Synonymy of the water mite families Malgasacaridae and Zelandothyadidae (Acari: Hydrachnidia) reveals an interesting disjunct distribution in the Southern Hemisphere
Gerecke, Reinhard1 ; Judson, Mark L.I.2 and Cook, David R.3
1✉ Institut de Systématique, Évolution, Biodiversité (ISYEB), UMR 7205 CNRS, MNHN, UPMC, EPHE, Muséum national d’Histoire naturelle, Sorbonne Universités, 57 rue Cuvier, CP 53, 75005 Paris, France. University of Tübingen, Department of Evolution and Ecology, Auf der Morgenstelle 28E, D-72076 Tübingen, Germany (current address).
2Institut de Systématique, Évolution, Biodiversité (ISYEB), UMR 7205 CNRS, MNHN, UPMC, EPHE, Muséum national d’Histoire naturelle, Sorbonne Universités, 57 rue Cuvier, CP 53, 75005 Paris, France.
37725 N. Foothill Drive S., Paradise Valley, Arizona, 85253, U.S.A.
2017 - Volume: 57 Issue: 3 pages: 617-631
https://doi.org/10.24349/acarologia/20174181ZooBank LSID: 8FD4E52A-AB6C-44B6-8ADF-8F91C46DE5F8
Keywords
Abstract
The family Zelandothyadidae Cook, 1983 was originally proposed as a subfamily without attribution to a family, due to uncertainties concerning its relationships. It was subsequently elevated to family rank, without discussion, in a key published by Harvey & Growns (1998). Shortly thereafter, Harvey (1998) provided justification for this change in rank and transferred to Zelandothyadidae the subfamily Australiothyadinae Cook, 1986, comprising the single species Australiothyas swaini Cook, 1986, from Tasmania. In the same work, Harvey (1998) tentatively placed Zelandothyadidae in the Hydryphantoidea. Obviously, Zelandothyadidae is a family of enigmatic phylogenetic relationships, with the attributed genera combining character states previously considered diagnostic for either the superfamily Eylaoidea or the superfamily Hydryphantoidea (Cook 1983; Harvey 1998).
The family Malgasacaridae Tuzovskij, Gerecke & Goldschmidt, 2008 was proposed for the monotypic genus Malgasacarus Tuzovskij et al., 2008. The similarities between Malgasacarus rarus Tuzovskij et al., 2008, described from a forest area in eastern Madagascar, and the two species of Zelandothyas, Z. diamphida Cook, 1983 and Z. hyporheica Smit, 1996, both from the South Island of New Zealand, prompted us to carry out a broader investigation of these genera, based on (1) examination of a new species of Zelandothyas, (2) the description of the previously unknown larval and deutonymphal stages of Zelandothyas diamphida and (3) a revision of Malgasacarus rarus.
The following abbreviations are used in the descriptions: Cx-I (etc.) = first coxae (etc.); H = height; L = length; I-L = first leg; P-1 (etc.) = palp, first segment (etc.); W = width. Measurements are given in μm.
Malgasacaridae Tuzovskij et al. 2008: 163-164. New subjective synonymy.
Diagnosis — Adults (deutonymphs and larvae unknown for Australiothyadinae): Palp with P-1-4 rather stout, P-4 with two heavy dorsodistal setae; P-5 slender, finger-like, with apical setae little modified, not claw-like.
Discussion — In contrast to Cook (1986), who preferred to keep Australiothyas and Zelandothyas in two unassigned subfamilies, Harvey (1998) considered them to represent sister-groups and placed the monotypic subfamilies in Zelandothyadidae. Instead of giving a diagnosis for this newly defined taxon, he made reference to "substantial resemblances" between the two taxa noted by Cook (1986) in the description of Australiothyas swaini. If we analyse the situation in detail, the only common character states are found in the palp, as given above in the diagnosis. Transitional forms in several basal hydryphantid genera (Cook 1974) show that the “chelate” hydryphantoid palp (with pointed dorsodistal extension of P-4) developed from the stable fusion of a dorsodistal seta to the segment surface. The situation found in Zelandothyadidae, with two strong dorsodistal setae on P-4, could represent an early evolutionary stage from which a chelate palp developed.
A further common feature of Zelandothyadinae and Australiothyadinae is the presence of a mix of morphological conditions that does not allow their attribution to any of the known superfamilies. However, this gives no significant information for a taxonomic assignment: where Australiothyas is eylaoid-like, Zelandothyas is hydryphantoid-like, and vice versa. Data from the original description and a revision of the holotype and paratype of Australiothyas swaini (Museum Victoria, slides K 387–388) show that Australiothyadinae differ from Zelandothyadinae in many important respects: (1) Idiosoma covered by well-developed dorsal and ventral shields separated by a narrow dorsal furrow; (2) glandular pores round, not slit-shaped; (3) coxae bearing very few setae; (4) Cx-I medially fused; (5) leg setation reduced, not forming a continuous hypertrichous cover of segments; (6) leg claws simple; (7) genital field with unstalked acetabula; (8) mouth opening associated with a membranous ring; (9) gnathosoma without infracapitular teeth; (10) cheliceral claws small.
