialization (e.g. Schulte 1979, Pfingstl 2013a), and usually the substrate serves them both a habitat (or protection against desiccation and dislodgment) and a food source (Bücking et al. 1998, Pfingstl 2013a) (Figure 8). Most Ameronothridae, Podacaridae and several Fortuyniidae and Selenoribatidae have been observed feeding on diverse types of green algae, as for example the microphytes Pleurococcus sp. or Chlorococcus sp., the filamentous algae Lyngbia sp. or Rhizoclonium riparium, macrophytes Enteromorpha sp. or Prasiola crispa and seaweed Porphyra sp. (Peckham 1967, Schulte 1976b, Pugh 1995, Pfingstl 2013a). Lichens are also an important food source for intertidal mites (e.g. Block and Convey 1995), but the mites feed predominantly on the phycobionts, i.e. the algal cells, of the lichens (e.g. Schulte 1976b, Bücking et al. 1998). In some cases Ameronothrus species were reported to feed on fungi (Luxton 1966, Schulte 1976b). Certain fortuyniid and selenoribatid species as well as the ameronothrid A. maculatus are known to primarily graze on cyanobacteria (Schuster 1979, Søvik 2004, Krisper and Schuster 2008). Carnivory has not yet been confirmed in any intertidal oribatid species. Schuster (1977) once inferred that Selenoribatidae may be carnivorous as he found pieces of tardigrades in their guts, but soon after he withdrew his idea (Schuster 1979).



Food preferences may differ among the developmental stages (Luxton 1966). Adults and tritonymphs of most Ameronothrus species feed on micro- and macroalgae, while the larvae, proto- and deutonymphs feed exclusively on microalgae. This sometimes leads to a spatial separation of the stages, with the younger ones staying in higher littoral areas (Schulte 1976b, Bücking et al. 1998). In most Ameronothrus species a correlation between habitat and type of food can also be observed, accordingly: (1) inhabitants of rocky intertidal shores predominantly feed on epilithic algae (algivorous), (2) mites dwelling on hard substrates in supralittoral areas prefer lichens (lichenivorous) and (3) species occurring in salt marshes mostly graze on fungi (fungivorous) (Schulte 1976b).arFeeding activity and defecation of Ameronothrus species follow a tidal rhythm, with maxima between the high tides (Schulte 1976b, Pugh and King 1986). This suggests that they feed as much as possible during low tide and then rest during high tide when they are submerged. Schulte (1976a) demonstrated that inhabitants of the lower eulittoral, exposed to longer periods of submergence, start feeding earlier and feed more quickly during low tide than do populations of the upper eulittoral, which are subject to shorter periods of inundation and hence have more time to feed.
There is a large amount of literature – enough to fill a book – dealing with physiological adaptations of ameronothroid mites but more than 90 % of it focuses on the ability of Antarctic Podacaridae to survive the extreme low temperatures and conditions of polar areas. These studies (e.g. Sømme and Block 1984, Convey 1994a, Block and Convey 1995, Worland and Lukešová 2000, Worland and Convey 2001, Deere and Chown 2006, Hawes et al. 2007, Benoit et al. 2008) are important to understand the mechanisms of survival in such cold environments, but studies investigating physiological adaptations to the marine environment are few, so our knowledge is sketchy. arIntertidal oribatid mites are resistant to salt and fresh water and as mentioned above, they can survive inundation for weeks and sometimes months (e.g. Schuster 1979, Pfingstl 2013b) (Table 3). How osmoregulation works in these animals is unclear but probably they possess the ability of hypertonic regulation to water of lower osmotic values than their hemolymph, such as fresh water, and to water of higher values, i.e. salt water, they are probably poikilosmotic (Schuster 1979). It has been suggested that littoral mites use their coxal glands for osmoregulation (Alberti and Storch 1977) or that they reduce ion resorption from excretory waste filtrate if their coxal glands are degenerated (Woodring 1973).
Ameronothroid mites have colonized various microhabitats of the marine littoral, even though this represents only a spatially restricted habitat. Ameronothridae and Podacaridae show various degrees of association with the intertidal ecosystem, including (1) typical intertidal species, e.g. A. marinus; (2) so-called transition species, able to dwell in both environments, e.g. A. lineatus; and (3) typical terrestrial species, as for example P. auberti (Schulte and Weigmann 1977) (see Table 2). There is a distinct latitudinal component to this habitat specificity: species at lower latitudes in each hemisphere are restricted to littoral environments while species found at higher latitudes show a greater affinity for terrestrial habitats (Schulte et al. 1975, Marshall and Convey 2004). Furthermore, several northern hemisphere Ameronothridae are also able to colonize brackish and freshwater habitats, such as marine ponds and estuaries (e.g. Schulte et al. 1975). Especially A. maculatus shows high freshwater tolerance and can frequently be found on the shores of coastal rivers (e.g. Schulte et al. 1975, Schuster 1986); in one case individuals were found in a small river 50 km upstream from the Mediterranean coast (Travé 1963). Some members of Ameronothridae can also be found far inland in typical terrestrial habitats, as for example the high arctic Ameronothrus lapponicus (Dalenius 1963) or the Scandinavian Ameronothrus dubinini, which was discovered a few kilometers inland (Laine et al. 1988). Within the littoral environment, Ameronothridae may occur on sediment-free rocky coasts (e.g. Bücking et al. 1998), on sediment-rich hard substrates (e.g. Schulte et al. 1975), in salt-marshes (e.g. Luxton 1966) and in washed-ashore flotsam or tidal debris (e.g. Schulte et al. 1975, Pugh and MacAlister 1994). There are also reports of quite unusual habitats. For example, the Antarctic Alaskozetes antarcticus was found near seal wallows or around penguin rookeries (Peckham 1967, Block and Convey 1995) but also on dead birds and well-rotted seal carcasses (Goddard 1979); Ameronothrus lineatus was collected from the nests of three different seabirds on Spitsbergen (Coulson et al. 2009) and Antarcticola georgiae and Halozetes belgicae were sampled from whale bones on sub-Antarctic coasts (e.g. Goddard 1979, Pugh and MacAlister 1994). Independent of microhabitat and taxon, investigations have shown that intertidal and terrestrial species show generally high niche specificity whereas transition species are eurytopic and may occur in a wide range of niches (Schulte et al. 1975, Marshall et al. 1999).


In contrast to Ameronothridae and Podacaridae, the subtropical and tropical Fortuyniidae and Selenoribatidae are stenotopic inhabitants of the littoral area, i.e. transition species or terrestrial species are lacking in these taxa. Collectively, Selenoribatidae species may inhabit a wide variety of intertidal habitats, e.g. sandy beaches, boulder beaches, rocky cliffs and mangrove roots (e.g. Karasawa and Aoki 2005, Pfingstl 2013a). Mangrove forests are one of the most important environments for tropical intertidal oribatid mites (Karasawa and Hijii 2004a) and some species, e.g. Carinozetes mangrovi, seem to be exclusively associated with these habitats (Pfingstl et al. 2014). Fortuyniidae species, on the other hand, display a very narrow range of habitats. Nearly all of them inhabit rocky shores; only three out of 20 species have been reported to occur occasionally in mangrove forests (e.g. Pfingstl 2015c). Pfingstl (2015c) suggested that the homogeneous morphology of the genus Fortuynia may be associated with this limited habitat preference. Several Fortuynia species may also dwell in empty barnacles or oyster shells, and are the only intertidal mites known to do so (Hammen 1963; Aoki 1974; Luxton 1990a, 1992; Marshall and Pugh 2002; Krisper and Schuster 2008). There is one record of F. smiti found in a riverine environment which represents the most unusual occurrence of this taxon (Pfingstl 2015c).



