Rediscovery of the water mite Atractides circumcinctus Schwoerbel, 1956 (Acari: Hydrachnidia) in the Black Forest National Park
Gerecke, Reinhard1
1University of Tübingen, Department of Evolution and Ecology, Auf der Morgenstelle 28E, D-72076 Tübingen, Germany.
2Arachnology, Senckenberg, Senckenberganlage 25, D-60325 Frankfurt, Germany.
2017 - Volume: 57 Issue: 3 pages: 665-671
https://doi.org/10.24349/acarologia/20174186ZooBank LSID: FCAED8A2-D48F-422C-AB4E-5CE6FE99895A
Keywords
Abstract
The water mite species Atractides (s. str.) circumcinctus Schwoerbel, 1956 was first described from a single male (Schwoerbel 1956a) from a helocrenic spring at about 1000 m asl. on Mt Schauinsland (Central Black Forest, district Freiburg). Morphological information on more specimens, including the first description of the female, was given by Schwoerbel (1958) who added further locality records: springs in the valley of the Haslach stream, the upper course of the Bohrerbach, and the Sauterquelle spring at Mt Feldberg. All records come from the Central Black Forest in the district of Freiburg, but only the latter site can be precisely localized nowadays. Since then, this species was not found again and the type material is in an unsatisfactory state of conservation (Gerecke 2003).
Since the establishment of a National Park in the northern part of the Black Forest ( http://www.schwarzwald-nationalpark.de/nationalpark/ ![]() ), authorities are interested in observing the environmental change expected under nature protection conditions. Of particular interest is the documentation of the status quo of the limnology of the area, under hydrogeological, floristic and faunistic aspects, with a particular focus on spring habitats. In the course of monitoring 21 springs draining to the Schönmünz stream (Murg catchment), the rare water mite Atractides circumcinctus was discovered at two sites. The aim of this paper is to give a redescription of the species, to contribute to our understanding of the variability of its diagnostic features, and to distinguish it from the most similar species in the genus.
), authorities are interested in observing the environmental change expected under nature protection conditions. Of particular interest is the documentation of the status quo of the limnology of the area, under hydrogeological, floristic and faunistic aspects, with a particular focus on spring habitats. In the course of monitoring 21 springs draining to the Schönmünz stream (Murg catchment), the rare water mite Atractides circumcinctus was discovered at two sites. The aim of this paper is to give a redescription of the species, to contribute to our understanding of the variability of its diagnostic features, and to distinguish it from the most similar species in the genus.
The sampling methods during the collections made in 2015 followed the standard proposed by Franz et al. (2006). In order to collect a sufficient number of specimens and to get more insight into habitat preference, on 25.08.2016 additional sampling was done in two sectors of spring 804 (eucrenal = spring mouth area and hypocrenal = 100 m downstream), dedicated mostly to the extraction of water mites. All material was sorted in the laboratory from fresh sediment samples and conserved in 99 % undenaturated ethanol. Specimens selected for microscopical investigation were slide-mounted in glycerine jelly (material collected in 2015) or Hoyer's fluid (material collected in 2016), one specimen from site D 804 was sequenced in the AIM laboratory (Advanced Identification Methods GmbH, Munich); sequences and trace files can be found within the Barcode of Life Database (BOLD – HYPERLINK " http://www.boldsystems.org ![]() ") under the processID AIMEG006-17.
") under the processID AIMEG006-17.
Measurements are given in μm.
Abbreviations (for further explanations see Gerecke 2003, Figs 2-8): I-IV-L-1-6 – Legs I-IV, segments 1-6; Ac-1-3 – acetabula 1-3; Cx-I-IV – coxae I-IV; D-1-4 – dorsalia 1-4 (dorsal muscle attachment platelets); Dgl-1-5 – dorsoglandularia 1-5; dL – dorsal length; H – height; HA – proximal height; HB – central height; HC – distal height; L – length; lL – lateral length; mL – median length; P-1-5 – palp segments 1-5; S-1/-2 – proximal/distal sword seta on I-L-5; V-1-3 – ventralia 1-3 (ventrocaudal muscle attachment platelets); Vgl-1-3 – ventroglandularia 1-3; vL – ventral length; W – width.