Of particular significance are the shape of glandular pores, the leg setation and the form of the mouthparts, indicating completely different modes of food uptake. Nevertheless, we follow the classification proposed by Harvey (1998) until it can be replaced by a better arrangement.The synonymy of Malgasacaridae with Zelandothyadidae is discussed below.
Diagnosis — Adults (only known for Zelandothyas; some features extrapolated from deutonymphs of Malgasacarus): With integument soft, papillate, papillae channelled, appearing stellate in apical view. Glandular pores slit-shaped. Lateral eyes with dark red pigmentation, near anterior idiosomal margin, generally on paired sclerotized bars orientated at right angles to longitudinal axis of body. Anterior dorsum usually with a frontal plate, variously composed of trabecular structures, surrounding a central dorsal projection bearing post-ocular setae; anterior part of this plate may be detached to form a separate, inverse-Y-shaped sclerite (lateral eye sclerites and frontal shield lacking in a juvenile female of Zelandothyas balloti sp. nov.). Frontal eye unpigmented, lying on sclerite between postocularia or reduced (possibly remaining in Zelandothyas as an indistinct structure in membranous area anterior to postocularia). Idiosomal chaetome formula (following Tuzovskij 1987): 2-2-4-4-6-4-4-4-2-4-0. Cx-II separate from Cx-III, not meeting in midline, all coxae bearing fine setae in dense rows, secondary sclerotization forming extensive associated apodemes, especially on posterior pairs. At least three pairs of acetabula (Malgasacarus probably polyacetabulate), these round or oval in shape and slightly stalked. Legs with numerous, fine, hair-like setae that are little differentiated in size and shape, without swimming setae; claws with numerous, fine, lateral clawlets in two rows, antiaxial row shorter than paraxial one. Mouthparts protrusible (gnathosoma attached to a tube of soft integument); basal cheliceral segments fused medially from tip to cheliceral furrow, cheliceral claws long and slender, flexible; palp neither chelate nor uncate, P-4 with two heavy dorsodistal setae, P-5 slender, with several small, barely modified terminal setae.
Deutonymph: Dorsal sclerites and coxal apodemes present (Malgasacarus) or absent (Zelandothyas). Idiosomal chaetome as in adults. Numbers of setae on coxal plates and appendages lower than in adults. Genital field lacking genital flaps and gonopore, consisting of two to three pairs of round, shortly-stalked acetabula on a short, longitudinal sclerite bearing two to four setae in lateral or anterior position.
Larva: Only known for Zelandothyas (see description below).
Discussion — The family Malgasacaridae was proposed in a rather precipitous manner by Tuzovskij et al. (2008), without proper consultation with the junior authors of that paper. Based on a re-examination of the holotype of Malgasacarus rarus and the study of a second specimen, important new morphological details are provided here, allowing a better understanding of its systematic position. Zelandothyas and Malgasacarus are evidently closely related and the differences between them (see discussion below, under Malgasacarus) do not warrant separation at the familial or even subfamilial level. Although Malgasacarus remains incompletely known, we see no reason to adopt separate monotypic subfamilies for these two genera and therefore place Malgasacarus in Zelandothyadinae.
In order to accommodate Malgasacarus, the diagnosis of Zelandothyadinae is here modified from Cook’s (1983) definition. Potential synapomorphies are: (1) integumental papillae with canalization (appearing star-shaped in apical view); (2) lateral eye capsules attached to transverse, rod-shaped plates; (3) frontal shield built up from trabecular sclerite structures including postocularia; (4) glandular pores slit-shaped; (5) long apodemes associated with coxal plates (posteromedially and laterally at Cx-II, anteriorly at Cx-III, posteriorly at Cx-IV); (6) leg claws with lateral clawlets arranged in a short antiaxial and a long paraxial row; (7) distal hypostome with a group of hooked, ventrally-directed teeth; (8) basal segments of chelicerae medially fused; (9) cheliceral claw stylet-like, but weakly sclerotized and flexible; (10) P-4 with two strong mediodistal setae.
The following character states of postlarval Zelandothyadinae are probably plesiomorphic: (1) all glandularia free in soft integument; (2) frontal eye developed (uncertain in Zelandothyas); (3) coxae in four groups; (4) leg setation little differentiated; (5) genital field with well-developed flaps and three pairs of acetabula (ascertained only in Zelandothyas); (6) excretory pore unsclerotized; (7) palp simple, neither uncate nor chelate, with terminal setae on P-5 little modified.