Given the strong ecological connection of ameronothroid mites to the littoral zone habitat, dispersal by active migration would be possible only along the coastline, but the fact that these mites have also reached geologically young and remote oceanic islands clearly shows that there are other ways of dispersal. There are several dispersal mechanisms that may be responsible for long-distance transport of these flightless arthropods, but the scarcity of evidence has allowed no consensus on their reality or relative importance. An overview of these possible dispersal mechanisms follows. ar(1) Anthropogenic dispersal seems unlikely as intertidal mites do not occur on commercially used plants or soils. Although the transport of these mites in bilge tanks of large vessels cannot be excluded, shipping activities are recent in geological times and may not have seriously influenced present distribution and population patterns. Nevertheless, with growing globalization activities this mode of dispersal may well have an impact on patterns in the near future.ar(2) Aerial dispersal (anemochory) is another possible mechanism but experimental data on this topic are rare and controversial. In an early study (Glick 1939), in which insect traps were installed on airplanes, mites were trapped on various occasions at different altitudes, but nearly all trapped specimens belonged to phoretic mite taxa that use insects as transport hosts. In a later study (Gressitt and Yoshimoto 1974), using fine meshe aerial nets in Alaska, a few oribatid mites were caught drifting in the wind, and a more recent study (Lehmitz et al. 2011) demonstrated that oribatid mites can be dispersed by wind up to at least 160 m above ground level, though the majority of these wind-transported mites consisted of species usually found in tree habitats. In contrast, a similar survey found no evidence of wind dispersal of mites (Coulson et al. 2003). Moreover, powerful storms would be necessary to propel flightless arthropods long distances of a few thousand kilometers and even if this would happen, these animals would not survive the extreme conditions, such as low pressure and freezing temperatures at high altitudes (Pugh 2003). Intertidal oribatid mites are neither phoretic on insects, nor do they inhabit trees and therefore aerial dispersal is a quite unlikely mode of long-distance transport for them.ar(3) Bird-mediated transport has been discussed as another possible way of reaching and colonizing remote islands (e.g. Schatz 1991, 1998; review by Lebedeva and Lebedev 2008) but again, evidence is scarce and authors are of different minds as to the importance of this mode of distribution. In a long-term study of birds in polar areas, several terrestrial mites were found on a few occasions, either in bird nests or plumage (Lebedeva and Lebedev 2008, Lebedeva 2012) and hence birds are believed by these authors to be the main suppliers of soil mites to isolated archipelagos.arConsidering the terrestrial nature of high Arctic and Antarctic Ameronothridae, it is indeed possible that dispersal occasionally occurs in this way. For example, it would explain the already mentioned mysterious findings of Alaskozetes coriaceus and Ameronothrus bilineatus in hemispheres opposite from their main distributions (Schuster 1966, Weigmann 1975b). In the above-mentioned study of seabird nesting colonies on Spitsbergen, A. lineatus individuals were found in a few nests (Coulson et al. 2009), hence the basic prerequisite for dispersal by birds was met. Nevertheless, the same study could find no evidence of the mites being actually transported the by birds. Pugh (1997) dismissed a priori bird-mediated transport of oribatid mites to oceanic islands, arguing that mites dispersed by birds usually show specific morphological adaptations to attach to the bird’s body. Marine-associated ameronothroid mites show no morphological modification for phoresy, though their long hook-like claws may facilitate such transport. While transport in bird plumage remains a possible, if improbable, dispersal mechanism for Ameronothridae, it almost certainly has no role for Fortuyniidae or Selenoribatidae: species of these exclusively intertidal families do not dwell in the vicinity of seabird nests or colonies. ar(4) Another potential method to reach remote islands is through transport by ocean currents (hydrochory). First evidence was provided by a study in which pleuston nets on boat trips between the islands of the Galápagos archipelago were used after a heavy storm, and a remarkable number of living oribatid mite individuals was found floating in debris or directly in the sea (Peck 1994). To survive such a transport, tolerance of long periods of submersion is necessary, which littoral mites possess, as discussed above (e.g. Schuster 1979, Pugh et al. 1987a). Coulson et al. (2002) further demonstrated that even terrestrial arthropods with different biologies are able to survive in salt water for over 14 days and therefore transoceanic dispersal by mites and collembolans may be a common phenomenon. Based on mean survival times of Bermudian intertidal oribatid mite species, individuals are able theoretically to survive transport in seawater along the Gulf Stream over a distance of 3,000 km (Pfingstl 2013b). Moreover, the floating behaviour of certain Fortuynia species (Pfingstl 2013a) as well as the possession of buoyancy-giving plastrons in supposedly all ameronothroid taxa, would contribute to their hydrochorous dispersal. Therefore, most authors (e.g. Schatz 1991, 1998; Coulson et al. 2002; Pfingstl 2013b) agree that long-distance dispersal of oribatid mites to oceanic islands is mainly hydrochorous.
An extensive fossil record since the Middle Devonian (388 ± 3,8 mya) indicates that oribatid mites are an ancient taxon (Norton et al. 1988). A molecular genetic study (Schaefer et al. 2010) even suggests that oribatid mites were among the first terrestrial colonizers and had already originated in the Precambrian (571 ± 37 mya). The colonization of land started in the interstitial zone of coastal habitats and finally resulted in the occupation of all terrestrial habitats on earth. Marine-associated mites as well as the exclusively marine halacarid mites demonstrate that some of them not just evolved from water to land but later also from land to water.
Schuster (1966) was the first to theorize about the origin of the intertidal lifestyle in oribatid mites. He suggested two possible ways: (a) terrestrial ancestors successively colonized the fringe of freshwater bodies, then brackish water habitats and finally the marine littoral; (b) fully terrestrial ancestors began to feed on washed-ashore flotsam, rich in organic matter, and subsequently specialized on other food resources present in the intertidal zone. Schuster admitted that both scenarios could have been concurrent, but considered it more likely that at least the Ameronothridae took the former evolutionary pathway. Indeed, certain members of Ameronothrus can be found regularly in estuaries (e.g. Schulte et al. 1975) and can withstand long periods of inundation not only with salt water but also with fresh water (Schuster 1979). Pfingstl (2013b) also discovered an unexpectedly high fresh water tolerance in the exclusively intertidal selenoribatid Carinozetes bermudensis and suggested it may be a relictual trait. A high general water tolerance was without a doubt a preadaptation necessary for mites to invade aquatic habitats, but it provides no evidence of evolutionary pathway, since some typical terrestrial species, e.g. Euzetes globulus, also can tolerate long periods of inundation (Schuster 1966).arSchulte and Weigmann (1977) provided another evolutionary scenario (Figure 9A) hypothesizing that a terrestrial ancestor of ameronothrid mites, inhabiting cold and wet soils, occupied larger portions of the world’s land masses and a warming of the world’s atmosphere scattered these animals to cooler locations. Subsequent glaciation events pushed these mites closer to the shore and finally forced them to invade polar coastlines. The chances of survival were higher near the warmer sea and so these coastal species radiated in the littoral environment and spread along the coast to warmer climates and this spreading happened independently in the Northern and Southern hemisphere. Marshall and Convey (2004) agreed that all ameronothroid lineages must ultimately have had an earlier terrestrial ancestry but they argued that at least recent colonization patterns of podacarid mites of the southern hemisphere cannot be explained with the aforementioned hypothesis. They stated that species living exclusively in intertidal habitats of the Antarctic and Sub-Antarctic are more primitive than those in supralittoral/terrestrial environments of coastal areas and proposed that colonization by ameronothrid (podacarid) mites in the southern hemisphere has proceeded from marine habitats to terrestrial areas following glacial retreat. However, a molecular genetic investigation of several Antarctic species of Podacaridae (Halozetes, Alaskozetes, Podacarus) found no evidence of speciation from an intertidal to a terrestrial group or vice versa (Mortimer et al. 2011). Although the above-mentioned theories (Schuster 1966, Schulte and Weigmann 1977, Marshall and Convey 2004) offer different opinions on recent evolutionary patterns, all agree on a monophyletic origin of the Ameronothroidea and their littoral lifestyle.