Material examined — Black Forest National Park, Schönmünzach valley: Site D 804, S Großbrunnen, spring at lower forest road, 710 m, 8°17'46.38"E, 48°34'5.69"N; 01.07.2015, eucrenal, 2 ♀♀, slide-mounted; 25.08.2016, eucrenal, 6 ♂♂, 7 ♀♀, 2 deutonymphs (4 ♂♂, 4 ♀♀, 3 deutonymphs slide-mounted, 2 ♂♂, 3 ♀♀ in ethanol); hypocrenal, 1 ♀, in ethanol. Site D 821, spring of upper affluent to Stumpenteich stream, 740 m, 8°19'44.97"E, 48°34'32.55"N; 25.09.2015, eucrenal, 1 ♀, slide-mounted.
Senckenberg, collection Schwoerbel: Holotype ♂, "Hydrachnellae 457 Pionae Atractidae Atractides; A. circumcinctus TYPUS ♂ ADA6–Quelle am Schauinsland, 1000 m, 27"; "Sauterquelle Feldberg/Schw., Okt., 1960", 1 ♂; "Schwarzwald 1077", 1 ♂; "Hydrachnellae 294 Pionae Atractidae Atractides; A. circumcinctus P.Typus ♀. Bohrerbach, Oberlauf, in Moosen Freiburg, Jan, 1954", 1 ♀; "Quelltümpel Haslachtal 20.3.1957, 1174", 1 ♀; "Schwarzwald 1081", 4 ♀♀ on one slide; "Schwarzwald 1067", 1 ♀; three slides without locality information, marked "paratypoid", numbered "786", "797", "801", each bearing 1 ♀.
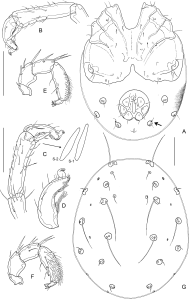
Redescription — Both sexes: Colour of sclerotized parts greyish brown, idiosoma with extended red areas. Integument with a very fine striation, dorsal and posteroventral muscle insertions unsclerotized, glandularia small, round (maximum diameter of Dgl-3, 20-23), setae of Dgl-1,-3, and -4, and Lgl-2 very long, all others short (Fig. 1G); coxal field with strong sexual dimorphism; apodemes of Cx-II directed laterally; Legs without sexual dimorphism, claws with claw blade and ventral clawlet; I-L-5/6: Figs 1B-D; I-L-5 extremely elongate, in the centre slightly narrowed and here with a ventral seta barely differing from the surrounding ones, dorsal and ventral margin diverging only towards distal segment end; setae S-1 and -2 close to each other (separation < maximum W S-2), rather stout and apically rounded; S-1 slightly shorter and more slender than S-2; I-L-6 (Fig. 1D) strongly shortened and curved, equally narrowed from base to tip. Genital field with subtriangular acetabula in a weakly curved line. Excretory pore smooth, Vgl-1/2 generally separate, but often very close (Fig. 1A, arrow), occasionally fused. Gnathosoma without particularly projecting rostrum, palp with slight sexual dimorphism in proportions; P-2 ventral margin without extensions, weakly concave in basal part, distally forming an obtuse angle; P-4 maximum H near proximoventral seta, sword seta insertion between ventral seta insertions, in centre of segment or slightly shifted to distal part; sectors between ventral setae nearly equal in length or distal sector slightly shortened.
Males (n = 4; palp and I-L, n = 3; gnathosoma and chelicera, n = 1) — Idiosoma L/W 440-510/240-400, venter: Fig. 1A; coxal field L/W 270-290/350-360, fused to form an extended shield including also Vgl-3 and V-1 (visible as a finely dotted area); posterior margin of coxal shield medially deeply indented, in lateral parts equally rounded; Cx-I+II mL 105-130, lL 184-210, W 233-250. I-L-5/6: Fig. 1C; I-L-5 dL/vL 141-146/123-124, ratio 1.15-1.19; HA 29-32, HB 38-40, HC 40-41, ratio dL/HB 3.53-3.76; S-1 L/W 50-53/7-8, ratio 6.5-7.6; S-2 L/W 55-56/9-11, 5.1-6.1; separation S-1-2, 6-7, L ratio S-1/-2, 0.91-0.95; I-L-6 dL 75-77, HA 25-26, HB 18, HC 16; ratio dL/HB 4.2-4.4; dL ratio I-L-5/6, 1.8-1.9. Genital field as long as wide, L/W 106-112/110-116, anterior margin with projecting border of secondary sclerotization, posterior margin indented, gonopore L 45-54, flanked by a line of 7-8 fine setae, 3-5 further setae scattered at the lateral and posterior plate margins; maximum diameter Ac-1, 29-34; Ac-2, 31-39; Ac-3, 29-30. Gnathosoma L 80; chelicera L/H 148/31 (ratio 4.8), L ratio basal segment/claw 2.3. Palp: Figs 1E-F; total L 242-255; L/H (ratio, rel. L [%]) P-1, 25-26/20-21 (1.2-1.3, 10-11); P-2, 52-55/38-39 (1.4, 21-22); P-3, 52-53/30-34 (1.6-1.7, 21); P-4, 88-94/30-33 (2.9, 36-37); P-5, 24-27/10-11 (2.4-2.5, 10-11); L ratio P-2/P-3, 1.00-1.04; P-2/P-4, 0.59-0.60; P-3/P-4, 0.56-0.59; P-4 with dense cover of fine, hair-like setae.