The larva of Zelandothyas (described below) fits the diagnosis of the superfamily Hydryphantoidea given by Smith & Cook (2001) in terms of the following character states: (1) legs with 6 movable segments; (2) dorsum without sclerotized plates; (3) lateral eyes not on a common plate; (4) leg tarsi with unmodified claws, empodium present; (5) humeral setae (Oe) simple; (6) one pair of acetabula at anterior margin of Cx-II; (7) P-5 cylindrical, thumb-like, inserting at a distance from the tip of P-4. However, most of these character states are plesiomorphic and Hydryphantoidea are probably a para- or polyphyletic group (Cook 1974; see also molecular evidence given by Dabert et al. 2016). The following larval characters justify the family rank of Zelandothyadidae: (1) posteroventral area bearing setae E2 (Ai) while setae E1 (Ae) are reduced – a situation not reported for any hydryphantoid family, but known from the phylogenetically distant Hydrachnoidea; (2) strong development of setae Ve, otherwise known only from representatives of Wandesiinae (Hydryphantidae), these setae being reduced in all other water mites; (3) presence of a frontal eye and slender solenidia in proximal part of leg tarsi, otherwise only known in Protziinae (Hydryphantidae); (4) legs without trichobothria, as opposed to their presence on the telofemora in all known larvae of Hydryphantidae.
The phylogenetic relationships between Zelandothyadidae and other water mite families remain uncertain. Some of the above-mentioned character states of postlarval stages (shape of leg claws, modification of cheliceral claws and development of hypostomal teeth) are highly derivative. The flexible cheliceral claws obviously cannot be used for piercing sclerotized prey, which is in stark contrast to the strongly hardened claws found in eylaoid families. Perhaps the task of piercing has been taken over by the hypostomal teeth, but if so, it is clear that they would only be able to penetrate thin, weakly sclerotized integuments. A palp with terminal setae of P-5 unmodified (i.e. not claw-like) is a plesiomorphic condition found in all Eylaoidea. Within this superfamily, Limnocharidae, with their palps rather reduced in size, are most similar to Zelandothyadidae, whereas Eylaidae and Piersigiidae have larger, more leg-like palps. An additional similarity with limnocharid mites is provided by the fusion of cheliceral basal segments. The development of rod-shaped, tuberculate sclerites can also be observed in eylaoid mites, with a strikingly similar arrangement in representatives of Limnocharidae, and fusion to form a trabecular frontal plate in Piersigiidae. Similarities to Hydryphantoidea are more numerous, but these are mostly plesiomorphies, such as adult genital field with three pairs of acetabula flanked by lateral flaps (only known for the genus Zelandothyas), gnathosoma with a small mouth opening, not surrounded by a membranous fringe, and the larval characters listed above.
Type species: Zelandothyas diamphida Cook, 1983, by original designation.
Diagnosis — Adults: Mature specimens with a reticulate, large-meshed frontal plate extending from level of lateral eyes to behind level of dorsoglandulare 3, including postocular setae; frontal eye probably absent (see discussion below). Genital field with movable genital flaps partially covering three pairs of acetabula. P-3 with two or more ventral setae.
Deutonymph: Differing from adults in: complete absence of dorsal sclerites (lateral eyes and postocular setae not associated with sclerotized platelets, but see discussion); lower numbers of setae on coxal plates and appendages; coxae without associated apodemes; provisional genital field lacking genital flaps and gonopore, consisting of two pairs of round, shortly stalked acetabula, flanked on each side by a short longitudinal sclerite bearing two setae.
Larva: Dorsum and posterior venter without sclerotized plates. Idiosomal chaetome complete, except for reduced E1 (Ae); setae Ve well developed (chaetome formula 2-2-4-4-4-4-4-4-2-2-2). Lateral eyes with two pairs of lenses lying free in the membranous integument. Coxae separate; one actabulum (“urstigma”) on each side at anterior margin of Cx-II. Anal plate transverse, triangular, bearing only setae E2 (Ai). Gnathosoma rhombic, basal cheliceral segments fused medially. P-4 strongly elongated and distally narrowed, with one proximolateral and two dorsal setae, plus strong, curved distal claws. P-5 inserting in proximal half of P-4, bearing 6 long, pennate setae.
Zelandothyas diamphida Cook 1983: 2–3, Figs 1–9, 11.
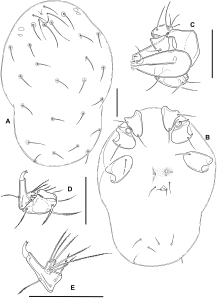


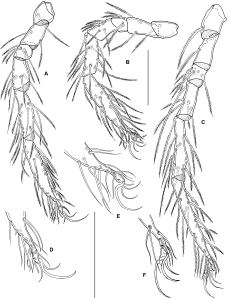




Material examined — Protonymphal skin with larval sclerites (slide 1), undissected deutonymph extracted from protonymphal skin (slide 2), New Zealand, South Island, Whangamoa R. 3 km NE Whangamoa Saddle, 6 Nov. 1982, D.R. Cook DC 43-82 (Canadian National Collection, Ottawa).