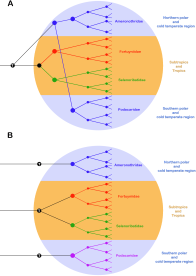


Procheş (2001), on the other hand, suggested that land-to-sea transition in secondary marine organisms, including ameronothroid mites, took place independently in three distinct latitudinal bands (northern cold-temperate, tropical, southern cold-temperate) (Figure 9B). He suggested that species interactions, i.e. competition, drove the origin of marine association in subtropical and tropical selenoribatid and fortuyniid mites, while glaciation is thought to be responsible for that of polar ameronothrid/podacarid mites (e.g. Schulte and Weigmann 1977, Procheş and Marshall 2001).
Schulte and Weigmann (1977) provided a thoughtful hypothesis concerning the monophyletic origin of the intertidal lifestyle which lead to the taxonomic unification of Ameronothridae and Podacaridae. The authors plausibly demonstrated that there are not enough morphological features separating these two taxa and indeed, presently there are only two characters which do not overlap: the presence in all Ameronothrus species (but absence in all Podacaridae) of brachytracheae in legs and the absence of setae it on leg IV of all Podacaridae (versus the presence in Ameronothridae). From a morphological point of view, the monophyly of this group seemed to be validated. arHowever, the molecular genetic study of Schäffer et al. (2010), encompassing selected oribatid mite species from diverse families, including A. maculatus and P. auberti, demonstrated a paraphyletic position of the two taxa, which clearly contradicts the conclusion of Weigmann and Schulte (1977). Wallwork (1964) had already argued that certain similar traits of the Podacaridae and Ameronothridae may have evolved independently. Both taxa inhabit predominantly cold and temperate regions and similar environmental properties may have led to similar convergent morphologies. The biogeographic distribution of Ameronothridae and Podacaridae – in opposite hemispheres without a noteworthy overlap – may point to an independent origin rather than to a common ancestry. The two often neglected and exceptional monotypic genera Aquanothrus and Chudalupia, occurring in ephemeral rock pools at modest elevations in semi-arid or arid regions of North America, South Africa and Western Australia, have been considered relictual ameronothrid species (Norton et al. 1997) that might have been linked biogeographically in the distant past. But there is evidence from another molecular genetic study (Mortimer et al. 2011) that Aquanothrus is not closely related to Podacaridae as the ceratozetoid genus Magellozetes intervenes between Aquanothrus and the Podacaridae. Consequently, these data also indicate that Ameronothridae and Podacaridae are supported as different groupings and that the current disjunct distribution, in opposite areas of the world, may not be associated with the relictual occurrences of Aquanothrus and Chudalupia. The biogeographic pattern of Ameronothroidea rather supports the theory of Procheş (2001), in which land-to-sea transitions took place independently in three distinct latitudinal bands, with the Ameronothridae in the northern cold-temperate, the Fortuyniidae and Selenoribatidae in the tropical, and the Podacaridae in the southern cold-temperate region. This theory is further supported by results of Krause et al. (2016) who investigated the evolution of aquatic life by oribatid mites using molecular genetics. They concluded that Fortuyniidae and Selenoribatidae colonized salt water habitats only once whereas the Ameronothridae invaded these habitats independently and hence they suggested that Ameronothroidea are a polyphyletic taxon. arBehan-Pelletier and Eamer (2007) argued that the evolution of respiratory organs in Oribatida was a critical step in the invasion of aquatic habitats, and indeed the development of plastron respiration was surely one of the most important adaptations allowing the colonization of the intertidal zone. Comparing plastron respiration systems in Ameronothroidea further supports the theory of independent land-to-sea transition events. Fortuyniidae and Selenoribatidae use similar plastrons and breathe with tracheal organs while submerged (Pfingstl and Krisper 2014), whereas Ameronothridae use different plastron organs and breathe across the thin weakly sclerotized cuticle during submergence (Pugh et al. 1987a, 1990; Messner et al. 1992).
In summary, biogeographic, molecular genetic and plastron respiration data contradict the theory of a monophyletic Ameronothroidea in its current sense, and suggest multiple independent origins of their marine-associated lifestyle.arNevertheless, biogeographic patterns are largely incomplete and many new taxa and occurrences remain to be found (Pfingstl and Schuster 2014). Furthermore, the recent molecular genetic studies (Schäffer et al. 2010, Mortimer et al. 2011, Krause et al. 2016), while informative, are certainly preliminary, as they include very few ameronothroid taxa and concentrated on different research questions. Finally, plastron structures were investigated in very few species, none of which was a member of Podacaridae.
Several authors (e.g. Subías 2004, Marshall and Pugh 2002) do not accept the monogeneric Tegeocranellidae as a member of Ameronothroidea because they do not consider the juvenile morphology to be important for systematic considerations. Given the above-mentioned doubts about the monophyly of Ameronothroidea, it may seem reasonable to agree with these authors. Nonetheless, Behan-Pelletier (1997) provided seven synapomorphies supporting a relationship among Tegeocranellidae, Fortuyniidae and Selenoribatidae. The most obvious synapomorphy is certainly the shared type of juvenile plication, namely a centrodorsal gastronotic plate framed by deep lateral and ventral folds and wrinkles, only present in immatures of these three taxa. Pfingstl and Krisper (2014) demonstrated that this specific type of plication plays an important role in plastron respiration of juvenile fortuyniid and selenoribatid mites. Though not investigated yet in tegeocranellid immature stages, they may use the same plastron mechanism and this could be a further indication of a common origin. Another morphological character associated with plastron respiration may also support a close relationship between Tegeocranellidae and Fortuyniidae. The already-mentioned van der Hammen’s organ of fortuyniid mites is part of the plastron system, and Behan-Pelletier (1997) noticed similar structures in Tegeocranellidae but refrained from considering these traits homologous. Considering a close relation between Tegeocranellidae and Fortuyniidae/Selenoribatidae, occupation of semiaquatic freshwater habitats may represent the more ancestral ecology. Based on these indications, the following scenario might be considered: a terrestrial ancestor colonized semiaquatic and aquatic freshwater habitats, e.g. ponds and streams, then some descendants diversified within these environments, evolving the Tegeocranellidae, and others colonized the marine littoral either by migrating downstream into brackish waters or by inhabiting coastal freshwater bodies that eventually became connected to the open ocean and evolved there to the Fortuyniidae and Selenoribatidae.
Ameronothroid intertidal oribatid mites have successfully colonized all shorelines, from biologically depauperate ice-cold polar coasts to lush humid tropical shores. Although air-breathing and similar in overall morphology to typical terrestrial taxa, they have evolved specific adaptations to the marine littoral, as for example modified tarsal claws, plastron respiration and ovoviviparity. Daily tidal cycles and associated wave action are the most important stress factors shaping these adaptations. Hydrochorous dispersal is considered the main long-distance transport mechanism in fortuyniid and selenoribatid mites, whereas bird-mediated transport may be responsible for dispersal events in northern ameronothrid mites.