Females (n = 7, gnathosoma and chelicera n = 1) — Idiosoma L/W 560-630/380-500, venter: Fig. 2A; coxal field L/W 250-305/370-420, coxae in 3 groups, with a narrow border of secondary sclerotization, posteromedial margin of Cx-I+II broadly rounded, posterior margin of Cx-IV weakly convex to nearly straight, perpendicular to the longitudinal axis; Cx-I+II mL 110-120, lL 195-225, W 240-274. I-L-5 dL/vL 168-181/139-161, ratio 1.1-1.2; HA 35-40, HB 44-46, HC 45-47, ratio dL/HB 3.7-4.0; S-1 L/W 55-61/6-8, ratio 7.4-10.2; S-2 L/W 59-63/10-12, ratio 5.0-6.1; separation S-1-2, 6-7, L ratio S-1/-2, 0.93-1.03; I-L-6 dL 77-86, HA 25-29, HB 18-21, HC 16-17; ratio dL/HB 4.1-4.5; dL ratio I-L-5/6, 2.02-2.35. Genital field L/W 162-193/180-210, pre- and postgenital sclerites strong, genital plates L/W 100-120/38-49, weakly curved, anteriorly pointed, posteriorly rounded, bearing about 10 pairs of fine, hair-like setae; maximum diameter Ac-1, 36-45; Ac-2, 34-42; Ac-3, 31-35. Gnathosoma L 108; chelicera L/H 177/38 (ratio 4.7), L ratio basal segment/claw 2.4. Palp: Fig. 2B; total L 247-295; L/H (ratio, rel. L [%]) P-1, 27-29/21-23 (1.2-1.4, 10-11); P-2, 53-66/32-41 (1.5-1.7, 21-23); P-3, 56-71/29-35 (1.9-2.2, 23-24); P-4, 86-99/27-30 (3.1-3.5, 33-35); P-5, 25-30/10-11 (2.5-3.0, 10-11); L ratio P-2/P-3, 0.90-0.98: P-2/P-4, 0.62-0.70; P-3/P-4, 0.65-0.72. As often in Atractides, female palp more slender than in males (L/H in males P-2 < 1.45, P-3 < 1.9, P-4 < 3.0), with a less dense cover of fine setae. Furthermore, P-3 relatively longer (in males, L ratio P-2/P-3 ≥1.0, P-3/P-4 < 0.6).