Supplementary description — Deutonymph: Integumental papillae with a finely undulating surface (Figure 3D); whole dorsum and posterior part of venter (Figures 3A–B) without sclerotization; lateral eyes and postocular setae not associated with sclerotized plates. Cx-III without the modified medial setae described for the adult female. Gnathosoma and palp as in Figure 3C. Measurements: idiosoma L/W 500/420; maximum L Cx-I 145; Cx-II 140; Cx-III 130; Cx-IV 120; maximum acetabulum diameter 30; gnathosoma L 170; L/H P-1 5/28, P-2 23/21, P-3 25/24, P-4 49/23, P-5 23/6.
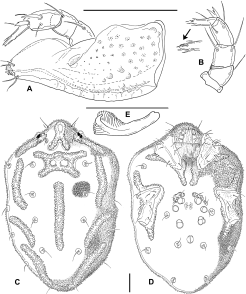
Larva: Dorsum (Figure 1A) without sclerotized plates, setae similar in size and shape, except for Mp-2 (Oi relatively long) and E2 (Ai rather short). Frontal area with two medial pits (a narrow anterior one and a rounded posterior one), one of which probably represents the frontal eye, surrounded by paired setae Lp1, Mp1-2 (Fch, Fp, Oi); lateral eyes not encapsulated, with two thin lenses; Lp2 (Vi) between frontal setal group and lateral eyes, a further pair of setae (Ve) posterior to lateral eyes at lateral idiosomal margin. Central and posterior dorsum with setae Hu, Mh1-4, Lh1-3 and V3 (Oe, Hi, He, Sci, Sce, Li, Le, Si, Se). All coxae subtriangular, with rounded medial margins, separate from each other (Figure 1B); setation: C1-2 on Cx-I, C-3 on Cx-II, C-4 on Cx-III. Shape and setation of leg segments as in Figure 2. Leg claws and empodium slender, empodium distinctly longer than claws. One flatly convex acetabulum (“urstigma”) at anterior margin of each Cx-II; an additional pair of weakly developed protonymphal acetabula centrally on venter between Cx-III. Anal pore on a roughly triangular platelet that is wider than long, flanked by setae E2 (Ai); E1 (Ae) reduced. Setae V1 (Pe) anterolateral to anal platelet, V2 (Pi) and V4 (Ci) in ventrocaudal area. Gnathosoma rhombic in dorsal view (Figure 1C), maximum width at level of palp insertions, rostrum forming an equilateral triangle. Basal cheliceral segments fused medially, claws short and pointed. Palp as in Figures 1D–E. P-1 short, rounded; P-2 with 2 long, pennate setae; P-3 bearing 1 dorsodistal seta; P-4 rather long, gradually narrowed distally, with 3 setae (2 near proximal margin, 1 dorsally in proximal half) and strong, curved distal claws; P-5 inserting in proximal half of P-4, bearing 6 long, pennate setae. Measurements: idiosoma L/W 630/450; maximum L Cx-I 95; Cx-II 100; Cx-III 115; gnathosoma L 140; palp not measured (in oblique position); leg segments 1-6: I-L 45, 40, 30, 60, 95, 150; II-L 55, 45, 30, 48, 85, 148; III-L 65, 45, 35, 65, 115, 185.
Discussion — There is little doubt that the pre-adult stages described here belong to Zelandothyas. They were collected together with adults of Z. diamphida and probably represent this species, sharing with them the shape of the gland pores and leg claws, and the stout palp with the characteristic dorsodistal setae on P-4. Differences between adults and deutonymph, as described in the diagnosis, are not uncommon within water mites: as fast-growing predatory stages, deutonymphs often are characterized by less extensive sclerotization. It should also be borne in mind that the single available deutonymph was extracted from a protonymphal skin. It is thus possible that it was incompletely developed and that older, eclosed deutonymphs might prove to have lateral eye plates and/or a frontal plate. The larval and deutonymphal skins are not in a suitable position for measuring or observing a number of details. In view of their fragility, we decided not to attempt a remounting of the specimens.
(Figure 4)
ZOOBANK: 3AAC087C-BCA8-4198-9F43-39436503EBC8 ![]()


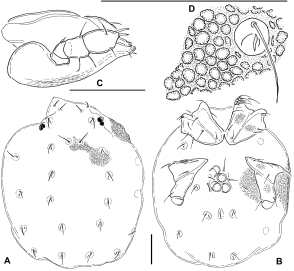
Type series — Holotype male, New Zealand, South Island, Northern part; small affluent of Pelorus River near Middy Hut, about 30 m upstream of confluence, rocks, stones, gravel, dead wood, weak flow; 15 Jan. 1990 leg. A. Ballot; slide-mounted. Paratypes: two females, same site and date, undissected in liquid collection. All deposited in Senckenberg Museum Frankfurt (Schwoerbel collection).