Based on recent findings it is suggested that the families currently included in Ameronothroidea do not share a proximate common ancestry. They probably have colonized coastal environments at least three times, once in northern hemispheric polar and temperate areas by the Ameronothridae, once in the Tropics by ancestors of Fortuyniidae and Selenoribatidae and once in Antarctic and Sub-Antarctic regions by the Podacaridae. Accordingly, the family Ameronothridae sensu lato – i.e. sensu Weigmann and Schulte (1977) – is a polyphyletic grouping, and the unification of Ameronothridae with Podacaridae no longer seems justified. The two families probably evolved parallel in opposite Polar regions. The Fortuyniidae, Selenoribatidae and Tegeocranellidae, on the other hand, may still represent a monophyletic lineage which has evolved from a tropical ancestor that already showed an association with freshwater habitats.arAs Marshall and Convey (2004) already pointed out, a more complete understanding of the evolution of marine-associated mites will require thorough morphological and molecular phylogenetic investigations. Moreover, a comprehensive and holistic approach is needed to reveal natural relationships among alleged members of Ameronothroidea and to reconstruct their evolutionary pathway from a terrestrial ancestor to the sea.
This work was funded by the Theodor-Körner Fund, therefore my sincerest thanks go to this Fund and its sponsors; in difficult times when I considered abandoning my scientific career, I was awarded with the prestigious Theodor-Körner prize, which firstly gave me enough confidence to continue my research and secondly supported me financially to perform the present studies. My work would not have been possible without this support. Thanks also to G. Krisper for contributing interesting ideas, thoughts and pictures. Special thanks to Roy A. Norton for providing numerous constructive comments and for helping to improve the quality of this work. I also want to thank the Centre for Electron Microscopy Graz (FELMI) and its team for realizing the electron micrographs.
Abd-el-Hamid M.E. 1973 — Acari (Oribatei) aus Ägypten: Selenoribates ghardaqensis nov. sp. am Roten Meer — Anz. österr. Akad. Wiss., mathemat. naturwiss. Kl., 8: 53-56.
Alberti G. 1974 — Fortpflanzungsverhalten und Fortpflanzungsorgane der Schnabelmilben (Acarina: Bdellidae, Trombidiformes) — Z. Morph. Tiere, 78: 111-157. doi:10.1007/BF00298469 ![]()
Alberti G., Storch V. 1977 — Zur Ultrastruktur der Coxaldrüsen actinotricher Milben — Zool. Jb. Anat., 109: 394-425.
Alberti G., Norton R.A., Adis J., Fernandez N.A., Franklin E.N., Kratzmann M., Moreno A.I., Weigmann G. and Woas S. 1997 — Porose integumental organs of oribatid mites (Acari, Oribatida). 2. Fine structure — Zoologica, Stuttgart, 146, 33–114.
Aoki J. 1974 — The first record of the intertidal Oribatid genus, Fortuynia, from Asia — Annot. Zool. Jpn., 47(3), 170–174.
Balogh J. 1965 — A synopsis of the world Oribatid (Acari) genera — Acta Zool. Hung., 11: 5-99.
Balogh J., Balogh P. 1992 — The Oribatid Mites Genera of the world. Vol. 1 — Hungarian Natural History Museum: Budapest. pp. 263.
Banks N. 1896 —New North American spiders and mites — Transact. Amer. Entomol. Soc., 23: 57-77.
Bartsch I. 2004 — Geographical and ecological distribution of marine halacarid genera and species (Acari: Halacaridae) — Exp. Appl. Acarol., 34: 37-58. doi:10.1023/B:APPA.0000044438.32992.35 ![]()
Bayartogtokh B., Chatterjee T. 2010 — Oribatid mites from marine littoral and freshwater habitats in India with remarks on world species of Thalassozetes (Acari: Oribatida) — Zool. Stud., 49: 839-854.
Bayartogtokh B., Chatterjee T., Chan B.K.K., Ingole B. 2009 — New species of marine littoral mites (Acari: Oribatida) from Taiwan and India, with a Key to the world's species of Fortuynia and notes on their distributions — Zool. Stud., 48: 243-261.
Behan-Pelletier V.M. 1997 — The semiaquatic genus Tegeocranellus (Acari: Oribatida: Ameronothroidea) of North and Central America — Can. Entomol., 129: 537-577. doi:10.4039/Ent129537-3 ![]()
Behan-Pelletier V.M. 2015 — Review of sexual dimorphism in brachypyline oribatid mites — Acarologia, 55: 127-146.
Behan-Pelletier V.M., Eamer B. 2007 — Aquatic Oribatida: Adaptations, constraints, distribution and ecology — In: Morales-Malagara J.B., Behan-Pelletier V.M., Ueckermann E., Pérez T.M., Estrada-Venegas E.G., Badii M. (Eds.). Acarology XI: Proceedings of the International Congress. México: Instituto de Biología and Facultad de Ciencias, Universidad Nacional Autónoma de México; Sociedad Latinoamericana de Acarología. p. 71-82.
Behan-Pelletier V.M., Eamer B. 2010 — The first sexually dimorphic species of Oribatella (Acari, Oribatida, Oribatellidae) and a review of sexual dimorphism in the Brachypylina — Zootaxa, 2332: 1-20.
Benoit J.B., Yoder J.A., Lopez-Martinez G., Elnitsky M.A., Lee Jr. R.E., Denlinger D.L. 2008 — Adaptations for the maintenance of water balance by three species of Antarctic mites — Polar Biol., 31: 539-547. doi:10.1007/s00300-007-0385-9 ![]()
Berlese A. 1896 — Acari, Myriapoda et Scorpiones hucusque in Italia reperta. Ordo Cryptostigmata II (Oribatidae) — Padua: Portici. pp. 98.
Berlese A. 1910 — Acari nuovi. Manipoli V-VI — Redia, 6, 199-234.
Berlese A. 1916 — Centuria prima di Acari nuovi — Redia, 12: 19-67.
Block W., Convey P. 1995 — The biology, life cycle and ecophysiology of the Antarctic mite Alaskozetes antarcticus — J. Zool., London, 236: 431-449. doi:10.1111/j.1469-7998.1995.tb02723.x ![]()
Bücking J., Ernst H., Siemer F. 1998 — Population dynamics of phytophagous mites inhabiting rocky shores – K-strategists in an extreme environment? — In: Ebermann E. (Ed.). Arthropod Biology: Contributions to Morphology, Ecology and Systematics. Biosystematics and Ecology Series 14. Vienna: Österreichische Akademie der Wissenschaften. p. 93-143.
Coetzee L., Marshall D.J. 2003 — A new Halozetes species (Acari: Oribatida: Ameronothridae) from the marine littoral of southern Africa — African Zool., 38: 327-331.
Coggi A. 1900 — Nuovi Oribatidi Italiani — Bullettino della Societa Entomologica Italiana, (30), 309–324.
Convey P. 1994a — Growth and survival strategy of the Antarctic mite Alaskozetes antarcticus — Ecography, 17: 97-107.
Convey P. 1994b — Sex ratio, oviposition and early development of the Antarctic oribatid mite Alaskozetes antarcticus (Acari: Cryptostigmata) with observations on other oribatids — Pedobiologia, 38: 161-168.
Coulson S.J., Hodkinson I.D., Webb N.R., Harrison J.A. 2002 — Survival of terrestrial soil-dwelling arthropods on and in seawater: implications for trans-oceanic dispersal — Funct. Ecol., 16: 353-356. doi:10.1046/j.1365-2435.2002.00636.x ![]()
Coulson S.J., Hodkinson I.D., Webb N.R. 2003 — Aerial dispersal of invertebrates over a high-Arctic glacier foreland: Midtre Lovénbreen Svalbard — Polar Biol., 26: 530-537. doi:10.1007/s00300-003-0516-x ![]()
Coulson S.J., Moe B., Monson F., Gabrielsen G.W. 2009 — The invertebrate fauna of High Arctic seabird nests: the microarthropod community inhabiting nests on Spitsbergen, Svalbard — Polar Biol., 32: 1041-1046. doi:10.1007/s00300-009-0603-8 ![]()
Dalenius P. 1963 — Studies on the Oribatei (Acari) of the Torneträsk territory in Swedish Lapland. V. Some interesting species — Ark. Zool., 16: 1-8.