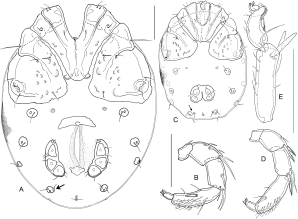


Deutonymph (n= 3; palp: n = 1) — Idiosoma L/W 300-370/220-300, venter: Fig. 2C; coxal field L/W 155-170/205-240, coxae in 3 groups, Cx-I+II mL 40-55, lL 100-115, W 145-160. I-L-5/6: Fig. 2E; I-L-5 dL/vL 86-89/75-78, ratio 1.1; HA 23, HB 26-29, HC 27-29, ratio dL/HB 3.1-3.3; S-1 L/W 34-35/5-6, ratio 5.8-6.8; S-2 L/W 38-40/6-7, ratio 5.7-6.3; distance S-1-2, 3, L ratio S-1/-2, 0.9; I-L-6 dL 53-54, HA 21, HB 15, HC 12; ratio dL/HB 3.5-3.6; dL ratio I-L-5/6, 1.6-1.7. Genital field L/W 45/66-67; maximum diameter Ac-1, 15-19; Ac-2, 18. Gnathosoma L 63-67; Palp: Fig. 2D; total L 155; L/H (ratio, rel. L [%]) P-1, 16/12 (1.3, 10); P-2, 35/22 (1.6, 23); P-3, 33/18 (1.8, 21); P-4, 53/17 (3.1, 34); P-5, 18/8 (2.3, 12); L ratio P-2/P-3, 1.06: P-2/P-4, 0.66; P-3/P-4, 0.62. As typical for deutonymphs, many setae are reduced and the genital field bears only two pairs of Ac. Platelets Vgl-1/2 are distinctly separate in all specimens. Proportions of L-I-5/6 are less extreme than in adults, but I-L-6 is shortened and typical in shape.
In important morphological features and proportions of sclerotized parts and appendages, the specimens from the Black Forest National Park agree well with the holotype and other specimens recorded by Schwoerbel (1958) from springs located in more southern parts of this mountain range. An analysis of the newly detected populations allows the refinement of the definition of this rare species, and contributes to a better understanding of its morphological variability.
Females from coll. Schwoerbel lie within the variability of the populations from the National Park in nearly all measurements. The holotype male, unfortunately in a bad state and with several important details difficult to evaluate, is generally a little larger in size, but the other males in coll. Schwoerbel lie within the variability range of the National Park specimens. A good agreement is found in important proportions.
Our new data allows to complete the combination of diagnostic character states of A. circumcinctus as follows: (1) integument smooth, with a very fine lineation; (2) glandularia not particularly enlarged, diameter about 20; (3) coxal field with sexual dimorphism, in males fused into a coxal plate including also Vgl-3, in females in three parts, Vgl-3 separate; (3) I-L with characteristic proportions of distal segments (I-L-5 extremely elongated, I-L-6 very short, L ratio 1.8-1.9 in males, > 2.0 in females); (4) I-L-5 in central part slightly narrowed, ventral seta in this sector not thickened; (5) setae S-1/-2 thickened, apically rounded, close to each other; (6) I-L-6 proximally strongly thickened, dorsal and ventral margins distally strongly converging; (7) genital field with relatively small Ac (maximum diameter Ac-3, 39 in males, 45 in females) lying in a weakly curved line; (8) anal pore smooth; (9) Vgl-1/-2 in general separate, but located very close and in oblique view often appearing to be fused, occasionally fused; (10) palp except for the more dense dorsal setation of P-4 without sexual dimorphism, the latter segment rather stout P-4.
In general, fusion or separation of the posteroventral setal platelets Vgl-1/-2 is an important character for the distinction of Atractides species. However, Gerecke (2003) reports for A. fonticolus (K. Viets, 1920) a sexual dimorphism (Vgl-2 fused in males, but separate in females), and fused Vgl-1+2 can occasionally be observed as individual aberrations also in species reported to have these platelets separate (Gerecke 2014). The observations on the populations presented here and a revision of the old collection material shows that A. circumcinctus is a particular case of intraspecific variability of this character state. As this phenomenon was overlooked so far, only specimens with fused Vgl-1+2 can be correctly identified using the keys published in Gerecke (2003) and Gerecke et al. (2016).
Instead, the most distinctive character state of the species concerns the extreme proportions of the terminal segments of I-L: Values of the I-L-5/6 L ratio as high as described above are not known in any other European Atractides species. On the base of this character, male A. circumcinctus should key out in Gerecke et al. (2016) at couplet 10 ("length ratio I-L-5/6 ≥ 1.8" – remaining species: "length ratio I-L-5/6 < 1.8"), and females at couplet 78 ("length ratio I-L-5/6 > 2.0" – remaining species: "length ratio I-L-5/6 < 2.0").
In the European fauna, the most similar species (combining a finely striated integument with a relatively long I-L-5 and relatively short I-L-6, ratio 1.6-1.7) is A. vaginalis (Koenike, 1905). Both species have in common also the shape of I-L-6 (basally thick, distally strongly narrowed), but A. vaginalis differs in the triangular arrangement of the generally larger acetabula (maximum diameter often > 45), absence of a projecting anterior margin of the male genital field, smaller glandularia (maximum diameter < 20) and the presence of a distinctly thickened ventral seta on I-L-5. Furthermore, the data given here show that P-4 of A. vaginalis is generally more slender (L/H in male 3.2, in female 3.5).