Diagnosis — Frontal plate of mature adults narrow (Figure 4A), consisting of a few trabeculae that surround five meshes (2 unpaired anteriorly, median pair, 1 unpaired posterior); postocular setae located close together on a small sclerite that either extends dorsally from the frontal plate or, when the plate is not developed, lies in the membranous integument. Gnathosoma and its appendages slender, rostrum not set off from gnathosomal base; basal cheliceral segment highest proximally, near insertion of main tracheae; P-2 with four ventral setae, P-4 highest in distal part, L/H > 3.5.
Derivatio nominis — Named after the collector of the species, Andreas Ballot, who at the time was a student of J. Schwoerbel (Univ. Konstanz), but now works as a phycologist at the Norwegian Institute of Water Research (Oslo).
Description — Integumental papillae blunt, typically ribbed and appearing irregularly stellate in apical view (Figure 4B), distributed all over idiosoma except for a medial area between Cx-I, where densely arranged longitudinal folds are developed, which suggests that this part of idiosoma can be inflated (e.g. during ingestion of prey). No modified setae with multiplied tips (as described for Z. diamphida) found at medial margin of Cx-III. Legs with dense, little-differentiated setation. Genital field with pre- and postgenital sclerites not developed; genital flaps porose, bearing 20–30 setae (these a little longer than maximum width of flaps), lateral margin evenly convex, medial margin slightly undulating, bluntly protruding at levels of acetabular interspaces. Acetabula long-oval, on short, porose stalks, gonopore area irregularly papillate. Gnathosoma elongate, with ventral margin nearly straight in anterior part, rostrum not set off from gnathosomal base. Mouth opening small, membranous; 3 fine, ventrally-directed teeth at distal end of hypostomal plate (Figure 4H). Basal cheliceral segment with dorsal and ventral margins slightly converging from insertion of main tracheae to distal edge; distal 2/3 of claws with a line of very fine denticles, weakly sclerotized, ending in fine, flexible tips that are easily bent in all directions (Figure 4F). Palp (Figures 4G, I) with P-1 very short, rounded; P-2 trapezoidal, with very short ventral margin, bearing 1 dorsodistal seta; P-3 with slightly convex dorsal and ventral margins, height greatest near distal end, with 1 dorsodistal seta and a row of 4 setae extending along distal half of ventral margin; P-4 slender, in its distal part with 6 setae (2 fine and long, set off from distal margin ventrally and dorsally, 2 stronger, dorsodistally situated, and 2 finer, short setae, placed mediodistally and laterodistally); P-5 rod-shaped, tip bearing 4 fine, unmodified setae.
Male: Ejaculatory complex (Figures 4D–E) generally of typical hydryphantid form: anterior keel highly elevated and extending to level of proximal arm insertion; posterior keel weakly elevated, not set off from the posterior surface; anterior and posterior arms well developed; proximal horns reduced (this is the only significant departure from ground-plan). Measurements: idiosoma L/W 2000/1400; lateral eye sclerite L 280, frontal plate L/W 435/170; maximum L/W Cx-I+II 290/290, L posterior apodeme 95; maximum L/W Cx-III+IV, 360/190; L anterior apodeme 160, posterior apodeme 50; L/H IV-L-3, 200/70; IV-L-4, 245/65; IV-L-5, 260/65; IV-L-6, 230/82; genital flap L 210; acetabula 1–3 maximum diameters 60/60/50, stalk L 20/30/35; ejaculatory complex L 235, proximal chamber W 80; gnathosoma L 360; chelicera basal segment L/H 215/65, claw L 130; L/H P-1, 10/40; P-2, 35/30; P-3 50/35; P-4, 98/25; P-5, 35/8. Female: Two paratypes differing in development of dorsal and ventral sclerites, probably due to a difference in age. The generally more weakly sclerotized specimen (Figure 4C), with large pores on coxal plates, has a smaller idiosoma, but the dimensions of its sclerites show little difference, suggesting allometric growth of the membranous body. It is further characterized by a complete lack of anterodorsal sclerites, such that the paired lateral eye capsules, as well as the unpaired sclerite bearing postocularia, lie free in the membranous integument. Moreover, the posterior apodeme of Cx-II is developed as only a small, narrow projection, while Cx-III+IV are completely devoid of apodemes. In the larger female, the dorsal sclerites and coxal apodemes are developed as described for the male. The large specimen contains seven round eggs. Measurements – Idiosoma L/W 1600–2800/1200–1800; lateral eye sclerite L 0–500, frontal plate L/W 0–800/0–400; maximum L/W Cx-I+II, 340–350/340, L posterior apodeme 20–270; Cx- III+IV, 410–430/250–290, L anterior apodeme 0–280, posterior apodeme 0–140; genital flap L 215–250; acetabula 1–3 maximum diameter 70/65–70/60–85, stalk L 20/30/35; egg diameter 190; mouthparts not measured. Discussion — The most significant differences separating Z. balloti from the previously described Zelandothyas species (Z. diamphida and Z. hyporheica) are its much less robust palp, the presence of 4 (as opposed to 2) ventral setae on P-2, and the higher L/H ratio of P-4 (3.5, as opposed to <3.0), which is highest towards its distal end (rather than in the middle). It also differs from them in having a more elongate basal cheliceral segment that is highest proximally, near insertion of main tracheae (as opposed to distinctly distad) and a less extended frontal sclerite, with the postocular setae inserting on a common sclerite (rather than in meshes surrounded by the sclerite trabeculae). We suppose that lateral ocular and frontal sclerites develop with increasing age and are therefore highly variable, possibly reaching a species-specific shape only in old adults (see also Smit 1996).