Dalenius P., Wilson O. 1958 — On the soil fauna of the Antarctic and of the Sub-Antarctic Islands. The Oribatidae (Acari) — Ark. Zool., 11: 393-425.
Deere J.A., Chown S.L. 2006 — Testing the beneficial acclimation hypothesis and its alternatives for locomotor performance — Amer. Nat., 168: 630-644. doi:10.1086/508026 ![]()
Engelbrecht C.M. 1974 — The genus Halozetes (Oribatei: Acari) on Marion Island — Navors. Nas. Mus., Bloemfontein, 3: 1-25
Engelbrecht C.M. 1975 — New ameronothroid (Oribatei: Acari) taxa from the Republic of South Africa and the islands Gough and Marion — Navors. Nas. Mus., Bloemfontein, 3: 53-88.
Ermilov S.G., Tolstikov A.V., Mary N., Schatz H. 2013 — Oribatid mites (Acari, Oribatida) from riverine environments of some islands in Oceania — Zookeys, 318: 47-57.
Fielden L.J., Knolhoff L.M., Villarreal S.M., Ryan P. 2011 — Underwater survival in the dog tick Dermacentor variabilis (Acari: Ixodidae) — J. Insect Physiol., 57: 21-26. doi:10.1016/j.jinsphys.2010.08.009 ![]()
Glick P.A. 1939 — The distribution of insects, spiders and mites in the air — Tech. Bull., 673: 1-151.
Goddard D.G. 1979 — Biological observations on the free-living mites of Signy Island in the maritime Antarctic — Brit. Antarct. Surv. Bull., 49: 181-205.
Grandjean F. 1954 — Essai de classification des Oribates (Acariens) — Bull. Soc. Zool. France, 78: 421-446.
Grandjean F. 1955 — Sur un acarien des iles Kerguelen. Podacarus auberti (Oribates) — Mém. Mus. Nat. Hist. Natur. Paris, 8: 109-150.
Grandjean F. 1966 — Selenoribates mediterraneus n.sp. et les Selenoribatidae (Oribates) — Acarologia, 8: 129-154.
Grandjean F. 1968 — Schusteria littorea n.g., n.sp. et les Selenoribatidae (Oribates) — Acarologia, 10: 116-150.
Gressitt J.L., Yoshimoto C.M. 1974 — Insect dispersal studies in Northern Alaska — Pac. Insects, 16: 11-30.
Halbert J.N. 1920 — The acarina of the seashore — Proceedings of the Royal Irish Academy, 35(B), 106–152.
Hammen van der L. 1960 — Fortuynia marina gen. nov., sp. nov., an oribatid mite from the intertidal zone in Netherlands New Guinea — Zool. Meded., 37: 1-9.
Hammen van der L. 1963 — Description of Fortuynia yunkeri nov. spec., and notes on the Fortuyniidae nov. fam. — Acarologia, 5: 152-167.
Hammer M. 1955 — Alaskan Oribatids — Acta Arctica, 7: 5-36.
Hammer M. 1966 — Investigations on the oribatid fauna of New Zealand — K. Dan. Vidensk. Selsk. Biol. Skr., 15: 1-108.
Hawes T.C., Bale J.S., Worland M.R., Convey P. 2007 — Plasticity and superplasticity in the acclimation potential of the Antarctic mite Halozetes belgicae (Michael) — J. Exp. Biol., 210: 593-601. doi:10.1242/jeb.02691 ![]()
Karasawa S., Aoki J. 2005 — Oribatid Mites (Arachnida: Acari: Oribatida) from the Marine Littoral of the Ryukyu Archipelago, Southwestern Japan — Species Div., 10: 209-233.
Karasawa S., Hijii N. 2004a — Effects of microhabitat diversity and geographical isolation on oribatid mite (Acari: Oribatida) communities in mangrove forests — Pedobiologia, 48: 245-255.
Karasawa S., Hijii N. 2004b — Morphological modifications among oribatid mites (Acari: Oribatida) in relation to habitat differentiation in mangrove forests — Pedobiologia, 48: 383-394.
Koch L. 1879 — Arachniden aus Sibirien und Novaya Zemlya, eingesammelt von der Schwedischen Expedition im Jahre 1875 — Kong. Sv. Vet. Akad. Handl., 16: 3-136.
Krause A., Pachl P., Schulz G., Lehmitz R., Seniczak A., Schaefer I., Scheu S., Maraun M. 2016 — Convergent evolution of aquatic life by sexual and parthenogenetic oribatid mites — Exp. Appl. Acarol., 70: 439¬-453.
Krisper G., Schuster R. 2008 — Fortuynia atlantica sp. nov., a thalassobiontic oribatid mite from the rocky coast of the Bermuda Islands (Acari: Oribatida: Fortuyniidae) — Ann. Zool., 58: 419-432. doi:10.3161/000345408X326753 ![]()
Laine A., Orell M., Itämies J. 1988 — The distribution and ecology of the mite Ameronothrus dubinini Sitnikova, 1977 (Acarina, Oribatei) in northern Finland — Ann. Zool. Fenn., 25: 253-256.
Lebedeva N.V. 2012 — Oribatid mites transported by birds to polar islands — In: Hempel G., Lochte K., Matlshov G. (Eds) Arctic marine biology. Bremerhaven: Helmholtz Gemeinschaft. p. 152-161.
Lebedeva N.V., Lebedev R. 2008 — Transport of oribatid mites to polar areas by birds — In: Bertrand M., Kreiter S., McCoy K.D., Migeon A., Navajas M., Tixier M-S., Vial L. (Eds) Integrative Acarology. Montpellier: Euraac. p. 359-367.
Lehmitz R., Russel D., Hohberg K., Christian A., Xylander W.E.R. 2011 — Wind dispersal of oribatid mites as mode of migration — Pedobiologia, 54: 201-207.
Lindquist E.E., Krantz G.W., Walter D.E. 2009 — Order Mesostigmata — In: Krantz G.W., Walter D.E. (Eds). A manual of Acarology – Third edition. Texas: Texas Tech University Press p. 430-564.
Lohmann H. 1907 — Die Meeresmilben der Deutschen Südpolar-Expedition 1901-1903 — In: Drygalski E. (Ed). Deutsche Südpolar-Expedition 1901-1903. Berlin, Reimer Verlag. p. 361-413.
Luxton M. 1966 — Laboratory studies on the feeding habits of saltmarsh Acarina, with notes on their behavior — Acarologia, 8: 163-174.
Luxton M. 1967 — Two new mites from the rocky shore of North Auckland — New Zeal. J. Mar. Fresh. Res., 1: 76-87. doi:10.1080/00288330.1967.9515194 ![]()
Luxton M. 1984 — More marine littoral mites from New Zealand — New Zeal. J. Marine Freshw. Res., 18: 291-302. doi:10.1080/00288330.1984.9516051 ![]()
Luxton M. 1986 — A new species of Fortuynia (Acari: Cryptostigmata) from the marine littoral of Kenya — J. Nat. Hist., 20: 65-69. doi:10.1080/00222938600770061 ![]()
Luxton M. 1990a — Local distribution and habitat of mites (Acari) on the sea coast of New Zealand — J. Royal Soc. New Zeal., 20: 419-426. doi:10.1080/03036758.1990.10426720 ![]()
Luxton M. 1990b — The marine littoral mites of the New Zealand region — J. Royal Soc. New Zeal., 20: 367-418. doi:10.1080/03036758.1990.10426719 ![]()
Luxton M. 1992 — Oribatid mites from the marine littoral of Hong Kong (Acari: Cryptostigmata) — In: Morton B. (Ed.). The marine flora and fauna of Hong Kong and southern China III. Proceedings of the Fourth International marine Biological Workshop: The marine Flora and Fauna of Hong Kong and Southern China. Hong Kong: Hong Kong University Press. p. 211–227.
