Occurrence of the mite Ophionyssus natricis (Acari: Macronyssidae) on captive snakes from Panama
Miranda, Roberto J.1 ; Cleghorn, John E.2 ; Bermudez, Sergio E.3 and Perotti, Maria A.4
1✉ Departamento de Investigación en Entomología Médica, Instituto Conmemorativo Gorgas de Estudios de la Salud, Panamá. Grupo de Estudios con Ectoparásitos.
2Escuela de Biología, Universidad de Panamá. Grupo de Estudios con Ectoparásitos.
3Departamento de Investigación en Entomología Médica, Instituto Conmemorativo Gorgas de Estudios de la Salud, Panamá. Grupo de Estudios con Ectoparásitos.
4Evolutionary Biology and Ecology Section, School of Biological Sciences, University of Reading, United Kingdom.
2017 - Volume: 57 Issue: 2 pages: 365-368
https://doi.org/10.1051/acarologia/20164161Keywords
Abstract
The exotic pet trade has reached worldwide proportions and involves a variety of wild animals from invertebrates to vertebrates. The movement of these exotic pets incorporates the translocation of many zoonotic diseases and vectors, most of which are still poorly studied and understood. Invertebrates, either pets or vectors are perhaps the most understudied. Chelicerates are a good example, with a huge body of literature lecturing on ticks (Ixodida) but very little on small parasitic mites of exotic pets (Masan et al., 2012). Ophionyssus natricis (Gervais, 1844) is the most widespread mite-pest of captive reptiles, particularly snakes (Squamata). This preference for reptiles seems to be characteristic of the genus Ophionyssus. Of the 16 species of this genus, 15 have been reported as parasites of reptiles, with only one species described from a mammal (Moraza et al., 2009).
Bites of O. natricis cause damages on the skin of captive reptiles, and if the level of infestation is high enough or out of control, it will consequently lead to irritation, anemia and even death (Beck and Pantchev, 2006, Hoppman and Wilson, 2007, Rataj et al., 2011). In addition, this mite is able to transmit pathogens as hemogregarines, Aeromonas spp., being the mechanical vector of hemorrhagic septicemia, caused by the motile anaerobic bacillus Aeromonas hydrophila, and responsible of or associated with the still under investigation "Inclusion Body Disease" (Camin, 1948; Yunker, 1956; Chang and Jacobson, 2010; Mariana et al., 2011). O. natricis is of great concern to zoos, pet shops and private collections of reptiles, especially due to its ability to spread fast from a single parasitized animal to others in the same enclosure or nearby (Rodríguez and Lazcano, 1992). Despite being pests of specific reptile hosts, O. natricis can opportunistically move to other hosts, like other blood sucking mites do, particularly in the domestic environment, e.g. like those in the families Macronyssidae, Dermanyssidae and Laelapidae (O'Donel Alexander, 1984), and whenever there is a shortage of food or the infestation is too severe, implying high competition for resources, the mites are able to affect humans, such as pet owners, keepers and handlers of infested snakes (Hoppman and Wilson, 2007; Rataj et al., 2011; Amanatfard et al., 2014). Ophionyssus natricis was first described from native European snakes and later became associated with captive reptiles of different biogeographical regions (Fain, 1962; Domrow, 1985; Fain and Bannert, 2000; Paredes-León et al., 2008), with just a handful of papers mentioning infestation of wild snakes (Yunker, 1956, Simonov and Zinchenko, 2010). To date, O. natricis have been reported in Africa (Yunker, 1956), Europe (Beck and Pantchev, 2006; Simonov and Zinchenko, 2010), Oceania (Domrow, 1985), Asia (Mariana et al., 2011), and America (Schroeder, 1934; Camin, 1948). Specifically for the Neotropical region, O. natricis has been only reported from Mexico (Rodríguez and Lazcano, 1992; Paredes-León et al., 2008) and Nicaragua (Rimbaud et al., 2006).
This is the first report documenting O. natricis on captive snakes from the City of Panama, Panama. Occurrence of mites was recorded between May and June 2015, from homes that kept reptiles as pets, captive in small micro-cosmos. The findings included five species of snake-hosts: Boa constrictor Linnaeus, 1758, Epicrates maurus Gray 1849, Corallus ruschenbergerii (Cope, 1876), Corallus caninus (Linnaeus, 1758) and Python regius (Shaw, 1802). The snakes had been living in captivity in their respective homes for several months (9-48 months). In all cases they were maintained in individual terrariums constructed with wooden frames, glass walls, and with wood paper as substrate. The owners indicated that the snakes were more aggressive than usual, and any severe skin damage was observed and recorded. After collection of mites from the snakes, all snakes and terrariums were treated with Fipronex® and the success of the control became evident days later.