Type species — Malgasacarus rarus Tuzovskij, Gerecke & Goldschmidt, 2008, by original designation.
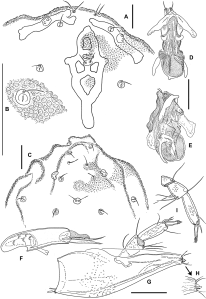
Diagnosis — Deutonymph (adults and larvae unknown): Dorsum (Figure 5C), bearing 4 rod-shaped lateral plates and 3 unpaired medial plates: (1) an inverse-Y-shaped frontal plate; (2) a transverse anterodorsal plate, consisting of a transverse bar with a dorsomedial projection bearing the unpaired frontal eye and paired postocular setae, this plate posteriorly with weakly developed, paired, medial projections or connected by three narrow extensions to a further transverse bar; (3) a longitudinal, rod-shaped posterodorsal plate. Trichobothria Fp sensu Tuzovskij et al. 2008 (preocularia) and all glandularia free in the membranous integument, not associated with platelets, glandular openings slit-shaped. Venter (Figure 5D) with Cx-II separate from Cx-III, not meeting in midline. Extended apodemes posteriorly and laterally on Cx-I+II, anteriorly and posteriorly on Cx-III+IV; these apodemes, as well as the secondary sclerotization along medial margins of Cx-III+IV, covered by tubercles as described for dorsal plates. Cx-I unfused medially. Coxal setae not numerous (Cx-I, 9, Cx-II, 8-9, Cx-III, 5-6, Cx-IV, 3). Provisional genital field between Cx-III+IV, with 3 pairs of short-stalked, round acetabula and 4 pairs of short setae lying on small sclerites anterolaterad of acetabulum 1; gonopore absent. Excretory pore smooth. Gnathosoma (Figures 5A, D) connected to idiosoma by a membranous tube, retractable into the membranous area between medial margins of Cx-I; gnathosomal base slender and elongate, rostrum moderately developed, hypostome bearing several fine, pointed teeth at anterior edge. Basal cheliceral segments fused to each other medially, claws long and stylet-shaped, with rather numerous ventral teeth; palps (Figures 5A–B) five-segmented, stout, neither chelate nor uncate, P-4 with two strong dorsodistal setae, P-5 bearing 7 fine setae, 4 of which near tip of segment but not claw-shaped. Leg segments bearing thin, weakly differentiated setae, mostly finely pennate, but some (mostly on dorsal segment surfaces and on terminal segments) are finer and simple; no swimming hairs developed; leg claws with one main tip, flanked on both sides by fine clawlets which are antiaxially lower in number and restricted to distal part, paraxially higher in number, forming a pecten that extends over more than half of the medial surface.
Discussion — The specimen on which the definition of this family was based is not an adult as stated by Tuzovskij et al. (2008), but a deutonymph (see below). Consequently, a comparison of diagnostic features is only possible with deutonymphs of potential sister taxa.
Zelandothyas deutonymphs differ from those of Malgasacarus mostly in the morphology of the genital field (only two pairs of acetabula) and P-2 (bearing ventral setae). The significance of a further important complex of characters – the development of dorsal plates and coxal apodemes – cannot be addressed given the present state of knowledge. As known from representatives of many other water mite families, and as observed in females of Z. balloti, these sclerites are not yet present immediately after hatching and develop during the growth of an individual. We would expect that small lateral eye- and frontal-sclerites would be developed in fully-formed Zelandothyas deutonymphs.
Although adults of Malgasacarus are not yet known, we hypothesize that they will prove to be multiacetabulate, since the number of acetabula is generally higher in adults than in deutonymphs. If so, this would provide an additional difference from Zelandothyas.
Based on the data now available, we find a high morphological agreement between Malgasacarus and Zelandothyas, making it clear that we are dealing with a sister group relationship between them.
Malgasacarus rarus Tuzovskij, Gerecke & Goldschmidt 2008: 164, Figs 1–16.