Märkel K. 1964 — Die Euphthiracaridae Jacot 1930 und ihre Gattungen (Acari, Oribatei) — Zoologische Verhandelingen, Leiden, 67, 3–78.
Marshall D.J., Convey P. 2004 — Latitudinal variation in habitat specifity of ameronothroid mites (Oribatida) — Exp. Appl. Acarol., 34: 21-35. doi:10.1023/B:APPA.0000044437.17333.82 ![]()
Marshall D.J., Gremmen N.J.M., Coetzee L., OConnor B.M., Pugh P.J.A., Theron P.D., Ueckermann E.A. 1999 — New records of Acari from the sub-Antarctic Prince Edward Islands — Polar Biol., 21: 84-89. doi:10.1007/s003000050338 ![]()
Marshall D.J., Pugh P.J.A. 2000 — Two new species of Schusteria (Acari: Oribatida: Ameronothroidea) from marine shores in southern Africa — Afr. Zool., 35: 201-205. doi:10.1080/15627020.2000.11657091 ![]()
Marshall D.J., Pugh P.J.A. 2002 — Fortuynia (Acari: Oribatida: Ameronothroidea) from the marine littoral of southern Africa — J. Nat. Hist, 36: 173-183. doi:10.1080/00222930010002775 ![]()
Messner B., Adis J., Franklin Ribeiro E. 1992 — A comparative study on plastron structures in mites (Acari) — Dtsch. Entomol. Z., 39: 159-176. doi:10.1002/mmnd.19920390124 ![]()
Michael A.D. 1882 — Further notes on British Oribatidae — J. Royal Microsc. Soc., London, 2: 1-18.
Michael A.D. 1888 — British Oribatidae. Vol. 2 — London: Ray Society. pp. 337-657.
Michael A.D. 1903 — Acarina (Oribatidae). Expedition Antarctique Belge. Resultats du Voyage du S.Y. Belgicae n 1897-1898-1899 — Rap. Sci. Zool., 10: 1-6.
Mortimer E., Jansen van Vuuren B., Lee J.E., Marshall D.J., Convey P., Chown S.L. 2011 — Mite dispersal among the Southern Ocean Islands and Antarctica before the last glacial maximum — Proc. Royal Soc. B, 278: 1247-1255. doi:10.1098/rspb.2010.1779 ![]()
Niedbała W. 1986 — Several species of moss-mites (Acari, Oribatida) from the Antarctic coastal zone — Pol. Polar Res., 7: 119-126.
Norton R.A.1994 — Evolutionary aspects of oribatid mite life histories and consequences for the origin of the Astigmata — In: Houck M. (Ed.). Mites. Ecological and evolutionary analyses of life history patterns. New York: Chapman and Hall. p. 99-135. doi:10.1007/978-1-4615-2389-5_5 ![]()
Norton R.A. and Alberti G. 1997 — Porose integumental organs of oribatid mites (Acari, Oribatida). 3. Evolutionary and ecological aspects — Zoologica, Stuttgart, 146, 115–143.
Norton R.A., Alberti G. and Woas S. 1997 — Porose integumental organs of oribatid mites (Acari, Oribatida). 1. Overview of types and distribution. — Zoologica, Stuttgart, 146, 1–31.
Norton R.A., Behan-Pelletier V.M. 2009 — Suborder Oribatida — In: Krantz G.W., Walter D.E. (Eds). A manual of Acarology – Third edition. Texas: Texas Tech University Press. p. 430-564.
Norton R.A., Bonamo P.M., Grierson J.D., Shear W.A. 1988 — Oribatid mite fossils from a terrestrial Devonian deposit near Gilboa, New York — J. Palaeontol., 62: 259-269. doi:10.1017/S0022336000029905 ![]()
Norton R.A., Graham T.B., Alberti G. 1997 — A rotifer-eating ameronothroid (Acari: Ameronothridae) mite from ephemeral pools on the Colorado Plateau — In: Mitchell R., Horn J., Needham G.R., Welbourn W.C. (Eds). Acarology IX, Proceedings of the IXth International Congress of Acarology. Columbus: Ohio Biological Survey. p. 539-542.
Norton R.A. and Sidorchuk E.A. 2014 — Collohmannia johnstoni n. sp. (Acari, Oribatida) from West Virginia (U.S.A.), including description of ontogeny, setal variation, notes on biology and systematics of Collohmanniidae — Acarologia, 54(3), 271–334.
Oudemans A.C. 1903 — Notes on the Acari. Fifth series — T. Entomol., 45: 123-150.
Peck S.B. 1994 — Sea-surface (pleuston) transport of insects between islands in the Galápagos Archipelago, Ecuador — Ann. Entomol. Soc. America, 87: 576-582. doi:10.1093/aesa/87.5.576 ![]()
Peckham V. 1967 — Studies of the Mite Alaskozetes antarcticus (Michael) — Antarct. J. U.S., 2: 196-197.
Pfingstl T. 2013a — Habitat use, feeding and reproductive traits of rocky-shore intertidal mites from Bermuda (Oribatida: Fortuyniidae and Selenoribatidae) — Acarologia, 53: 369-382. doi:10.1051/acarologia/20132101 ![]()
Pfingstl T. 2013b — Resistance to fresh and salt water in intertidal mites (Acari: Oribatida): implications for ecology and hydrochorous dispersal — Exp. Appl. Acarol., 61: 87-96. doi:10.1007/s10493-013-9681-y ![]()
Pfingstl T. 2013c — Revealing the diversity of a once small taxon: the genus Selenoribates (Acari, Oribatida, Selenoribatidae) — Zookeys, 312: 39-63. doi:10.3897/zookeys.312.5478 ![]()
Pfingstl T. 2013d — Thalassozetes barbara n. sp. (Acari, Oribatida), a new intertidal species from the coast of Barbados — Acarologia, 53: 417-424.
Pfingstl T. 2015a — An interesting case of sexual dimorphism in intertidal mites: Fortuynia dimorpha sp. nov. (Acari, Oribatida, Fortuyniidae) — Syst. Appl. Acarol., 20: 567-578. doi:10.11158/saa.20.5.10 ![]()
Pfingstl T. 2015b — Morphological diversity in Selenoribates (Acari, Oribatida): new species from coasts of the Red Sea and the Indo-Pacific — Int. J. Acarol., 41: 356-370. doi:10.1080/01647954.2015.1035321 ![]()
Pfingstl T. 2015c — The intertidal Fortuyniidae (Acari: Oribatida): new species, morphological diversity, ecology and biogeography — Zootaxa, 3957: 351¬-382.
Pfingstl T. 2016 — Marine associated mites (Acari: Oribatida: Ameronothroidea) of the African continent: biogeography, new species and distribution records — Syst. Appl. Acarol., 21: 320-334. doi:10.11158/saa.21.3.7 ![]()
Pfingstl T., Jagersbacher-Baumann J. 2016 — Indications of parthenogenesis and morphological differentiation in Hawaiian intertidal Fortuynia (Acari, Oribatida) populations — Zool. Anz., 260: 11-24. doi:10.1016/j.jcz.2015.11.004 ![]()
Pfingstl T., Krisper G. 2014 — Plastron respiration in marine intertidal oribatid mites (Acari, Fortuyniidae and Selenoribatidae) — Zoomorphology 133: 359-378. doi:10.1007/s00435-014-0228-5 ![]()
Pfingstl T., Schuster R. 2012a — Carinozetes nov. gen. (Acari: Oribatida) from Bermuda and remarks on the present status of the Family Selenoribatidae — Acarologia, 52: 377-409.