Further European species with a rather high I-L-5/6 ratio (for details see Gerecke 2003) are Atractides fonticolus (K. Viets, 1920), A. pennatus (K. Viets, 1920), A. legeri (Motaș, 1927) and A. angustiporus (K. Viets, 1930). The former three form a distinctive species group, differing from both A. vaginalis and A. circumcinctus in a lineated integument and the sexual dimorphism in the shape of P-2 (with a ventral projection in males). Atractides angustiporus, only known in the female sex from two populations in the Pyrenees, is in need of redescription. Major differences from A. circumcinctus are a wider separation of setae S-1/2, the shape of I-L-6 (enlarged, not narrowed, in distal part), and acetabula in triangular arrangement as in A. vaginalis.
Habitat and Biology – Except for the record from the upper course of the Bohrerbach, all known records of Atractides circumcinctus come from spring habitats, mostly (rheo)helocrenes. Documents in the Schwoerbel archive (Gerecke unpubl.) suggest that he had a rather strict viewpoint concerning the longitudinal zonation in lowest order streams, assigning to the spring stream also sectors at rather low distance (about 50 m) from the spring mouth. The new data support this interpretation of the habitat preference: In 25.08.2016, only one specimen was found in the hypocrenal sector of spring D 804, the remaining 15 in the swampy area surrounding the spring mouth, in summer densely overgrown by Chrysosplenium. Here, the species was part of a rich water mite coenosis with 14 other species, among which Hydrovolzia placophora (Thor, 1901) was the dominant.
Nothing is known about the life cycle of Atractides circumcinctus. Living specimens attract attention by their fast movements, being much more hasty then other spring-typical Atractides species.
The specimens described here were detected in the course of a pioneer project for long term monitoring selected springs in the Schönmünz valley, financed and organized by the Black Forest National Park. Terry Gledhill (FB; Ambleside) corrected the language and contents of a former draft of this paper. Jérôme Morinière (Advanced Identification Methods – AIM GmbH), Munich, is acknowledged for sequencing and genetical analysis of one of the investigated specimens.
Franz H., Gerecke R., Stur E., Wiedenbrug S. 2006 — Vorschläge für die langfristige Umweltbeobachtung, Ausblick. — In: Gerecke R., Franz H. (eds): Quellen im Nationalpark Berchtesgaden. Lebensgemeinschaften als Indikatoren des Klimawandels. Nationalpark Berchtesgaden, Forschungsbericht 51 pp. 255-263
Gerecke R. 2003 — The water mites of the genus Atractides (Acari: Parasitengona: Hygrobatidae) in the W palaearctic region. A revision. — J. Linn. Soc. London 138, 141-376. doi:10.1046/j.1096-3642.06-0.00051.x ![]()
Gerecke R. 2014 — The water mites of the genus Atractides Koch, 1837 (Acari, Hydrachnidia: Hygrobatidae) in Corsica and Sardinia. — Zoosystema 36(4), 735-759. doi:10.5252/z2014n4a3 ![]()
Gerecke R., Gledhill T., Pesic V., Smit H. 2016 — Acari: Hydrachnidia III. — In: Gerecke R. (ed.) Süßwasserfauna von Mitteleuropa 7/2-3: Chelicerata: Acari III. Springer Spektrum Elsevier. 1-429.
Schwoerbel J. 1956 — Über seltene und neue Milben aus dem südlichen Schwarzwald (Hydrachnellae, Acari). — Arch. Hydrobiol., Suppl. 24. 3(1), 67-85.
Schwoerbel J. 1958 — Zur Kenntnis der Wassermilbenfauna des südlichen Schwarzwaldes (Hydrachnellae, Acari). 4. Beitrag. — Mitt. Bad. Landesver. Naturkde. Naturschutz (N.F.) 7(2), 133-144.



2016-12-04
Date accepted:
2017-01-27
Date published:
2017-05-23
Edited by:
Kreiter, Serge

This work is licensed under a Creative Commons Attribution 4.0 International License
2017 Gerecke, Reinhard
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