Material examined — Holotype deutonymph, Madagascar, Ranomena (Fianarantsoa), spring area of stream that intersects railway NW of tunnel 18, 21°29'45.9"S, 047°24'07.5"E, 1017 m a.s.l., 21 Aug. 2001, leg. R. Gerecke & T. Goldschmidt, collecting site 43a (Senckenberg Museum Frankfurt; not deposited in Gerecke coll. as stated by Tuzovskij et al. (2008)); one deutonymph, Madagascar, Madiorano (Fianarantsoa), stream that intersects railway W of tunnel 18, at railroad km 51.2, 21°31'32.2"'S, 047°26'05.3"E, 650 m a.s.l., riffle, 18 Aug. 2001, leg. R. Gerecke & T. Goldschmidt, collecting site 31a (Senckenberg Museum Frankfurt).
Redescription (mainly based on non-type deutonymph 31a; characters given in subfamilial diagnosis not repeated) – Idiosoma slightly flattened, pointed anteriorly and posteriorly, with prominent shoulders (maximum width in distal quarter); integumental papillae conical and with ridges like a limpet shell (appearing stellate in apical view). All dorsal plates (Figure 5C) without porosity, but densely covered with fine tubercles. Anterodorsal plate consisting of two transverse bars – a larger anterior and a shorter posterior one – connected by three rungs; eye-bearing lateral plates straight, all other lateral plates slightly curved, with medial margin concave on second, but slightly convex on third and fourth plates.
All coxae with extended, densely tuberculate, secondary sclerotization, forming apodeme-like extensions posteromedially and laterally on Cx-I+II, and anteriorly and posteriorly on Cx-III+IV (Figure 5D); Cx-I separated medially by a membranous area with longitudinal folding that extends anteriorly and covers the gnathosomal insertion ventrally. Provisional genital field consisting of paired platelets (that on left side subdivided into a larger anterior and a smaller posterior platelet) bearing four setae on each side and three pairs of round, short-stalked acetabula arranged around the (closed) gonopore rudiment; acetabulum 3 slightly larger than others, longitudinally split on left side into two parts on a common stalk. Excretory pore surrounded by a circular area of smooth integument, not associated with sclerotized parts.
Gnathosoma (Figure 5A) with rostrum moderately developed; hypostome with a short anterior projection bearing a group of ventrally-directed teeth. Palp (Figures 5A–B): P-1 short, without setae; P-2 convex dorsally and ventrally, bearing a dorsomedial seta near distal end; P-3 subquadrate, bearing 2 distal setae, 1 ventromedially and 1 dorsolaterally; P-4 with slightly convex dorsal and ventral margins, bearing in distal part 1 thin seta medially and 3 setae dorsolaterally, 1 of which is long and the other 2 strong and serrate; P-5 thin, relatively long, slightly narrowed distally, bearing a solenidion near the middle and 6 short setae at the tip.
Legs with uniform setation; claws pointed, with numerous lateral clawlets, arranged in two rows, a long, pecten-like paraxial row, and a shorter antiaxial row.
Measurements — Idiosoma L/W 900/620; frontal plate L/W 180/117; anterodorsal plate L 90, anterior W 250, posterior W 200; posterodorsal plate L 315; Lateral plates 1-4 L 200, 280, 225, 290; C-I+II L/W 265/247; Cx-III+IV L/W 324/220; maximum acetabula diameter ca. 30; gnathosoma L 148; L/H P-1, 13/58; P-2, 54/45; P-3, 45/45; P-4, 78/38; P-5, 67/18; basal chelicera segment L/H 110/36, claw L 74; leg segments 1-6 L: I-L: 25, 78, 85, 112, 127, 134; II-L: 27, 72, 81, 112, 127, 134; III-L: 27, 67, 72, 112, 133, 133; IV-L: 65, 94, 108, 156, 161, 145.
Habitat and distribution – The two known specimens were collected in rather different habitats: a hygropetric area near a large cascade of a middle-order stream and a swampy helocrene. Intensive fieldwork carried out at the two sites and their surroundings in April 2011 did not produce any further specimens. The precise habitat preferences of this species therefore remain enigmatic.
Discussion — During the initial stages of this investigation we hypothesized that unusual larval specimens found parasitizing water bugs in northern Madagascar (Koenike, 1898, Gerecke 2004) might represent the larval stage of Malgasacarus. Meanwhile, those larvae have been investigated in detail and found to belong to the superfamily Eylaoidea. Their characteristics include a well-developed dorsal and ventral sclerotization, specialized setae anteriorly on Cx-I, presence of trichobothria on leg telofemora, and unique modifications of the gnathosoma not found in larvae of any known water mite taxon (unpubl. obs.). Because the larva of Zelandothyas described here differs strongly from these larvae in most respects, and in view of the considerable similarity between deutonymphs of Zelandothyas and Malgasacarus, we consider our initial hypothesis to be false.