Pfingstl T., Schuster R. 2012b — First record of the littoral genus Alismobates (Acari: Oribatida) from the Atlantic ocean, with a redefinition of the family Fortuyniidae based on adult and juvenile morphology — Zootaxa, 3301: 1-33.
Pfingstl T., Schuster R. 2014 — Global distribution of the thalassobiontic Fortuyniidae and Selenoribatidae (Acari, Oribatida) — Soil Organisms, 86: 125-130.
Pfingstl T., Lienhard A., Jagersbacher-Baumann J. 2014 — Hidden in the mangrove forest: the cryptic intertidal mite Carinozetes mangrovi sp. nov. (Acari, Oribatida, Selenoribatidae) — Exp. Appl. Acarol., 63: 481-495. doi:10.1007/s10493-014-9802-2 ![]()
Pletzen R., Kok D.J. 1971 — Oribatei — In: Zinderen Bakker E.M., Winterbottom J.M., Dyer R.A. (Eds). Marion and Prince Edward Islands. Cape Town: A.A. Balkema. p. 314-326.
Procheş S. 2001 — Back to the sea: secondary marine organisms from a biogeographical perspective — Biol. J. Linn. Soc., 74: 197-203. doi:10.1111/j.1095-8312.2001.tb01386.x ![]()
Procheş S., Marshall D.J. 2001 — Global distribution patterns of non-halacarid marine intertidal mites: implications for their origins in marine habitats — J. Biogeogr., 28: 47-58. doi:10.1046/j.1365-2699.2001.00513.x ![]()
Pugh P.J.A. 1995 — Air-breathing littoral mites of sub-Antarctic South Georgia — J. Zool., London, 236: 649-666. doi:10.1111/j.1469-7998.1995.tb02737.x ![]()
Pugh P.J.A. 1997 — Acarine colonization of Antarctica and the islands of the Southern Ocean: the role of zoohoria — Polar Rec., 33: 113-122. doi:10.1017/S0032247400014431 ![]()
Pugh P.J.A., King P.E. 1986 — Activity rhythms in littoral Acari under laboratory conditions — J. Exp. Marine Biol. Ecol., 100: 37-45. doi:10.1016/0022-0981(86)90154-1 ![]()
Pugh P.J.A., MacAlister H.E. 1994 — Acari of the supralittoral zone on sub-Antarctic South Georgia — Pedobiologia, 38: 552-565.
Pugh P.J.A., Mercer R.D. 2001 — Littoral Acari of Marion Island: ecology and extreme wave action — Polar Biol., 24: 239-243. doi:10.1007/s003000000203 ![]()
Pugh P.J.A. 2003 — Have mites (Acarina:Arachnida) colonized Antarctica and the islands of the Southern Ocean via air currents? — Polar Record, 39: 239-244.
Pugh P.J.A., King P.E., Fordy M.R. 1987a — A comparison of the structure and function of the cerotegument in two new species of Cryptostigmata (Acarina) — J. Nat. Hist., 21: 603-616. doi:10.1080/00222938700770371 ![]()
Pugh P.J.A., King P.E., Fordy M.R. 1987b — Ambulacral structure in the terrestrial moiety of the intertidal Acari, and its relationship with the lifestyle of the Acari — Acarologia, 28: 3-13.
Pugh P.J.A., King P.E., Fordy M.R. 1990 — Respiration in Fortuynia maculata Luxton (Fortuyniidae: Cryptostigmata: Acarina) with particular reference to the role of van der Hammen's organ — J. Nat. Hist., 24: 1529-1547. doi:10.1080/00222939000770881 ![]()
Richters F. 1907 — Die Fauna der Moosrasen des Gaussberges und einiger südlicher Inseln — In: Drygalski E. (Ed). Deutsche Südpolar-Expedition 1901-1903. Berlin, Reimer Verlag. p. 259-302.
Schaefer I., Norton R.A., Scheu S., Maraun M. 2010 — Arthropod colonization of land – Linking molecules and fossils in oribatid mites (Acari, Oribatida) — M. Phylogenet. Evol., 57: 112-121. doi:10.1016/j.ympev.2010.04.015 ![]()
Schäffer S., Koblmüller S., Pfingstl T., Sturmbauer C., Krisper G. 2010 — Ancestral state reconstructions reveal multiple independent evolution of diagnostic morphological characters in the "Higher Oribatida" (Acari), conflicting current classification schemes — BMC Evol. Biol., 10: 246. doi:10.1186/1471-2148-10-246 ![]()
Schatz H. 1991 — Arrival and establishment of Acari on oceanic islands — In: Dusbábek F., Bukva V. (Eds) Modern Acarology Prague: Academia Prague and SPB Academic Publishing, The Hague, Vol. 2. p. 613-618.
Schatz H. 1998 — Oribatid mites of the Galápagos Islands-faunistics, ecology and speciation — Exp. Appl. Acarol., 22: 373-409. doi:10.1023/A:1006097928124 ![]()
Schubart H. 1970 — Ameronothrus schusteri n. sp., eine neue Oribatidae von der Küste Jugoslawiens (Arachnida: Acari) — Senckenberg. Biol., 51: 425-432.
Schubart H. 1971 — Vergleichende Morphologie und taxonomische Revision der Oribatidenfamilie Ameronothridae (Arachnida: Acari) mit Bemerkungen über jahreszeitliche Verschiebungen des Geschlechterverhältnisses [PhD Thesis] — Kiel: Christian-Albrechts-Universität. pp. 78.
Schubart H. 1975 — Morphologische Grundlagen für die Klärung der Verwandtschaftsbeziehungen innerhalb der Milbenfamilie Ameronothridae (Acari, Oribatei) — Zoologica, 123: 23-91.
Schulte G. 1973 — Vertikalwanderungen küstenbewohnender Milben (Acari, Oribatei) — Neth. J. Sea Res., 7: 68-80. doi:10.1016/0077-7579(73)90033-1 ![]()
Schulte G. 1975 — Holarktische Artareale der Ameronothridae (Acari, Oribatei) — Veröff. Inst. Meeresforsch. Bremerhaven, 15: 339-357.
Schulte G. 1976a — Gezeitenrhythmische Nahrungsaufnahme und Kotballenablage einer terrestrischen Milbe (Oribatei: Ameronothridae) im marinen Felslitoral — Marine Biol., 37: 265-277. doi:10.1007/BF00387612 ![]()
Schulte G. 1976b — Zur Nahrungsbiologie der terrestrischen und marinen Milbenfamilie Ameronothridae (Acari, Oribatei) — Pedobiologia, 16: 332-352.
Schulte G., Schuster R., Schubart H. 1975 — Zur Verbreitung und Ökologie der Ameronothriden (Acari, Oribatei) in terrestrischen, limnischen und marinen Lebensräumen — Veröff. Inst. Meeresforsch. Bremerhaven, 15: 359-385.
Schulte G., Weigmann G. 1977 — The evolution of the family Ameronothridae (Acari: Oribatei) II. Ecological aspects — Acarologia, 19: 167-173.
Schuster R. 1957 — Haloribatula tenareae nov. gen., nov. spec., eine neue Oribatide aus dem mediterranen Eulitoral (Acari) — Zool. Anz., 159: 121-127.