The ectoparasites were removed manually by one of the authors (JEC), and preserved by placing them in vials with 70% ethanol. Once in the laboratory, a sample of 30 mites (randomly taken from the 5 snakes) was treated with 10% NaOH and mounted using Hoyer's medium. Species identification followed the key of Moraza et al. (2009). The slides were deposited into the "Dr. Eustorgio Mendez" Zoological Collection of the Gorgas Memorial Institute.
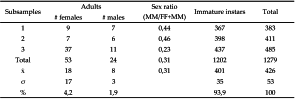


The number of mites collected on E. maurus, C. ruschenbergerii, C. caninus and P. regius was between 10-50 specimens. Remarkable was the infestation over Boa constrictor and its terrarium, estimating that there were approximately 4000 individuals, estimation based on three subsamples of 2 ml each (Table 1). Most specimens were immature stages and adults (females) (Figure 1). Empty puparia of Phoridae flies and some individuals of Glycycometus malaysiensis (Fain and Nadchatram, 1980) (Aeroglyphidae) were also present in this sample.
eginfigure[H]% figure1 entering ncludegraphics[width=argeurfigureunecolonne]../Articles/00436-Miranda/Figure1.pdf aptionHabitus of Ophionyssus natricis female, collected from Boa constrictor terrarium. Scale bar = 0.35 mm.ndfigure
Previous reports of mites parasitizing pet-reptiles in Panama include a tick species close to Amblyomma flavomaculatum on Varanus exanthematicus (Bosc, 1792) (Bermúdez and Miranda, 2011), the pterygosomatid mite Geckobiella stamii Hirst, 1917 on Iguana iguana (L. 1758) (Murgas et al., 2013) and the argasid tick Ornithodoros puertoricensis Fox 1947 on Varanidae and Pythonidae (Bermúdez et al., 2015).
The predominance of females (Table 1, sex ratio) and immature stages was noted earlier by Domrow (1985) and Mariana et al. (2011), and these two works emphasised that a single female could generate a large offspring. In fact, a female bias is the canon for haplodiploid species and O. natricis is one of them, this is also a common feature of many Macronyssidae. They reproduce by arrhenotokous parthenogenesis, where females are diploid and males are the result of unfertilized eggs (Oliver, 1966). Its nidicolous behavior corresponds well with acarine haplodiploid clades having adaptations to live in patchy or ephemeral environments (Perotti and Braig, 2009).
This mite species must be considered a risk to humans, pet owners or keepers, in addition to reptiles, due to the stress caused by the implicit bites and further infection (Schultz, 1975, Rataj et al., 2011, Amanatfard et al., 2014). There is also an extra risk for humans handling animals, especially if the snakes are not normally aggressive, because this behavior increases with high infestations of O. natricis (Amanatfard et al., 2014).
Finally, the infestations of captive animals by O. natricis should be considered as a serious risk in the trade business and maintenance procedures of exotic reptiles, and efforts should focus on keeping captive animals under periodic health checks and under treatment. The application of biological control methods including the use of predatory mites such as Laelapidae and/or Cheyletus (Schillinger et al., 2013) or the applications of low toxicity acaricide compounds (Rodríguez and Lazcano, 1992) have proven to be successful and affordable to keep captive pets in good health.
We are grateful to Lionel Schillinger (Clinique vétérinaire du village d'Auteuil, France) for providing literature.
Beck W., Pantchev N. 2006 — Schlangenmilbenbefall (Ophionyssus natricis) beim Grünen Leguan (Iguana iguana)- Ein Fallbericht — Kleintierpraxis, 51, 3-7.
Bermúdez S., Miranda R. 2011 — De mascotas exóticas y turistas: nuevas oportunidades para la introducción de ectoparásitos en Panamá — Bol. SEA, 48, 491-492.
Bermúdez S., Miranda R., Cleghorn J., Venzal J. 2015 — Ornithodoros (Alectorobius) puertoricensis (Ixodida: Argasidae) parasitizing exotic reptiles pets in Panama— Rev. FAVE- Cienc. Vet., 14, 1-5.
Camin J. 1948 — Mite transmission of a hemorrhagic septicemia in snakes — J. Parasitol, 34, 345-354.
Chang L.-W., Jacobson E.R. 2010 — Inclusion body disease, a worldwide infectious disease of boid snakes: A review — J. Exot. Pet Med., 19, 216-225.
Domrow R. 1985 — Species of Ophionyssus Megnin from Australian lizards and snakes (Acari: Dermanyssidae) — J. Aust. Entomol. Soc., 24, 149-153.