The second specimen of Malgasacarus rarus reported here differs from the holotype in having distinctly smaller general dimensions, with the idiosoma being about 30% and the idiosomal sclerites about 20 % shorter. Four important morphological differences between the two specimens were revealed by the revision of the holotype, in which (1) distinct integument papillosity is found only in restricted areas – over large parts the papillae are flattened, giving the integument a reticulate aspect; (2) the frontal sclerite is distinctly more slender and has less divergent posterior extensions; (3) the anterodorsal sclerite lacks a posterior bar and, consequently, the three transversal extensions; and (4) the gnathosoma is stouter, with a shorter rostrum. In view of these morphological differences and the different habitats in which the two specimens were collected, the possibility that two species might be involved cannot be excluded.
Two apparent differences from the original description are due to observational error or misinterpretation: (1) the genital field does not include the pre- and postgenital sclerites or a gonopore (it is closed, as is typical for deutonymphs); (2) the leg claws are not longitudinally divided into a dorsal and a ventral branch. In dorsoventral view it is evident that the clawlets of the shorter lateral row could suggest a longitudinal bifurcation. In fact, the shape of the claws of Malgasacarus agrees well with that described for Zelandothyas diamphida by Cook (1983) (Figure 5E).
A previous study of larval water mites from Madagascar deposited at MNHN Paris by the senior author was made possible by a grant from the EU SYNTHESYS programme. Mites collected by A. Ballot from New Zealand in 1990 were discovered during a project dedicated to the conservation of the J. Schwoerbel collection (Senckenberg Museum, Frankfurt), financed by the Deutsche Forschungsgemeinschaft (DFG). The DFG also provided financial support for a collecting trip in 2011 by the senior author to the area of the type locality of Malgasacarus rarus, carried out in cooperation with F. Rakotondraparany, R. Oliarinony (Univ. Antananarivo) and J. Ranaivo (Centre Vahatra, Antananarivo). Permits for field work and exportation were provided in 2001 and 2011 by the ministry of environment of Madagascar, in cooperation with Dr F. Rakotondraparany. Specimens were borrowed from Senckenberg Museum, Frankfurt, through the courtesy of Peter Jäger and Julia Altmann, and from Museum Victoria, Melbourne, through the courtesy of Simon Hinkley.
Cook D.R. 1974 — Water mite genera and subgenera — Mem. Amer. Ent. Inst., 21: VII + 1-860.
Cook D.R. 1983 — Rheophilic and hyporheic water mites of New Zealand — Contr. Amer. Ent. Inst., 21: I–II, 1-224.
Cook D.R. 1986 — Water mites from Australia — Mem. Amer. Ent. Inst., 40: I–IV, 1-568.
Dabert M., Proctor H., Dabert J. 2016 — Higher-level molecular phylogeny of the water mites (Acariformes: Prostigmata: Parasitengonina: Hydrachnidiae) — Mol. Phylogenet. Evol., 101: 75-90. doi:10.1016/j.ympev.2016.05.004 ![]()
Gerecke R. 2004 — The water mites of Madagascar (Acari: Hydrachnidia): a revised list completed by original material conserved at the Muséum national d'Histoire naturelle, Paris — Zoosystema, 26: 393-418.
Harvey M.S. 1998 — The Australian water mites: a guide to families and genera — In: Veroni, M. (ed.): Monographs on invertebrate taxonomy, vol. 4, CSIRO Publ., Collingwood, Australia: 1-150.
Harvey M.S, Growns J. 1998 — A guide to the identification of families of Australian water mites (Arachnida: Acarina) — Cooperative Research Centre for Freshwater Ecology, Identification Guide No. 18. Albury, Australia: Murray-Darling Freshwater Research Centre.
Koenike F. 1898 — Hydrachniden-Fauna von Madagaskar und Nossi-Bé — Abh. Senckenberg. naturf. Ges., Frankfurt/M., 21: 295-435.
Smit H. 1996 — New species of water mites from New Zealand, with remarks on the water mites from ponds and lakes (Acari, Hydrachnellae) — Acarologia, 37(1): 45-53.
Smith I.M., Cook D.R. 1991 — Water Mites — pp. 523-592 in: Thorp J., Covich A. (eds) Ecology and Classification of North American Freshwater Invertebrates. Academic Press, San Diego.
Tuzovskij P.V. 1987 — [Morphology and postembryonic development of water mites] — Moscow: Nauka, 1-176 (in Russian).
Tuzovskij P., Gerecke R., Goldschmidt T. 2008 — Malgasacaridae, a new family of water mites from Madagascar (Acariformes, Hydrachnidia) — Zoosyst. Ross., 16[2007]: 163-167.



2016-11-28
Date accepted:
2017-01-12
Date published:
2017-05-17
Edited by:
Auger, Philippe

This work is licensed under a Creative Commons Attribution 4.0 International License
2017 Gerecke, Reinhard; Judson, Mark L.I. and Cook, David R.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