Schuster R. 1963 — Thalassozetes riparius n. gen., n. sp., eine litoralbewohnende Oribatide von bemerkenswerter morphologischer Variabilität (Acari-Oribatei) — Zool. Anz., 171: 391-403.
Schuster R. 1966 — Hornmilben (Oribatei) als Bewohner des marinen Litorals — Veröff. Inst. Meeresforsch. Bremerhaven, Sonderband II: 319-327.
Schuster R. 1977 — Die Selenoribatidae, Eine Thalassobionte Familie der Hornmilben — Acarologia, 19(1), 155–160.
Schuster R. 1979 — Soil mites in the marine environment — Recent Advances Acarol., 1: 593-602.
Schuster R. 1986 — Bodenbewohnende Milben (Acari) aus Niedersachsen und anderen Regionen Deutschlands (BRD) — Braunschw. Naturkundl. Schriften, 3: 519-525.
Schuster R. 1989 — Transoceanic distribution of air-breathing littoral mites — Progr. Acarol., 1: 355-362.
Sitnikova L.G. 1975 — The family Ameronothridae Balogh, 1961 — In: Ghilyarov M.S. (Ed). Key to soil inhabiting mites. Sarcoptiformes. Moscow: Nauka Press. p. 232-234.
Sømme L., Block W. 1984 — Ecophysiology of two intertidal mites at South Georgia — Oikos, 42: 276¬–282.
Søvik G. 2003 — Observations on ovoviviparity and mixed-parity mode in arctic populations of Ameronothrus lineatus (Acari, Oribatida) — Acarologia, 43: 393-398.
Søvik G. 2004 — The biology and life history of arctic populations of the littoral mite Ameronothrus lineatus (Acari, Oribatida) — Exp. Appl. Acarol., 34: 3-20. doi:10.1023/B:APPA.0000044436.80588.96 ![]()
Søvik G., Leinaas H.P., Ims R.A., Solhøy T. 2003 — Population dynamics and life history of the oribatid mite Ameronothrus lineatus (Acari, Oribatida) on the high arctic archipelago of Svalbard — Pedobiologia, 47: 257-271.
Strenzke K. 1961 — Selenoribates foveiventris n. gen., n. sp., aus der unterirdischen Feuchtzone der Küste des Roten Meeres (Acarina: Oribatei) — Kieler Meeresforsch., 17: 89-93.
Strong J. 1967 — Ecology of terrestrial arthropods at Palmer Station, Antarctic Peninsula — Antarct. Res. Ser. Washington, 10: 357-371.
Subías L.S. 2004 — Listado sistemático, sinonímico y biogeográphico de los ácaros oribátidos (Acariformes: Oribatida) del mundo — Graellsia, 60: 3-305. Internet update 2016, available from: ( http://escalera.bio.ucm.es/usuarios/bba/cont/docs/RO_1.pdf ![]() )
)
Syamjith P.K., Ramani N. 2013 — Association of a mangrove dwelling euphthiracarid mite, Acrotritia clavata (Märkel 1964) (Acari: Oribatida) with the epilithic alga, Microspora sp. — Int. J. Acarol., 39: 615-619. doi:10.1080/01647954.2013.862568 ![]()
Thorell T. 1871 — Om Arachnider fran Spetsbergen och Beeren-Eiland — Öfvers. Kongl. Vet. Akad. Förh., 28: 683-702.
Travé J. 1963 — Écologie et biologie des Oribates (Acariens) saxicoles et arboricoles — Vie et Milieu, suppl. 14: 1-286.
Travé J. 1986 — Les taenidies respiratoires des Oribates — Acarologia, 27: 85-94.
Trouessart E.L. 1912 — The Acariens of the Scottish national Antarctic Expedition — In: Bruce W.S. (Ed). Report on the scientific results of the voyage of S.Y. "Scotia" during the years 1902, 1903 and 1904. Edinburgh: The Scottish Oceanographical Laboratory. p. 81-86.
Vitzthum H.G. 1942 — Acarina. In: Braun H.G. (Ed.). Klassen und Ordnungen des Tierreichs. Leipzig: Akademische Verlagsgesellschaft Becker and Erler Kom. Ges. p. 1011.
Wallwork J.A. 1962 — A redescription of Notaspis antarcticus Michael, 1903 (Acari: Oribatei) — Pac. Insects, 4: 869-880.
Wallwork J.A. 1963 — The Oribatei (Acari) of Macquarie Island — Pac. Insects, 5: 721-769.
Wallwork J.A. 1964 — A revision of the family Podacaridae Grandj. (Acari: Oribatei) — Acarologia, 6: 387-399.
Wallwork J.A. 1966 — More Oribatid mites (Acari: Cryptostigmata) from Campbell I. — Pac. Insects, 8: 849-877.
Wallwork J.A. 1967 — Cryptostigmata (Oribatid mites) — Entomol. Antarct., 10: 105-122. doi:10.1029/ar010p0105 ![]()
Wallwork J.A. 1970 — Acarina: Cryptostigmata of South Georgia — Pac. Insects Monogr., 23: 161-178.
Wallwork J.A. 1981 — A new aquatic oribatid mite from western Australia (Acari: Cryptostigmata: Ameronothridae) — Acarologia, 22: 333-339.
Walter D.E., Lindquist E.E., Smith I.M. 2009 — Order Trombidiformes — In: Krantz G.W., Walter D.E. (Eds) A manual of Acarology – Third edition. Texas: Texas Tech University Press. p. 430-564.
Weigmann G. 1975a — Labor- und Freilanduntersuchungen zur Generationsdauer von Oribatiden (Acari: Oribatei) — Pedobiologia, 15: 133-148.
Weigmann G. 1975b — Vorkommen von Ameronothrus (Acari, Oribatei) im Litoral Südafrikas — Veröff. Inst. Meeresforsch. Bremerhaven, 15: 65-67.
Weigmann G., Schulte G. 1975 — Ameronothrus schubarti n.sp. aus dem marinen Litoral Kaliforniens — Senckenberg. Biol., 56: 133-143.
Weigmann G., Schulte G. 1977 — The evolution of the family Ameronothridae (Acari: Oribatei) I. Morphological aspects — Acarologia, 19: 161-166.
Willmann C. 1931 — Moosmilben oder Oribatiden (Cryptostigmata) — In: Dahl F. (Ed). Die Tierwelt Deutschlands. Band 22. Jena: Fischer Jena. p. 79-200.
Willmann C. 1936 — Oribatiden von Bonaire und Curaçao — Zool. Jb. Syst., 67: 429-441.
Woas S. 2002 — Acari: Oribatida — In: Adis J. (Ed). Amazonian Arachnida and Myriapoda. Sofia-Moscow: Pensoft Publishers. p. 21-291.
Woodring J.P. 1973 — Comparative morphology, functions and homologies of the coxal glands in oribatid mites — J. Morph., 139: 407-430. doi:10.1002/jmor.1051390404 ![]()
Worland M.R., Convey P. 2001 — Rapid cold hardening in Antarctic microarthropods — Funct. Ecol., 15: 515-524. doi:10.1046/j.0269-8463.2001.00547.x ![]()
Worland M.R., Lukešová A. 2000 — The effect of feeding on specific soil algae on the cold-hardiness of two Antarctic micro-arthropods (Alaskozetes antarcticus and Cryptopygus antarcticus) — Polar Biol., 23: 766-774. doi:10.1007/s003000000150 ![]()



2016-12-14
Date accepted:
2017-03-21
Date published:
2017-05-05
Edited by:
Sidorchuk, Ekaterina

This work is licensed under a Creative Commons Attribution 4.0 International License
2017 Pfingstl, Tobias
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)