Fain A. 1962 — Les acariens mesostigmatiques ectoparasites des serpentes — Bull. Inst. Roy. Sci. Nat. Belgique, 38, 1-149.
Fain A., Bannert B. 2000 — Two new species of Ophionyssus Mégnin (Acari: Macronyssidae) parasitic on lizards of the genus Gallotia Boulenger (Reptilia: Lacertidae) from the Canary Islands — Int. J. Acarol, 26, 41-50.
Hoppman E., Wilson H. 2007 — Dermatology in Reptiles — J. Exot. Pet Med., 16, 210-224.
Mariana A., Vellayan S., Halimaton I., Ho T.M. 2011 — Acariasis on pet Burmese python, Python molurus bivittatus in Malaysia —Asian Pac. J. Trop. Med., 2011, 4, 227-228.
Masan P., Simpson C., Perotti M.A., Braig H.R. 2012 — Mites parasitic on Australasian and African spiders found in the pet trade; a redescription of Ljunghia pulleinei Womersley — PLoS ONE, 7, e39019.
Moraza M., Irwin N., Godinho R., Baird S.J.E., De Bellocq J.G. 2009 — A new species of Ophionyssus Mégnin (Acari: Mesostigmata: Macronyssidae) parasitic on Lacerta schreiberi Bedriaga (Reptilia: Lacertidae) from the Iberian Peninsula, and a world key to species — Zootaxa, 2007, 58-68.
Murgas D., Dutary S., Miranda R. 2013 — First report of Geckobiella stamii (Acari: Pterygosomatidae) parasitizing Iguana iguana (Squamata: Iguanidae) in Panama — Rev. Iber. Aracnol, 22, 97-98.
O'Donel Alexander J. 1984 — Arthropods and human skin — Berlin, Springer-Verlag.
Oliver J.H.J. 1966 — Notes on Reproductive Behavior in the Dermanyssidae (Acarina: Mesostigmata) — J. Med. Entomol., 29, 29-35.
Paredes-León R., García-Prieto L., Guzmán-Cornejo C., León-Regagnon V., Pérez T.M. 2008 — Metazoan parasites of Mexican amphibians and reptiles — Zootaxa, 1904, 1-166.
Perotti M.A., Braig H.R. 2009 — Phoretic mites associated with animal and human decomposition — Exp. Appl. Acarol., 49, 85-124.
Rataj A.V., Lindner-Knific R., Vlahovic K., Mavri U., Dovc A. 2011 — Parasites in pet reptiles — Acta Vet. Scand., 53, 1-20.
Rimbaud E., Pineda N., Luna L., Zepeda N., Rivera G. 2006 — Primer reporte de Ophionyssus natricis (Arthropoda, Acarina, Macronyssidae, Gervais 1953) parasitando Boa constrictor constrictor en Nicaragua — Bol. Parasitol. Esc. Med. Vet. Univ. Nac. Costa Rica, 7, 1.
Rodríguez M.L., Lazcano D. 1992 — First report of the mite Ophionyssus natricis (Acarina: Macronyssidae) from Mexico — Southwestern Nat., 37, 426.
Schillinger L.H., Morel D., Bonwitt J.H., Marquis O. 2013 — Cheyletus eruditus (Taurrus®): an effective candidate for the biological control of the snake mite (Ophionyssus natricis) — J. Wildl. Dis., 44, 654-659.
Schroeder C.R. 1934 — The snake mite (Ophionyssus serpentium Hirst) — J. Econ. Entomol., 28, 1004-1014.
Schultz H. 1975 — Human infestation by Ophionyssus natricis snake mites — Br. J. Dermatol., 93, 695-697.
Simonov E., Zinchenko V. 2010 — Intensive infestation of Siberian pit-viper, Gloydius halys halys by the common snake mite, Ophionyssus natricis — North West J. Zool., 6, 134-137.
Yunker C. 1956 — Studies on the snake mite, Ophionyssus natricis, in nature — Science, 124, 979-980.



2016-09-02
Date accepted:
2016-10-03
Date published:
2017-04-20
Edited by:
Bertrand, Michel

This work is licensed under a Creative Commons Attribution 4.0 International License
2017 Miranda, Roberto J.; Cleghorn, John E.; Bermudez, Sergio E. and Perotti, Maria A.
Download the citation
RIS with abstract
(Zotero, Endnote, Reference Manager, ProCite, RefWorks, Mendeley)
RIS without abstract
BIB
(Zotero, BibTeX)
TXT
(PubMed, Txt)



